Grantee Research Project Results
2010 Progress Report: Toxicological Evaluation of Realistic Emission Source Aerosol (TERESA): Investigation of Vehicular Emissions
EPA Grant Number: R832416C005Subproject: this is subproject number 005 , established and managed by the Center Director under grant R832416
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Health Effects Institute (2015 - 2020)
Center Director: Greenbaum, Daniel S.
Title: Toxicological Evaluation of Realistic Emission Source Aerosol (TERESA): Investigation of Vehicular Emissions
Investigators: Koutrakis, Petros , Godleski, John J.
Institution: Harvard University
EPA Project Officer: Chung, Serena
Project Period: October 1, 2005 through September 30, 2010 (Extended to September 30, 2011)
Project Period Covered by this Report: August 1, 2009 through July 31,2010
RFA: Particulate Matter Research Centers (2004) RFA Text | Recipients Lists
Research Category: Human Health , Air
Objective:
Because particulate and gaseous source emissions undergo many transformations once released into the atmosphere, it is likely that secondary and primary pollutants exhibit different toxicities. Most of the source-specific toxicity studies to date have focused on primary pollutants; therefore, there is a great need to investigate the relative toxicity of source-specific primary and secondary particles. We recently have probed directly into this question with the Toxicological Evaluation of Realistic Emission Source Aerosol (TERESA) Power Plant study, a research project funded by the Electric Power Research Institute (EPRI) and the previous Harvard EPA PM Center. This study was designed to investigate the relative toxicity of primary and secondary particulate emissions from coal-fired power plants, in situ, and to explore the relationship between secondary particle formation processes and particle toxicity. As part of the TERESA study, we have developed techniques and facilities to sample source emissions, form secondary particles inside a photochemical chamber, and expose animals to both primary and secondary particles (Ruiz, et al., 2006; Ruiz, et al., 2007a, b; Kang, et al., 2010). The TERESA approach forms an excellent foundation for future research, as it can be adapted readily to investigate other combustion sources. Using the developed technologies, this project extends this research to the toxicological investigation of primary and secondary pollutants from vehicular (mobile source) emissions released from the ventilation stack of a large roadway tunnel within the northeastern United States.
During the last year of the Center, we installed and optimized an exposure system that consists of (i) a sampling system to extract motor vehicle primary emissions from the plenum, (ii) a new, large photochemical chamber that can be used to form secondary organic aerosol from primary vehicular emissions and (iii) an improved output counter-current parallel-plate membrane diffusion denuder that has high removal efficiencies for both unreactive gases such as carbon monoxide (CO) and nitric oxide (NO), as well as for vehicular volatile organic compounds (VOCs) with relatively low diffusion coefficients. During the last year, this exposure system has been used to conduct animal exposures to primary particle emissions, aged primary plus secondary organic particles, and secondary organic aerosol only. In June 2010, the exposure system and preliminary results were presented at the meeting of the EPA PM Center Scientific Advisory Committee in Boston.
The specific hypotheses of this project are:
- Exposures to fresh and to photochemically oxidized mobile source emissions will induce cardiovascular responses in normal animals;
- Atmospheric photochemical processes enhance the toxicity of gases and particles emitted from motor vehicles; and
- Animal models of susceptible populations (e.g., spontaneously hypertensive rats) will have greater biological responses to particles originating from motor vehicles than the corresponding normal animal model.
Progress Summary:
Field study setup using an urban highway tunnel to provide mobile source emissions. Progressing from our laboratory development studies using the diluted exhaust of a single vehicle to a field study has involved several steps. Site preparation was extensive, and included installation of electric service, creation of secure areas for equipment and instrumentation, design and construction of a new large photochemical chamber and enclosure for the UV lamps, and design and construction of a sampling system to transport primary emissions from the plenum of the tunnel to the photochemical chamber.
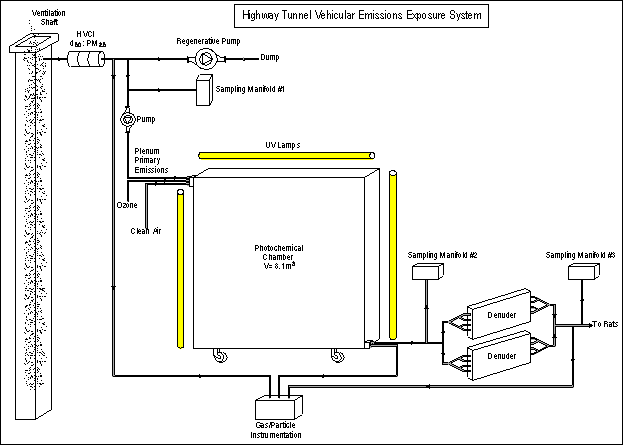
The exposure generation system is shown schematically below in Figure 1. The three main components are the primary emissions sampling system, the photochemical reaction chamber, and the parallel plate membrane denuder.
Figure 1. Exposure Generation System
The primary emissions sampling system consists of approximately 50 feet of a large diameter (2”) antistatic tube extending into the plenum, a size selective inlet (SSI) to remove particles larger than 2.5 μm, and a blower that pulls air from the plenum through the SSI. To keep particle losses to a minimum, the flow rate through the tube is 300 LPM; the SSI is a high volume cascade impactor (HVCI) modified to provide a 2.5 μm size cut at 300 LPM. A tee between the SSI and blower is connected to (i) a manifold for upstream sampling of vehicle exhaust, and (ii) a vacuum pump that is used to introduce a constant flow into the chamber.
The chamber is a well-mixed-flow reactor that is operated in a dynamic flow mode. It is a cube of approximately 8 m3 volume. Three surfaces (two sides and the top) are covered with 2 mil (0.05 mm) thick fluorinated ethylene propylene (FEP) Teflon film. The other three surfaces (front, back, and bottom) are constructed of aluminum coated with polyfluorinated alkene (PFA) Teflon. The photochemical chamber is housed within a ventilated enclosure with UV lamps mounted on both sides and the ceiling. A total of 180 UV lamps, mostly UV-350 nm fluorescent 40W lamps, provide a good representation of the ground-level solar spectrum. Also, 5% of the lamps are UVB313 nm fluorescent 40W lamps, used to enhance hydroxyl radical (·OH) production.
To produce secondary organic aerosol, direct tunnel primary vehicular emissions are diluted with clean air inside the chamber. Sufficient ozone is added to titrate the baseline nitric oxide concentrations in the chamber and expedite the initiation of the gas photochemical reactions. To maximize the secondary organic aerosol yield, and also to have adequate chamber flow output for animal exposures and particle characterization, the chamber performance is optimized by varying the dilution factor and chamber residence time.
Two high-flow, counter-current, parallel-plate membrane diffusion denuders are used in parallel with the system, to achieve high removal efficiencies for excess primary and secondary gases from the chamber output, including ozone and unreactive gases such as CO and NO, as well as VOCs with relatively low diffusion coefficients. These larger denuders provide enough sample flow to allow for both toxicological exposures and characterization of test aerosols. Each denuder is operated at a sample flow (QS) =16.7 L/min, while each purge channel flow (QP) = 30 L/min (two purge channels [60 L/min] per denuder). Wilkerson cylinders containing Hydrosil (potassium permanganate-coated molecular sieve) and activated carbon, followed by a HEPA filter are used to provide the zero-air required for each purge flow. Purge flows are measured using calibrated rotameters, and the absence of net convective flow across the membranes is verified by maintaining zero differential pressure at the midpoint of the membrane.
Figure 1 above also shows the three locations where sampling/monitoring manifolds are located: upstream of the chamber between the SSI and blower, downstream of the photochemical chamber between the chamber outlet and denuders, and downstream of the denuders at the point of exposure. A variety of continuous and integrated methods were used to characterize vehicular emissions, chamber performance, and animal exposures. These methods are listed below.
Sampling and Monitoring Methods
Integrated Sampling:
- Particle mass and elements on 47 mm Teflon membrane filters using PFA Teflon filter holders, by gravimetric analysis and X-ray fluorescence (XRF), respectively.
- EC/OC on 47 mm pre-fired quartz filters by thermal optical reflectance (TOR).
- Water soluble OC (Stone, et al., 2008) on 47 mm pre-fired quartz fiber filters.
- Aldehydes on DNPH cartridges by HPLC.
Continuous/Semi-continuous Monitoring:
- Continuous particle size distribution (15 nm to 500 nm particle electric mobility diameter) of secondary particles using a Scanning Mobility Particle Sizer (SMPS Model 3934, TSI Inc., Shoreview, MN).
- Continuous particle size distribution (500 nm to 20 µm particle aerodynamic diameter) of secondary particles using an Aerodynamic Particle Sizer (APS 3321, TSI Inc., Shoreview, MN).
- Continuous particle total number concentration using a Condensation Particle Counter (Model 3007 CPC, TSI Inc., Shoreview, MN).
- Continuous CO (Model 48C CO Analyzer, Thermo Environmental Instruments, Inc., Franklin, MA).
- Continuous NOx by chemiluminescence (Model 42C NO-NOx Analyzer, Thermo Environmental Instruments, Inc., Franklin, MA).
- Continuous O3 by UV photometry (Model 49C O3 Analyzer, Thermo Environmental Instruments, Inc., Franklin, MA).
- Continuous SO2 by pulsed fluorescence (Model 43C SO2 Analyzer, Thermo Environmental Instruments, Inc., Franklin, MA).
- Elemental and organic carbon (EC/OC) using an OCEC Carbon Aerosol Instrument (Model 3F Semi-Continuous Field Analyzer, Sunset Laboratory, Tigard, OR).
Exposure Atmosphere Generation. The exposure generation system described above is being used to conduct animal exposures to three types of atmospheres, as described below.
Primary mobile source emissions. Tunnel effluent introduced into the chamber contains both primary particles smaller than 2.5 µm in aerodynamic diameter and primary gases. No lamps are turned on for this atmosphere simulation (and no ozone is added to titrate NO). The chamber output passes through the two-denuder system to remove the primary gases and have the animals exposed only to primary particles from the plenum.
Aged plume. The tunnel effluent containing primary gases and particles is introduced into the chamber. Sufficient ozone is added to titrate the nitric oxide. The lamps are turned on and the emissions are photochemically oxidized to generate a mixture of aged primary particles and SOA. The chamber output passes through the two-denuder system to remove the excess primary and secondary gases and animals are exposed only to aged primary particles plus SOA.
Secondary organic aerosol only. The tunnel effluent is filtered to remove primary particles and introduced into the chamber with sufficient ozone to titrate the nitric oxide. The lamps are turned on and the emissions are photochemically oxidized to form SOA. The chamber output passes through the two-denuder system to remove the excess primary and secondary gases, and animals are exposed to SOA.
The toxicological testing in this study is performed using consistent mass concentrations for each exposure and each of the three exposure atmospheres. Each exposure is 5 hours long, and two exposures per day are conducted 4 days per week for 3 consecutive weeks. To accommodate two 5-hour exposures with characterization, it is necessary to operate the chamber in a dynamic rather than a batch mode.
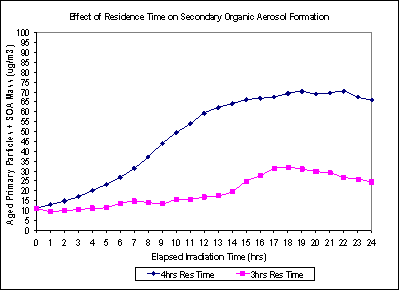
Our primary goal was to maximize the formation of SOA for the Aged Plume (aged primary plus secondary) and SOA Only exposures. We also wanted to minimize the time required to generate a stable chamber output. To do this, we add just enough ozone to the chamber to titrate the NO to NO2 and have a small excess of O3 before turning on the lights. We found that a longer residence time gave a higher SOA yield, Figure 2.
Figure 2. Effect of residence time on SOA formation. 35 LPM (4 hrs Res Time) and 45 LPM (3 hrs Res Time) with the SOA formed as a function of irradiation time from 0 (lights on) to 24 hrs.
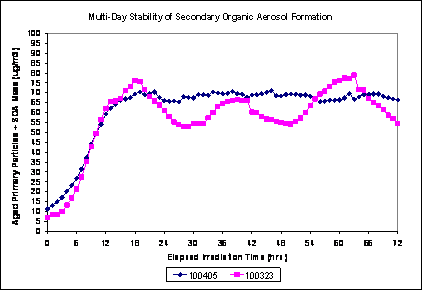
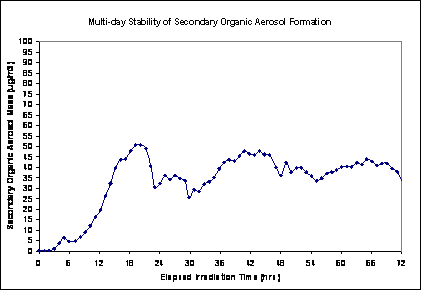
Because of the long time required to generate stable secondary mass concentrations in the chamber, it was decided to leave the lights on and run the chamber dynamically for the 4 days per week of exposure. We found that once a stable mass output of the chamber was achieved, the output remained fairly stable for the duration, as shown in Figure 3.
Figure 3. Multi-day stability of SOA formation, Aged Primary Plus Secondary scenario. PM concentration as a function of time using plenum air diluted with clean air, for 72 hours after turning lights on. Curves from two separate experiments, same conditions.
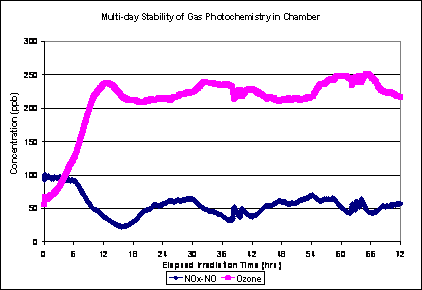
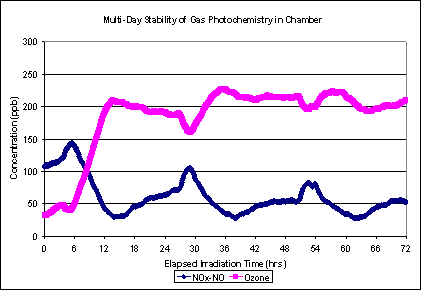
Similarly, the secondary gas concentrations show the same general pattern (Figure 4). The variability in Figures 3 and 4 can be explained by fluctuations in tunnel concentrations of gas phase organic molecules with particle forming potential, due to traffic patterns and changes in tunnel ventilation rates.
Figure 4. Multi-day Ozone and NOx-NOx curves, Aged Plume (Aged Primary Plus Secondary) scenario. Curves from one experiment starting March 23, 2010.
Figures 5 and 6, respectively, show the multi-day SOA and photochemical gas stability for the SOA Only exposure atmosphere (fluctuations reflect changes in precursor concentrations in the tunnel). This experiment uses filtered plenum air diluted with clean air at the same dilution factor as the experiments in Figures 3 and 4. The same trends are apparent.
Figure 5. Multi-day stability of SOA formation, SOA Only scenario (using filtered plenum air diluted with clean air, same dilution as Figures 3 and 4); showing PM concentration for 72 hours after the lights are turned ON.
Figure 6. Multi-day Ozone and NOx-NO curves, SOA Only scenario (same experiment as Figure 5).
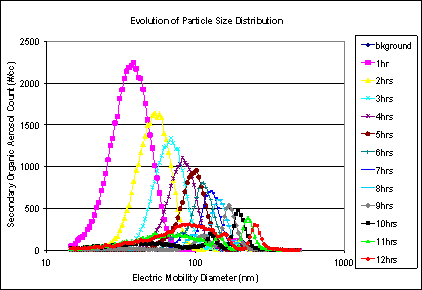
Figure 7 shows the evolution of the particle size distribution in the SOA Only scenario over time from Lights ON to 12 hours, showing first nucleation, then particle growth.
Figure 7. Evolution of particle size distribution, SOA Only scenario.
Atmospheres Generated for Animal Exposure Study. The exposure generation system has been used to conduct a number of exposures to each of the three atmospheres described in the previous section. As indicated above, following the coal power plant TERESA study, the exposure plan was refined to keep the total mass concentration of exposures consistent between scenarios.
Analysis of the integrated samples collected during exposures (described previously) is still pending, but the continuous data show that our methods allow us to generate reproducible exposures to primary particle mass, aged primary plus secondary particle mass, and secondary particle mass only, all at consistent concentrations. The available continuous exposure data are summarized below in Table 1.
Table 1. SMPS Mass measurements (average and standard deviation) for exposures.
| Scenario | Number of Exposures | SMPS Mass (μg/m3) | |
| Mean | Std Deviation | ||
| Primary Particles Only | 13 | 47.4 | 11.8 |
| Aged Primary Particles plus SOA | 24 | 48.7 | 9.3 |
| Secondary Organic Aerosol Only | 24 | 50.3 | 22.2 |
Animal Studies - Exposures. As described previously, animals were exposed to three different scenarios: Primary Particles from the tunnel plenum (P), Aged Primary Particles plus Secondary Organic Aerosol (P + SOA), and Secondary Organic Aerosol only [removing primary particles] (SOA). Control exposures to clean filtered air were done simultaneously.
Chamber output was passed through the parallel plate membrane denuders, as described previously, to remove excess unreacted primary gases and photochemical gases (O3 and NO2). The output was split between three manifolds, one for sampling and the other two for exposures, one on each of two exposure carts.
Animals were exposed to either fresh or photochemically aged aerosol or clean filtered air by inhalation in individual whole body chambers that also served as plethymographs for collection of respiratory data. Flow rate through each individual whole body chamber was maintained at 1.5 liters per minute. For each scenario, two sets of 5 hour exposures were run per day, 4 consecutive days per week, for 3 weeks. Each exposure session used equal numbers of animals exposed to the (fresh or aged) aerosol of the scenario and exposed to clean filtered air (sham) as controls. Up to 12 animals per group were exposed simultaneously. Clean filtered air for control exposures was prepared by passing room ambient air through Wilkerson cylinders containing Hydrosil (potassium permanganate-coated molecular sieve) to remove NO and NO2, and activated carbon to remove organic gases, followed by a HEPA filter to remove particles.
Animal Studies - Experimental Design. Animals were exposed 5 hours per day for 1 day, 2 days, 4 days or 12 days, depending on the end-point for which they were evaluated, as follows:
- Animals exposed 1 day for 5 hours, were used for Chemiluminescence within 2 hours of the end of the exposure (n=4/day).
- Animals exposed for 2 consecutive days for 5 hours/day, were used for Broncho-Alveolar Lavage (BAL) either 24 (n=4/exposure) or 48 hours (n=4/exposure) after the end of the second exposure. In a second set of animals tissues were harvested for histological analyses. Blood was collected from all this animals and CBC was performed, plasma and buffy coat samples were archived for analyses. A third set of animals (n=4/exposure) was used to analyze the level of methylation of three different gene promoters involved in vascular processes (eNOS, NOTCH4 and endothelin-1).
- Animals exposed for 4 consecutive days for 5 hours/day (n=8/week), were sacrificed 24 hours after the last exposure and used for BAL, histology and blood analyses.
- Finally, a set of animals implanted with blood pressure DSI PCA-40 transmitters (n=8/scenario) was used to monitor the changes in blood pressure with these exposures; these animals were exposed 4 days a week for 3 consecutive weeks and then sacrificed for histological analyses.
- Respiratory data were collected from a sample of animals in each group.
The distribution of animals in different groups and outcomes for this experiment are shown below in Table 2.
Table 2. Number of animals exposed for each outcome and scenario.
| Outcome/Scenario | P | P+SOA | SOA Only | ||||
| Exposed | Control | Exposed | Control | Exposed | Control | ||
| 1 Day Exposure | Chemiluminescence | 20 | 20 | 24 | 24 | 24 | 24 |
| 2 Days Exposure | Breathing Pattern | 20 | 20 | 18 | 18 | 18 | 18 |
| BAL 24 Hour | 12 | 12 | 24 | 24 | 24 | 24 | |
| BAL 48 Hour | N/A | N/A | 24 | 24 | 24 | 24 | |
| Histology | 8 | 8 | 24 | 24 | 24 | 24 | |
| CBC | 12 | 12 | 24 | 24 | 24 | 24 | |
| DNA Methylation | 4 | 4 | 2 | 2 | 12 | 12 | |
| 4 Days Exposure | Breathing Pattern | N/A | N/A | 12 | 12 | 12 | 12 |
| BAL 24 Hour | N/A | N/A | 12 | 12 | 12 | 12 | |
| Histology | N/A | N/A | 12 | 12 | 12 | 12 | |
| CBC | N/A | N/A | 12 | 12 | 12 | 12 | |
| 12 Days Exposure1 | Breathing Pattern | 2 | 2 | 2 | 2 | 2 | 2 |
| Blood Pressure2 | 4 | 4 | 4 | 4 | 4 | 4 | |
| Histology | 4 | 4 | 4 | 4 | 4 | 4 | |
1 Exposure to Primary Particles was for 10 days only.
2 Same animals used for P and P+SOA scenarios in a cross over design.
Results of Animal Exposure Study. Analyses of the data collected currently are under way, preliminary observations from the different end-points are summarized below.
Breathing Pattern: Minor changes are observed in animals exposed for 2 consecutive days, these changes include decreases in flows parameters (PIF, PEF and EF50) and volumes (Tv), with both the P and P + SOA scenario (no data reported for the SOA only scenario yet). These changes are consistent through the different exposure dates and groups and they seem to be potentiated with the repeated exposures.
For the P+SOA 4 days of exposure group, a rapid shallow breathing pattern is suggested with increases in 20% in the frequency and concomitant decreases in TI, Te Tv and MV, also, flow parameters (PIF, PEF and EF50) show a decrease in the exposed group.
Broncho- Alveolar Lavage (BAL): Total cell counts are similar between the groups, with a widening gap between the groups in the animals exposed for 4 consecutive days. Differential counts, protein and B-NAG concentrations are being analyzed, but no preliminary results can be added at this time for those outcomes.
Histology: Organs have been submitted for processing and H & E staining. No preliminary results can be added at this time for this outcome.
Blood Samples: Preliminary observations in the Complete Blood Counts (CBC) point to a decrease in circulating monocytes, eosinophiles and basophiles, with an increase in circulating lymphocytes and platelets.
Chemiluminescence: No apparent differences were observed between the groups.
DNA Methylation. The aim of this research is to define the role of epigenetic mechanisms specifically in vasoconstriction of blood vessels of the lung and heart after exposure to traffic-related ambient particles. Measuring the level of DNA methylation of three gene promoters involved in vascular processes (eNOS, NOTCH4 and endothelin-1) in rat heart and lung tissue will allow for comparison of particulate matter exposed versus unexposed tissues. Differences in methylation could implicate changes in expression levels of these genes providing a mechanism by which particulate matter induces vasoconstriction.
To date, sequencing has been conducted on a number of animal tissues in analyzing the eNOS promoter of rat heart and lung tissues and early indications point to differences in DNA methylation between control and exposed animals as well as differences between exposure scenarios. Analysis will continue in order to determine statistical significance and magnitude of differences. Additionally, we hope to be able to expand this area of research to the NOTCH4 and endothelin-1 gene promoters.
QA/QC
This project does not measure routine air quality data, such as that collected for ambient air, personal exposures, or micro-environmental measurements. Thus, no conventional data quality parameters are applicable. All measurements conducted in the testing and deployment of the apparatus used to simulate secondary aerosol formation from mobile source emissions have been made using instruments calibrated with standard methods. This includes gravimetric measurements of particle mass, semi-continuous measurements of particle size distribution, continuous measurements of particle number concentration, continuous measurements of pollutant gases, ion chromatographic measurements of sulfate, nitrate and ammonium ions, and thermal optical reflectance measurements of particulate elemental and organic carbon.
Future Activities:
Analysis of samples and data collected during these exposures is ongoing. Some of the specific activities include: chemical analysis of integrated samples collected during exposures (EC/OC, WSOC, trace elements); protein, BNAG from the bronchoalveolar lavage; DNA sequencing; and statistical analysis of results.
This project is the continuation of the TERESA study that was designed to compare the health effects of simulated secondary aerosol for coal-fired power plants with the effects from simulated secondary aerosol from vehicular sources. These efforts were supported by the previous Harvard/EPA PM Center, as well as by the current Center, and additional support was provided by both the Electric Power Research Institute (EPRI) and the U.S. Department of Energy (DOE). Analysis of data collected in the coal-fired power plant studies has been completed this year, and manuscripts from these studies (see publications) are in preparation, review at EPRI (to be included in a special supplement), or have been accepted for publication by Inhalation Toxicology. After the mobile source emission study health effects have been determined, we will publish a comparison paper that will include concentrated ambient particles, aged coal power plant aerosols, and mobile source emission aerosols.
References:
Kang, C. M., Gupta, T., Ruiz, P. A., Wolfson, J. M., Ferguson, S. T., Lawrence, J. E., Rohr, A. C., Godleski, J. and Koutrakis, P. (2010). Aged particles derived from emissions of coal-fired power plants: The TERESA field results. Inhalation Toxicology. accepted
Ruiz, PA, Lawrence, JE, Ferguson, ST, Wolfson, JM, Koutrakis, P. (2006). A counter-current parallel-plate membrane denuder for the non-specific removal of trace gases. Environmental Science & Technology 40(16):5058-5063.
Ruiz, P.A., et al., (2007a). Development and Evaluation of a Photochemical Chamber to Examine the Toxicity of Coal-Fired Power Plant Emissions. Inhalation Toxicology 19(8):597-606.
Ruiz, PA, Gupta, T, Kang, CM, Lawrence, JE, Ferguson, ST, Wolfson, JM, Rohr, AC, Koutrakis, P. (2007b). Development of an exposure system for the toxicological evaluation of particles derived from coal‑fired power plants. Inhalation Toxicology 19(8):607-619.
Savage, ST, Lawrence, J, Katz, T, Stearns, RC, Coull, BA, Godleski, JJ. (2003). Does the Harvard/US Environmental Protection Agency ambient particle concentrator change the toxic potential of particles? Journal of the Air & Waste Management Association 53(9):1088-1097.
Stone, EA, Snyder, DC, Sheesley, RJ, Sullivan, AP, Weber, RJ, Schauer, JJ. (2008). Source apportionment of fine organic aerosol in Mexico City during the MILAGRO experiment 2006. Atmospheric Chemistry and Physics 8(5):1249-1259.
Journal Articles on this Report : 7 Displayed | Download in RIS Format
| Other subproject views: | All 9 publications | 9 publications in selected types | All 9 journal articles |
|---|---|---|---|
| Other center views: | All 206 publications | 199 publications in selected types | All 199 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Coull BA, Wellenius GA, Gonzalez-Flecha B, Diaz E, Koutrakis P, Godleski JJ. The Toxicological Evaluation of Realistic Emissions of Source Aerosols study: statistical methods. Inhalation Toxicology 2011;23(Suppl 2):31-41. |
R832416 (2010) R832416C005 (2010) |
Exit |
|
|
Diaz EA, Lemos M, Coull B, Long MS, Rohr AC, Ruiz P, Gupta T, Kang C-M, Godleski JJ. Toxicological Evaluation of Realistic Emission Source Aerosols (TERESA)--power plant studies: assessment of breathing pattern. Inhalation Toxicology 2011;23(Suppl 2):42-59. |
R832416C005 (2010) R834798 (2013) R834798 (2014) R834798 (Final) R834798C005 (Final) |
Exit Exit |
|
|
Godleski JJ, Diaz EA, Lemos M, Long M, Ruiz P, Gupta T, Kang C-M, Coull B. Toxicological Evaluation of Realistic Emission Source Aerosols (TERESA)-power plant studies: assessment of cellular responses. Inhalation Toxicology 2011;23(Suppl 2):60-74. |
R832416C005 (2010) R834798 (2013) R834798 (2014) R834798 (Final) R834798C001 (Final) R834798C005 (Final) |
Exit Exit |
|
|
Godleski JJ, Rohr AC, Kang CM, Diaz EA, Ruiz PA, Koutrakis P. Toxicological Evaluation of Realistic Emission Source Aerosols (TERESA): introduction and overview. Inhalation Toxicology 2011;23(Suppl 2):1-10. |
R832416C005 (2010) R834798 (2013) R834798 (2014) R834798 (Final) R834798C005 (Final) |
Exit Exit |
|
|
Kang C-M, Gupta T, Ruiz PA, Wolfson JM, Ferguson ST, Lawrence JE, Rohr AC, Godleski J, Koutrakis P. Aged particles derived from emissions of coal-fired power plants: the TERESA field results. Inhalation Toxicology 2011;23(Suppl 2):11-30. |
R832416 (Final) R832416C005 (2010) R832416C005 (Final) R834798 (2013) R834798 (2014) R834798 (Final) |
Exit Exit |
|
|
Lemos M, Diaz EA, Gupta T, Kang C-M, Ruiz P, Coull BA, Godleski JJ, Gonzalez-Flecha B. Cardiac and pulmonary oxidative stress in rats exposed to realistic emissions of source aerosols. Inhalation Toxicology 2011;23(Suppl 2):75-83. |
R832416 (Final) R832416C005 (2010) R834798 (2013) R834798 (2014) R834798 (Final) R834798C001 (Final) R834798C005 (Final) |
Exit Exit |
|
|
Wellenius GA, Diaz EA, Gupta T, Ruiz PA, Long M, Kang CM, Coull BA, Godleski JJ. Electrocardiographic and respiratory responses to coal-fired power plant emissions in a rat model of acute myocardial infarction: results from the Toxicological Evaluation of Realistic Emissions of Source Aerosols Study. Inhalation Toxicology 2011;23(Suppl 2):84-94. |
R832416 (Final) R832416C005 (2010) R832416C005 (Final) R827353 (Final) |
Exit |
Supplemental Keywords:
traffic related particles, acute cardiovascular effects, primary and secondary particle effects, RFA, Health, Air, Scientific Discipline, Health Risk Assessment, Risk Assessments, particulate matter, Environmental Chemistry, Toxicology, biological mechanisms, chemical characteristics, autonomic dysfunction, biological mechanism , airborne particulate matter, cardiovascular vulnerability, chemical composition, animal model, oxidative stress, ambient particle health effects, PM, atmospheric particulate matter, automobile exhaust, ambient air quality, concentrated ambient particulates (CAPs), human health effects, traffic related particulate matterProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R832416 Health Effects Institute (2015 - 2020) Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R832416C001 Cardiovascular Responses in the Normative Aging Study: Exploring the Pathways of Particle Toxicity
R832416C002 Cardiovascular Toxicity of Concentrated Ambient Fine, Ultrafine and Coarse Particles in Controlled Human Exposures
R832416C003 Assessing Toxicity of Local and Transported Particles Using Animal Models Exposed to CAPs
R832416C004 Cardiovascular Effects of Mobile Source Exposures: Effects of Particles and Gaseous Co-pollutants
R832416C005 Toxicological Evaluation of Realistic Emission Source Aerosol (TERESA): Investigation of Vehicular Emissions
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
9 journal articles for this subproject
Main Center: R832416
206 publications for this center
199 journal articles for this center