Grantee Research Project Results
2011 Progress Report: Center for Child Environmental Health Risks Research
EPA Grant Number: R834514Center: Predictive Toxicology Center for Organotypic Cultures and Assessment of AOPs for Engineered Nanomaterials
Center Director: Faustman, Elaine
Title: Center for Child Environmental Health Risks Research
Investigators: Faustman, Elaine , Fenske, Richard , Griffith, William C. , Yost, Michael , Yu, Xiaozhong , Costa, Lucio G , Furlong, Clement , Thompson, Engelberta , Vigoren, Eric M. , Karr, Catharine J.
Current Investigators: Faustman, Elaine , Fenske, Richard , Griffith, William C. , Yost, Michael , Costa, Lucio G , Furlong, Clement , Thompson, Engelberta , Vigoren, Eric M. , Karr, Catharine J.
Institution: University of Washington
EPA Project Officer: Callan, Richard
Project Period: September 25, 2009 through September 24, 2016
Project Period Covered by this Report: August 1, 2010 through July 31,2011
Project Amount: $5,417,075
RFA: Children's Environmental Health and Disease Prevention Research Centers (with NIEHS) (2009) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
For more than a decade, researchers of the University of Washington Center for Child Environmental Health Risks Research (CHC) have been using a multidisciplinary research approach working in the lab, in the field, and in the community to understand the mechanisms that define children’s susceptibility to pesticides, identify the implications of this susceptibility for development and learning, and partner with our communities to translate our findings into risk communication, risk management, and prevention strategies. The Center has received a third cycle of funding from the National Institute of Environmental Health Sciences (NIEHS) and U.S. Environmental Protection Agency (EPA), which provided approximately 30% and 70% of the total Center funds, respectively. This report provides an overall description of the work done by each research project and facility core from August 1, 2010, through July 31, 2011. Separate reports for each project also are posted on the NCER Website.
The Center, directed by Dr. Elaine M. Faustman, is administratively housed within the Institute for Risk Analysis and Risk Communication, which is in the University of Washington’s School of Public Health. The Center includes partnerships with the Fred Hutchinson Cancer Research Center and the Yakima Valley community, located in the agricultural center of Washington State, to jointly sponsor a Community-Based Participatory Research (CBPR) Project aimed at reducing childhood pesticide exposure.
All Center efforts are highly integrated and comprise two field-based research projects, two laboratory-based research projects, three facility cores, an Administrative Core, and Faculty Development Investigator.
The specific objectives of the two field-based projects — the CBPR Project and the pesticide exposure pathways research project — are to:
- Improve our understanding of critical pathways of potential pesticide exposure for children; and
- Apply culturally appropriate interventions to reduce children’s exposure to pesticides.
The specific objectives of the laboratory-based research projects — a molecular mechanisms research project and a genetic susceptibility research project — are to:
- Identify cellular, biochemical and molecular mechanisms that cause adverse developmental neurotoxicity of pesticides; and
- Identify susceptibility factors for developmental neurotoxicity of pesticides.
The three facility cores are: Biomarkers and Exposure Assessment (BEA); Biostatistics, Modeling and Risk Characterization (BMRC); and Community Outreach and Translation (COTC). The cores are designed to support the research objectives and to put the research into a child-specific risk assessment context. Thus, the specific objectives of the facility cores are to:
- Provide core support for the development and application of risk assessment methods, enabling basic research on pesticide toxicity and exposure to inform risk decisions to protect children’s health from pesticides; and
- Foster partnerships between academic researchers and the community in which information requested by the community and basic research deficiencies/gaps are translated into studies that address the needs of both.
The Administrative Core provides fiscal oversight, resource management, coordination, and integration of Center activities. The COTC shares membership with the Administrative Core and includes the Center’s Faculty Development Investigator/Pediatric Health Specialist, Dr. Catherine Karr. Since 2009, Dr. Karr has served as the Center’s Faculty Development Investigator, as well as Pediatric Health Specialist. Center support and mentoring enable Dr. Karr to capitalize on the complementary and collaborative resources at the Center to navigate the transition to independent investigator in this subject area
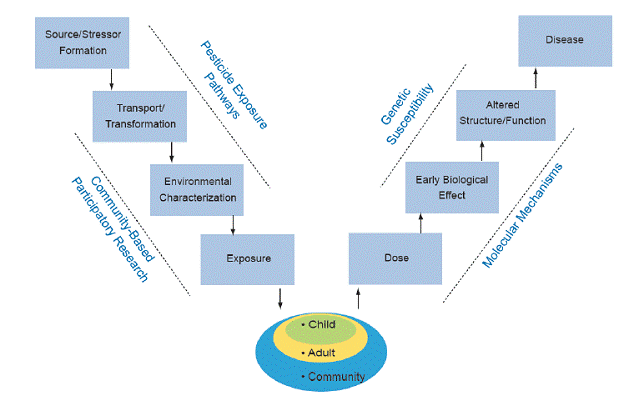
Work in the Center is organized around the Public Health paradigm “V-diagram,” which connects occurrence of disease in humans to the original source of the problem. Along the pathway from source/stressor to disease, the diagram identifies intermediate processes (which may be subject to public health intervention) and conditions (which may be observable for public health monitoring and hypothesis testing). This paradigm is central to the overall organization and integration of the various components of the Center. Using Global Positioning System (GPS) tools to relate human activities and land use helps identify how organophosphate (OP) pesticide (stressor) application is transferred/transported to create OP environmental conditions. The CBPR Project focuses on the take-home pathway of OP transfer potentially leading to exposure and resulting dose in children. The biological sampling efforts help identify susceptibility factors and genetic mechanisms relating dose to early biological effect. Biostatistical analyses and outreach link all these components together.
Figure 1.The Public Health paradigm “V-diagram” frames and integrates the Center’s research efforts:
Progress Summary:
Below, we provide highlights of Center progress between August 1, 2010, and July 31, 2011.
COMMUNITY-BASED PARTICIPATORY RESEARCH PROJECT (R834514C001)
A key feature of the Center is the CBPR Project, which identifies the multiple potential pathways that may contribute to pesticide exposure in adults and children living in agricultural communities. Unlike many other studies of potential pesticide exposure, the CBPR investigators have used a group randomized trial design. Advantages of such an approach are the potential to reach large numbers of people, disseminate messages about behavior across a diverse population, change community norms about behavior, and integrate new behavioral practices into existing community structures and organizations. Part of the study design requires involvement from the communities to guide us in planning intervention activities. The results of this research support the hypothesis that the take-home exposure pathway contributes to residential pesticide contamination in agricultural homes where young children are present.
The highlights of this past year include successful recruitment of families, completion of the data collection components of the thinning season (such as a baseline and a follow-up questionnaire, activity and diet diary, urine, blood, buccal cell, house and vehicle dust samples and using indoor and outdoor air monitors and deposition plates) and collection of hair/saliva for the Cortisol and Stress Study. Specifically, during this past year we were able to re-establish contact with 100 households (60 farmworker and 40 non-farmworker families) from previous CHC cohorts (51 farmworker and 40 non-farmworker families were recruited from CHC-2 cohort (2003-2010) and nine from the CHC-1 cohort (1998-2003). A total of 132 farmworkers and 90 non-farmworkers were re-contacted before reaching the goal of 60 farmworker and 40 non-farmworker households. Given that this is a highly mobile population, it is significant that we were able to recruit 100 cohort members to participate in our current study. Further, they all have consented to participate in the three data collection periods of the pre-thinning, thinning, and non-spray season. Importantly, we were able to collect urine, buccal cells, 3-Day Diet Survey and 7-Day Activity Diary on all 60 referent farmworkers' children (who now are between 7 and 12 years of age). Furthermore, we were able to collect blood samples from all but 5 of the 60 referent children. Similarly, for referent non-farmworkers' children, we were able to collect all needed samples on all 40 children except for two blood samples. For adults, we were able to collect all needed samples on all 60 farmworker adults except for one Day-5 urine and two vehicle dust samples. We were able to collect all samples on all 40 non-farmworker adults. We also collected indoor and outdoor air samples so we can better understand the influence of drift by proximity to spray events. Finally, we recruited 27 Hispanic farmworker and non-farmworker women who also are current participants in the CHC-3 study to further participate in the Cortisol and Stress study. These women are ages 18-49 and currently are neither pregnant nor taking any corticosteroid treatment. We have completed sample collection of hair and saliva for 15 participants. We will measure cortisol level in these samples reported to increase due to exposure to organophosphate pesticide.
PESTICIDE EXPOSURE PATHWAYS RESEARCH PROJECT (R834514C002)
The Pesticide Exposure Pathways Research Project’s overall aim is to develop a systematic understanding of residential pesticide exposures arising from proximity to agricultural spraying in rural agricultural communities where children live. The proximity exposure pathway is due to transport of pesticides from applied fields to homes and other settings. The mechanisms of transport include spray drift, volatilization, resuspension and deposition of particles containing pesticides. The pathways project studied both local pesticide application factors and human activity factors that contribute to childhood exposure to pesticides.
The highlights of this past year include completing air and particle sampling fieldwork phase I (March – May 2011) and proceeding to phase II (summer of 2011).
In collaboration with the CBPR Project, data were gathered on airborne exposures to organophosphate pesticide dusts and vapors, with the deployment of 46 polyurethane foam passive air samplers and 46 microslide/polypropylene deposition plates in a subset of 21 research participant’s homes, specifically positioned in a living room or kitchen at an average height of 1.5 meters) over a time period of 30 days to collect simultaneous measurements of particle deposition and airborne concentrations of the organophosphorus pesticide chlorpyrifos and its oxon analog. Overall, 95% of these homes agreed to participate in the air monitoring portion of the study. Responding to feedback from the field group, the plan of using 3M NonDamage Utility Hooks to hang the passive air sampler inside the home was changed to using free-standing ornament hangers for easier location near the deposition plates. Outdoors, a large passive air sampler disk, housed in a stainless steel chamber, was positioned more than 6 meters from the outside of the home at a height of 1.6 to 2.1 meters with these outdoor samplers remaining stable during heavy winds and rain and with no tampering with the instruments noted. In addition to the use of passive air sampler, the fieldwork team also deployed side-by-side active and passive samplers for 7-14 days at two calibration sites in the local communities, a public high school in town (non-proximal), and a weather station near agricultural fields (proximal). Both calibration sites were co-located with Washington State University (WSU) AgWeatherNet stations and Department of Ecology air monitoring sites that record real-time weather data and PM2.5, respectively.
Moreover, a total of 40 MicroDAQ data loggers also were deployed to collect real-time temperature inside and outside the homes. All participant homes were within a 10-mile radius of the nearest WSU AgWeatherNet station, which provided additional real-time data on temperature, precipitation, wind direction, wind speed, and relative humidity.
Finally, to assess the importance of proximity vs. potential take-home pathway, researchers used Global Positioning System (GPS) and identified eight homes as “proximal/farmworker,” two homes as “proximal/non-farmworker,” six homes as “non-proximal/non-farmworker,” and four homes as “non-proximal/farmworker,” with “proximal” defined as 5 – 200 meters and “non-proximal” as 550 – 1,200 meters from an agricultural field.
Currently, these sampling matrices are being analyzed for chlorpyrifos and oxon using liquid chromatography-mass spectrometry (LC-MS-MS) at the University of Washington Environmental Health Laboratory. In Phase II of the study, these samples will undergo extractions for azinphos methyl (AZM) and oxon. Furthermore, the deposition microslides were analyzed using ImageJ software to count and size dust particles by optical microscopy. This information will help researchers describe phase-partitioning of the semi-volatile pesticides and their transformation to the more toxic oxon. In addition, the measured ratios of indoor/outdoor air concentrations will be used to assess infiltration to indoor environments.
MOLECULAR MECHANISMS RESEARCH PROJECT (R834514C003)
Pesticide‐induced alterations in the regulatory dynamics of normal cell proliferation, differentiation and cell death result in altered central nervous system (CNS) morphogenesis. These alterations are correlated with subsequent deficits in learning and development. Thorough understanding of mechanisms of toxicity at the molecular, cellular and organism level is therefore important as these mechanisms may define “windows of susceptibility” for many neurodevelopmental toxicants as well as for pesticides. The Molecular Mechanism Research Project evaluates these mechanistic processes across several pesticide classes to understand the commonality and uniqueness of pesticide toxicity across dose, lifestage, cell type and behavioral domain. These efforts bring together the expertise from different laboratories and focus this expertise on the relationship between pesticide‐induced alterations in cell proliferation and death during embryonic/postnatal development and postnatal alterations in behavior. The laboratory of Dr. Elaine Faustman addresses the impacts of pesticides and model neurotoxicants on prenatal neurogenesis (gestational day 12‐17), and the laboratory of Dr. Lucio Costa evaluates the postnatal effects on gliogenesis.
In the last 2 years, the Faustman lab initiated studies to explore new in vitro models for the evaluation of neurogenesis, including proliferation and differentiation. The highlights of this year include the following:
- Establishing human neuronal stem cell cultures. Direct application of this culture system has provided critical information on chemical impacts on human cell proliferation and differentiation, which is relevant for neurogenesis in human tissues. We investigated the effects of chlorpyrifos (CP) and arsenic on proliferation and differentiation. Human neural stem cells (hNPCs) were expanded in serum free proliferation expansion medium (MEM) or differentiated in neural differentiation medium (HDM). Cells were incubated for 72 hours in either MEM or HDM with CP. Dose-dependent decreases in cell viability were observed in both cells cultured in proliferation MEM and differentiation HDM medium, with significantly greater effects of CP on cells in HDM.
- The Faustman lab further investigated whether CP differentially altered epigenetic markers during proliferation and differentiation. We examined protein expression of epigenetic markers. These initial results suggest that CP exposure in differentiating hNPCs resulted in significant alteration in expression of several epigenetic markers associated with transcriptional activation while exposure of cells to CP during proliferation showed fewer changes in these epigenetic factors. Differential alterations of epigenetic markers during proliferation and differentiation may provide cell stage specific mechanistic clues for examining effects on neural development. These examinations of the effect of pesticides on human cells should provide valuable information and help to interpret in vivo animal studies and more directly inform prediction for impacts in human populations.
- In addition, the Faustman lab is invested in optimizing biomarker methods to allow for analysis from human samples to profile the potential impacts of pesticides on exposed populations. Due to their non-invasive nature and low cost to collect, buccal epithelial cells have been used for the isolation of DNA and, in limited cases, for obtaining RNA for gene expression analyses. We have tested several types of DNA and RNA isolation methods. For DNA, an ethanol precipitation method following a proteinase K digestion (Gentra Puregene kit) was found to provide superior yields compared to a phenol extraction method and to a protocol using magnetic beads (Invitrogen Chargeswitch). DNA isolated with the optimal method is of high quality and able to be amplified using whole genome amplification methods to provide ample material for multiplexed genotyping arrays. With regards to RNA, a column-based isolation (Qiagen RNeasy) paired with a short proteinase K digestion step outperformed a phenol-chloroform (Invitrogen Trizol) extraction method. A comparison of whole transcriptome amplification methods showed that amplification products generated with the Nugen WT Ovation kit provided greater signals in RT-PCR assays than did those produced by the Ambion WT Expression kit.
- We characterized field samples collected by the CBPR Project to identify potential differences in farmworker and non-farmworker families in metabolite profiles with the Affymetrix Drug Metabolizing Enzyme and Transporter (DMET) Plus platform. We investigated if genotypes for key CYP450 genes might modify the urinary identifications of OP metabolites and this relationship and our analysis has identified genetic variation in the CYP3A5 gene (6986A>G, rs776746) (ANOVA, p < 0.05). Post-hoc testing showed that individuals with the G/G genotype had higher acetylcholinesterase (AChE) inhibition per unit AZ or DMTP compared to A/A individuals. Because the G allele produces a splice defect that is associated with lower enzyme activity, future PBPK modeling will determine if genotypic variation at this locus could be used to identify individuals who may be more sensitive to OP-association effects.
During the past year, work in the Costa lab has progressed according to plans, addressing the proposed specific aims. The Costa lab had shown previously that the OP insecticide diazinon and its active oxygen analog diazoxon induced oxidative stress, by measuring levels of reactive oxygen species (ROS), in rat cortical astrocytes. Both compounds were effective at concentrations ranging from 0.1 to 10 µM. The highlight of this year is that we now have found that exposure of astrocytes for 24 h to diazinon or diazoxon (both at 10 µM), followed by complete wash-out, and by co-culture of astrocytes with rat hippocampal neurons for an additional 48 h, significantly inhibits the ability of astrocytes to foster neuronal differentiation. Control astrocytes, when incubated with hippocampal neurons, promote neurite outgrowth. When astrocytes were pre-exposed to either diazinon or diazoxon, length of the longest neurite (previously determined by tau staining to be the axon [Guizzetti, et al., 2008]) decreased by 50%, while length of minor neurites decreased by 40%. There were no changes in the number of neurites/cell. Incubation of astrocytes with a known oxidant, H2O2, which was used as a positive control, induced oxidative stress in astrocytes, and reduced neuritogenesis in hippocampal neurons to an extent similar to the OPs. Astrocyte-mediated neuritogenesis has been shown to be mediated by permissive neuritogenic factors, which are expressed and secreted by astrocytes. We previously had shown that two extracellular matrix proteins, fibronectin and laminin, play a major role in astrocyte-mediated neuritogenesis. In a preliminary experiment, we found that diazinon and diazoxon decrease fibronectin protein level in the astrocyte lysate, as does H2O2. In contrast, neither OPs nor H2O2 had any effect on laminin levels.
GENETIC SUSCEPTIBILITY RESEARCH PROJECT (R834514C004)
Activities of the Genetic Susceptibility Research Project have contributed to a greater understanding of the role of gene-environment interactions for children’s susceptibility to OP insecticides. The project involves interactions with many other entities, including the Centers for Disease Control and Prevention (CDC), Environmental Protection Agency (EPA) Region 10, Agency for Toxic Substances and Disease Registry (ATSDR), the University of California Berkeley Center for the Health Assessment of Mothers and Children of Salinas (CHAMACOS), Washington State Department of Health, Washington State Pesticide Incident and Reporting Panel (PIRT), other U.S. government agencies, the United Kingdom (UK) Committee on Toxicity and several members of the UK Parliament. The overall goal of the Genetic Susceptibility Project is to develop specific biomarkers of exposure to OP compounds, and to use these biomarkers to explore gene-environment interactions related to genetic variability in the paraoxonase (PON1) gene, particularly with respect to OP exposures that occur during early development.
The highlights of this year include signficant progress on the first specific aim, which is to develop and validate immunomagnetic bead (IMB) isolation protocols for the biomarker proteins of interest: butyrylcholinesterase (BChE), acyl peptide hydrolase (APH), and AChE, and to use a proteomic approach to validate the use of these proteins as biomarkers of OP exposure:
- Recently, we added two additional potential biomarkers of toxicity and/or exposure, monocyte carboxylesterase (CES) and neuropathy target esterase (NTE), that are present in blood. The OP modification of BChE was initially determined by incubating plasma with 25 μM chlorpyrifos oxon (CPO) and 25 μM azinphos methyl oxon (AZO) to generate the ethyl and methyl modified active site serines. In both cases, the OP adducts aged to the mono acyl derivatives, mono-ethyl phosphoserine in the case of CPO and mono-methyl phosphoserine in the case of APMO. The mono-methyl phosphate added 94 Da to the active site peptide (chymotryptic digest) GES*AGAASVSLH, while the mono-ethyl phosphate added 107 Da to the active site peptide. We currently are determing the limit of detection of modified peptide by injecting different ratios of modified vs. unmodified peptide digests into the mass spectrometer (MS). A ThermoFisher LTQ-FT with a focusing lens was installed in the MS Facility, increasing the sensitivity of the instrument by at least five-fold.
- We currently are characterizing the CPO and APMO adducts of recombinant and red cell APH. The red cell APH has a much longer half-life than the plasma BChE. We have identified the peptide modified by OP exposure by mass spectrometry (collaboration with Dr. Mike MacCoss). We also have generated 15N-labeled standard APH by expression of APH in an E. coli system. The 15N-labeled APH will be used to quantify the RBC APH modified by exposure. We have added two additional target biomarker proteins, CES1 and NTE, to the panel of OP biomarker target proteins. These proteins should be accessible from the mononuclear cell fraction of blood, and also, in the case of NTE, from platelets. We characterized CES activity in an activity stained gel in analyzing an extract from a cultured human monocyte cell line. We have generated recombinant CES1, using a baculovirus expression system, and are in the process of purifying this protein. For assessment of NTE as a biomarker, we are cloning the active-site peptide, NTE esterase domain (NEST), into an expression system. Commercial antibodies that recognize human and mouse NTE also are available.
- The generation of anti active-site peptide antibodies is under way both for APH and BChE. We think that having very pure target active site peptide will allow for a more rapid identification of unusual modifications and thereby a better characterization of unusual exposures to ChE inhibitors. We currently are optimizing the isolation protocols for APH from the brains and red cells of OP-exposed mice. The optimization protocols that we developed during this past year for BChE, which included the use of special tubes and high temperature wash of the immunomagnetic beads to remove loosely bound antibodies, will facilitate the generation of highly pure APH. The two-step IMB protocol, where we first isolate highly purified target protein (BChE and APH), then follow with the digestion of the purified target protein and isolate the active-site peptides in a second step IMB purification, should produce a nearly pure modified and unmodified target active site peptide. This will facilitate the characterization of unusual adducts to the active site serine. We are transferring these protocols to Rudy Johnson at CDC. These same biomarkers are being used to assess exposure to OP-related compounds in aircraft-cabin air. Modification of BChE by exposure to OP-related compounds in engine oil leaks in aircraft cabin air was determined by incubation of BChE with the chemically synthesized active metabolite of tricresyl phosphate (TCP) CBCP or cyclic saligenin cyclic phosphate. Although the TCP does not inhibit BChE, its toxic metabolite CBDP is a very potent inhibitor. A manuscript describing the CBCP was published in 2010 (Schopfer, et al., 2010). Several adducts were observed; the 170 Da cresyl phosphoserine, a 186 Da adduct where the cresyl group was lost and the open ring saligenin was bound and an unusual aged adduct phosphate (+ 80 Da) where all acyl groups were lost. This is, to our knowledge, the first example of an aged organophosphate adduct where all of the acyl groups are lost on aging. In addition, a 156 Da adduct was observed that would be consistent with a phenyl phosphoserine adduct as observed when we inhibited BChE in vitro with phenyl saligenin cyclic phosphate. To set the magnetic bead protocol up for high-throughput, a magnet was fabricated that allows processing of 96 samples in a standard microtiter plate. We have been able to use rat liver microsomes to bioconvert TCP to active esterase inhibitor(s) in vitro, and are using this technique to inhibit BChE for detection of adducts by MS. Surprisingly, tri-paracresyl phosphate also converts to an inhibitor of some esterases in vivo. The anti-human APH antibodies that we generated also recognize mouse APH.
Progress made regarding specific aims 2 and 3, which use knockout and humanized mouse models to evaluate interactions among biomarkers of OP sensitivity, exposure and response during critical stages of development, includes:
- Working on methods for isolation of BChE from mouse plasma, which will require the use of a different antibody than the one currently used for isolation of human BChE. We are raising antibodies against CES, and purchasing anti-NTE antibodies, which will be useful for isolating these enzymes from mouse tissues.
- For the experiments characterizing PON1 status as a biomarker of sensitivity to chlorpyrifos (CP) and CPO during gestation, the initial chronic dose-response studies in pregnant and non-pregnant mice demonstrated steep dose-response curves for inhibition of BChE and AChE, with higher doses leading to weight loss and fetal abnormalities. PON1-/- mice were more sensitive than PON1+/+ mice to these effects of chronic CPO exposure. Fetuses were more resistant than dams to CPO-inhibition of BChE, and tgHuPON1R192 females were more resistant than females expressing tgHuPON1Q192. Continuing studies are using chronic gestational exposure in these dose ranges to determine the importance of PON1 status on maternal and fetal health in mice of all four PON1 genotypes. Recent FISH analyses revealed that the transgenic mice carry multiple copies of the transgenes on different chromosomes, which probably explains at least some of the variability in PON1 levels observed in pups from the same breeding pairs of mice. Mice now are being backcrossed to the PON1-/- background to elucidate the effect of copy number on PON1 activity levels, which we anticipate will produce “low expressers” and “high expressers” with lower interindividual variability.
We also have completed project aims from the previous funding period, one of which was to evaluate the effects of exposure to CPO during early postnatal development and to determine the role of the human PON1-Q192R polymorphism in modulating these effects, using multiple endpoints of OP toxicity. This aim involved exposure of transgenic and knockout mice to CPO throughout early postnatal development, followed by measurement of multiple endpoints of neurotoxicity, including neurochemistry, histopathology, neurobehavioral assessment and global gene expression patterns in different regions of the brain. Assessment of all of the indicated endpoints for these studies has been completed. PON1+/+, PON1-/-, tgHuPON1Q192 and tgHuPON1R192 mice were exposed to CPO at five different dosages (0.15 mg/kg/d, 0.18 mg/kg/d, 0.25 mg/kg/d, 0.35 mg/kg/d, and 0.50 mg/kg/d), or to vehicle alone, from postnatal day (PND) 4 to PND 21. A manuscript now in press (Cole, et al., Toxicological Sciences) describes changes in gene expression patterns and brain AChE activity following repeated exposure of mice to CPO. After repeated CPO exposure from PND 4 to 21, gene expression was measured on PND 22 using Affymetrix Mouse Genome microarrays. All four genotypes exposed to CPO (0.35 or 0.50 mg/kg/d) showed significant differences in gene expression compared with controls. Pathway analysis and Gene Set Analysis revealed multiple pathways and gene sets affected by CPO exposure, including genes involved in mitochondrial dysfunction, oxidative stress, neurotransmission, and nervous system development. Changes in gene expression were modulated differentially by the two Q192R alloforms, with the most overlap in significantly enriched gene sets occurring between the tgHuPON1Q192 and PON1-/- mice. These findings indicate that neonatal CPO exposure is associated with wide-ranging effects on gene expression in the brain, and that PON1 status can modulate these effects, even when PON1 levels are low during early development. Finally, we are preparing a related manuscript describing the results of neurobehavioral assessment of mice exposed repeatedly to CPO during the postnatal study. This study did not reveal significant effects on learning and memory (assessed with a water radial arm maze, Morris water maze, or contextual fear conditioning), motor function (assessed with a rotarod test and locomotor activity in an open field), auditory startle amplitude, prepulse inhibition of startle, early reflex development (reflex righting, negative geotaxis, cliff avoidance), or the appearance of developmental landmarks. Bodyweight gain and auditory startle latency were significantly affected by exposure of PON1-/- mice to CPO at 0.25 mg/kg/d and higher. Additionally, from PND 16-19 the PON1-/- mice exposed chronically to 0.18 or 0.25 mg/kg/d exhibited transient hyperkinesis in the 20-min period following CPO administration. PON1+/+ mice did not exhibit this hyperkinesis.
BIOMARKERS AND EXPOSURE ASSESSMENT FACILITY CORE
The Biomarkers and Exposure Assessment (BEA) Facility Core has focused on samples collected in the field and coordinating their analysis for biomarkers, chemical analysis, and genotype and phenotype characterization. A strong emphasis has been placed on providing analytical support for field samples collected by the CBPR Project and the Pesticide Exposure Pathways Research Project. The Core stores the results of the analyses and assists investigators in their interpretation.
BEA also has continued to provide support to the Pesticides Exposure Pathways Research Project and the CBPR Project for collection of samples in early spring of 2011 from our existing cohort in Yakima Valley. The Core helped investigators develop new biomarkers to span the range from exposure to early markers of potential disease. The Core has added several biomarkers of exposure to the Center by measuring 3,5,6-trichloro-2-pyridinol (TCPy), a specific metabolite of chlorpyrifos, and oxons of OPs. The Core collaborates closely with the University of Washington Environmental Health Laboratory (EHL) to carry out these analyses. The Core also has provided support to the Genetic Susceptibility Research Project through the identification and development of relevant biomarkers. The Core continues to monitor and maintain the Center’s biorepository and associated IRB applications and requests. Additional funding has been obtained from the National Children's Study to analyze samples in the biorepository collected in past CHC studies. The results of these additional studies will be used to inform the choice of the types of analyses that are being carried out on samples collected in our present study.
The Core also is collaborating on a paper that will be submitted in the near future on the samples collected in the CHC2-CBPR study and the types of analyses that were performed on these samples.
BIOSTATISTICS, MODELING, AND RISK CHARACTERIZATION FACILITY CORE
The Biostatistics, Modeling, and Risk Characterization Facility Core provides innovative statistical, modeling, and risk assessment capabilities to interpret results of the Center research. This Core has used high dimension statistical models that are innovative for exposure analysis and allow for successful interpretation and analysis of complex cohort studies. Collaboration with investigators on statistical designs and analysis is the start of an integrated process whereby this Core has improved the initial design and construct of studies to increase their power to detect statistically significant results. Some of the challenges the Core has taken into account when performing analyses include field samples with frequent values below quantification limits, missing data, multiple data collection protocols, combinations of random and fixed effects, repeated measures, and multiple spatial and temporal scales.
This year, the Core has been deeply involved in the design of the CBPR project to collect information on the cohort identified in previous CHC studies and re-recruited to conduct further CBPR research. Analysis of exposure data from the Child Health Center second funding cycle (CHC2)-CBPR has focused on using Bayesian Markov chain Monte Carlo methods using multivariate distributions in hierarchical models to understand the relationship between child and adult exposures from the take home pathway for orchard workers, trends in potential exposures across agricultural seasons, differences between exposures of farmworker and non-farmworker households, and sources of variability.
In the CHC2-CBPR, we collected urine samples across three agricultural seasons—spray, harvest and off season—on 3 days within each season so that we could separate within person variability from between person variability. This has proved valuable because when the large within person variability is separated out, we can characterize the smaller within person variability to understand and show how population members differ from each other. The seasonal trends in exposure characteristics have shown that the potential for exposures to dimethyl organophosphate pesticides increase for farmworkers in both the spray and harvest seasons and decrease to lower levels similar to the non-farmworkers in the off-season, while the potential exposures to diethyl organophosphate pesticides are low across these three seasons. Children of farmworkers showed trends in exposure patterns similar to the adults in their households indicating that the take home pathway appears to be a major part of their exposure to organophosphate pesticides.
To demonstrate the importance of separating within person variability from between person variability, we have used these methods to reanalyze a data set of urinary metabolites of organophosphate pesticides collected from children living in the same area as our population—the Yakima Valley. This study has been accepted for publication and shows that it is important to separate large within person variability to properly characterize and understand the between person variability, and that in the Yakima Valley there is large year to year variability due to different levels of pesticide use.
The data from the CHC2-CBPR study also includes the analysis of six organophosphate pesticides in the dust of homes and vehicles. In the dust, azinphosmethyl has the highest concentration and the homes and vehicles of farmworkers have higher concentrations than those of the non-farmworkers. Dust concentrations of organophosphate pesticides are significantly correlated with metabolites in the urine of both adults and children demonstrating that pesticides associated with the take home pathway are a source of exposure for children and possible re-exposure for the adult farmworkers.
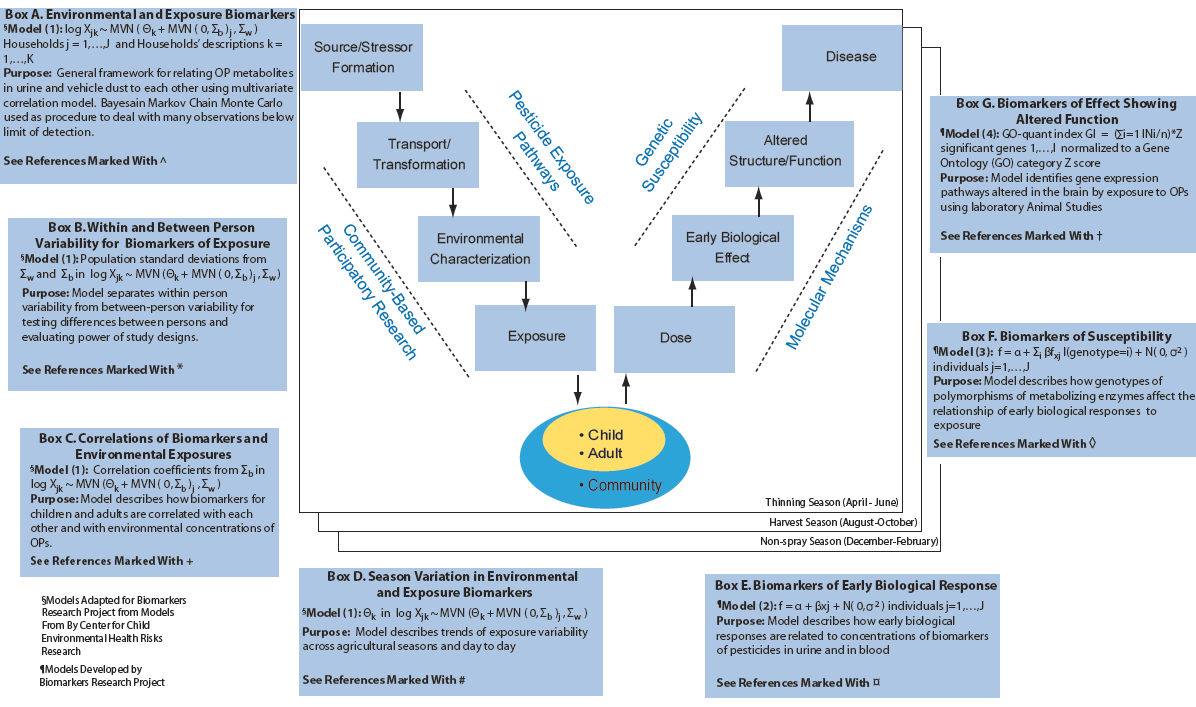
We also have been using the CHC2-CBPR study to relate metabolites in the urine to parent organophosphate pesticide concentrations in the blood. We have been able to show a significant relationship between the parent compounds in blood and metabolites in urine. Also, both the parent compounds and the metabolites are significantly correlated to cholinesterase inhibition showing that inhibition can be seen even at relatively low levels of exposure. During this year, the Core has developed an integrative tool for evaluating biomarkers across the Public Health paradigm. This tool, shown in Figure 2, was developed in conjunction with the targeted EPA biomarker grant entitled “Integrating Innovative Biomarkers of Environmentally Induced Disease for Children in Agricultural Communities.” (EPA grant number RD832733). We have organized this integrative tool around the Public Health paradigm “V-diagram,” which models the occurrence of disease in response to original sources of exposure (see Figure below). The diagram identifies intermediate processes (which may be subject to public health intervention) and conditions (which may be observable for public health monitoring and hypothesis testing) along the pathway from exposure source to health effect. While the Center Project focused on identifying the potential for a take home pathway of OP transfer, in this tool and its applications we focused on modeling biomarkers of exposure, susceptibility, and effect as shown in Figure 2, and used Center data to characterize these relationships.
Figure 2. Integrative tool containing models we developed in boxes A-G organized around the Public Health paradigm "V-diagram."
COMMUNITY OUTREACH AND TRANSLATION FACILITY CORE
The Community Outreach and Translation Core (COTC) is positioned within the Center to overcome the barriers involved in sharing, interpreting, translating, documenting, and archiving data and information in an interdisciplinary and true collaborative research setting. The goal of the COTC is to develop, implement and evaluate strategies to translate and apply the scientific findings of the Center into information for the public, policy makers, and clinical professionals to use to protect the health of children. The COTC shares membership with the Administrative Core and includes the Center’s Faculty Development Investigator/Pediatric Health Specialist, Dr. Catherine Karr.
During the reporting period, COTC members have been involved in a variety of outreach and translational activities, on both the local and national level:
- Dr. Karr is in charge of coordinating monthly Children’s Environmental Health Journal Club sessions and developing outreach materials (such as mailers sent to parents of all children born in Washington State (n~90,000) as part of the WA Department of Health (DOH) Child Profile health prevention program.The team currently is planning to host an External Advisory Committee meeting, a joint Center/PEHSU (Pediatric Environmental Health Specialty Unit; Dr. Karr is director) meeting, and also develop a continuing education course.
- Dr. Faustman has used her experiences with the Center to inform and guide the National Children’s Study (NCS), particularly the Pacific Northwest Center for the National Children’s Study (PNWNCS), for which Dr. Faustman also is the PI. Efforts to engage communities and educate stakeholders as part of the PNWNCS are actively under way, as the study currently is recruiting participants in Grant County, Washington. Grant County is one of 10 NCS locations that currently are piloting a door-to-door recruitment strategy to enroll women into the study. The study starts with enrolling women just before or during pregnancy and following their children as they grow up. This effort has benefited greatly from the wealth of knowledge that the Center has amassed since its inception under the leadership of Dr. Faustman and the CBPR Project team that has led community efforts to reduce children’s exposure to pesticides.
- Dr. Faustman is chairing and organizing a session at the American Public Health Association’s (APHA) Annual Meeting in Washington, DC, October 29th through November 2nd. The theme of this year’s annual meeting is “Healthy Communities Promote Healthy Minds and Bodies” and the title of Dr. Faustman’s session is “Formative Research in the National Children’s Study: Impacting the Research Design of the National Children’s Study.” This session will feature formative research projects in genomics, environmental exposures, and stress and describe how they are designed to meet this need. The scope and objective of these projects will be presented along with summary results to illustrate how these projects provide information to inform study visit measures for the NCS and to further the development of the NCS as a high quality platform for investigation of children’s health in many areas.
- Sharing lessons learned and working across Center research project and facility cores, whereby the Center’s CBPR Project was able to re-establish contact with 100 households from their cohort study in the Yakima Valley that took place in earlier years of the Center. Furthermore, the CBPR team worked with the Pesticide Exposure Pathways Research Project to collect indoor and outdoor air samples to better understand the influence of drift by proximity to spray events. These activities in the past year demonstrate the Center’s successful approaches in the field for engaging communities with the overall aim of protecting children’'s health. Other examples of cross-Center efforts include the analytical support and biorepository capacity provided by the Biomarkers of Exposure Faciltiy Core and the results interpretation capacity provided by the Biostatistics, Modeling, and Risk Characterization Facility Core. Finally, as described above, there is a great deal of collaboration among the COTC, Administrative Core, and Dr. Karr's efforts in her multiple roles in the Center.
- Engaging Dr. Karr in her role as a Pediatric Health Specialist in Clinical Translation and Needs Assessment included various activities, such as translation of Center-based activities and scientific discovery for health professionals who have responsibilities for child health in clinical or public health settings through educational material development and delivery. Examples are presentations at local area Grand Rounds, participation in public health advisory panels, serving as a member of the Washington State Environmental Biomonitoring Survey Advisory Committee, serving as an advisory on the Migrant Clinicians Network Adolescent Farmworker Occupational Health and Safety Advisory Committee, participating on the planning committee for the Joint Meeting NIEHS/EPA Children’s Research Centers-PEHSU Program (10/21/2010), presenting at EPA Region 2 Urban Pesticide Summit, NYC (9/14/2010): “Health Effects of Pesticides on Children: Focus on U.S. Urban Settings,” organizing the global health session and presenting “Beyond the Backyard: Building Children’s Environmental Health Capacity in SE Asia,” developing review articles, policy statements and technical reports for pediatric clinical providers and public health professionals on topics in child health impacts of environmental factors including pesticides. Moreover, Dr. Karr has delivered specialty lectures and grand rounds presentations on exposure to pesticides and other contaminants on child health outcomes.
- Evaluating the Center’s Impact on Pesticide Practices by Employing Tracking Translational Tools where a graduate student has begun conducting literature reviews relevant to this aim to identify methods that we can use to translate lessons learned from the burgeoning field of public health tracking to employ this tracking concept in our Center research as a tool to reduce child pesticide exposures and also evaluate our impact on pesticide practices awareness.
FACULTY DEVELOPMENT INVESTIGATOR
As the Faculty Development Investigator, Dr. Karr is developing her research portfolio related to environmental factors, particularly pesticides, and respiratory health of children in agricultural settings. In addition, she is developing her role as a mentor and clinician-educator in pediatric environmental health. Dr. Karr is participating as a co-investigator in the Center’s farmworker family cohort research and has included a respiratory health questionnaire to the ongoing assessment, which will be administered in late summer 2011, in collaboration with the CBPR Project. She will utilize exposure and mechanistic biomarker data on these children to evaluate associations between pesticide exposure, oxidative stress, inflammation, and respiratory health outcomes.
She is active in related and complementary research pursuits and collaborations. In August 2010, she replaced Dr. Matthew Keifer as PI of an NIEHS R-21 grant on ambient environmental factors and asthma when Dr. Keifer left the University of Washington. This is a longitudinal cohort study of children with asthma who reside in the agricultural Yakima Valley of Washington State. Dr. Karr has been asked to participate as a member of the Internal Advisory Panel for the UW NIOSH-sponsored Northwest Agricultural Safety and Health Center.
Dr. Karr engaged a medical student’s involvement in her R21 study for the UW Medical Student Research Training Program (Rebecca Mandell) in summer and fall 2010. Ms. Mandell was selected to present her experience with the study at the Western Student Medical Research Forum in Carmel, CA, in January 2010.
Dr. Karr serves as a core mentor for Ph.D. candidate Martha Perla (UW Health Services) who is conducting dissertation-related research that includes an assessment of the health impacts of pesticides on asthma using NHANES datasets.
Future Activities:
The highlights above illustrate the high volume and quality of the Center’s efforts aimed at reducing the adverse effects of environmental pesticide exposures in children. The Center continues to work to understand the mechanisms that define children’s susceptibility to pesticides, identify the implications of this susceptibility for development and learning, and partner with our communities to translate our findings into risk communication, risk management, and prevention strategies. Center researchers continue to work in the lab, in the field, and in the community to bring a unique and successful approach to the study of children’s environmental health.
Journal Articles: 178 Displayed | Download in RIS Format
| Other center views: | All 510 publications | 227 publications in selected types | All 178 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Armstrong JL, Fenske RA, Yost MG, Galvin K, Tchong-French M, Yu J. Presence of organophosphorus pesticide oxygen analogs in air samples. Atmospheric Environment 2013;66:145-150. |
R834514 (2012) R834514 (2013) R834514 (Final) R834514C002 (Final) |
Exit Exit |
|
|
Armstrong JL, Fenske RA, Yost MG, Tchong-French M, Yu J. Comparison of polyurethane foam and XAD-2 sampling matrices to measure airborne organophosphorus pesticides and their oxygen analogs in an agricultural community. Chemosphere 2013:92(4):451-457. |
R834514 (2013) R834514 (Final) R834514C002 (Final) |
Exit Exit Exit |
|
|
Armstrong JL, Fitzpatrick CF, Loftus CT, Yost MG, Tchong-French M, Karr CJ. Development of a unique multi-contaminant air sampling device for a childhood asthma cohort in an agricultural environment. Environmental Science: Processes & Impacts 2013;15(9):1760-1767. |
R834514 (2013) R834514 (Final) R834514C002 (Final) |
Exit Exit |
|
|
Armstrong JL, Dills RL, Yu J, Yost MG, and Fenske RA. A sensitive LC-MS/MS method for measurement of organophosphorus pesticides and their oxygen analogs in air sampling matrices. Journal of Environmental Science and Health, Part B 2014;49(2):102-108. |
R834514 (Final) R834514C002 (Final) |
Exit |
|
|
Armstrong JL, Yost MG, Fenske RA. Development of a passive air sampler to measure airborne organophosphorus pesticides and oxygen analogs in an agricultural community. Chemosphere 2014;111:135-143. |
R834514 (Final) R834514C002 (Final) |
Exit Exit Exit |
|
|
Asselbergs FW, Guo Y, van Iperen EP, Sivapalaratnam S, Tragante V, Lanktree MB, Lange LA, Almoguera B, Appelman YE, Barnard J, Baumert J, Beitelshees AL, Bhangale TR, Chen YD, Gaunt TR, Gong Y, Hopewell JC, Johnson T, Kleber ME, Langaee TY, Li M, Li YR, Liu K, McDonough CW, Meijs MF, Middelberg RP, Musunuru K, Nelson CP, O'Connell JR, Padmanabhan S, Pankow JS, Pankratz N, Rafelt S, Rajagopalan R, Romaine SP, Schork NJ, Shaffer J, Shen H, Smith EN, Tischfield SE, van der Most PJ, van Vliet-Ostaptchouk JV, Verweij N, Volcik KA, Zhang L, Bailey KR, Bailey KM, Bauer F, Boer JM, Braund PS, Burt A, Burton PR, Buxbaum SG, Chen W, Cooper-Dehoff RM, Cupples LA, deJong JS, Delles C, Duggan D, Fornage M, Furlong CE, Glazer N, Gums JG, Hastie C, Holmes MV, Illig T, Kirkland SA, Kivimaki M, Klein R, Klein BE, Kooperberg C, Kottke-Marchant K, Kumari M, LaCroix AZ, Mallela L, Murugesan G, Ordovas J, Ouwehand WH, Post WS, Saxena R, Scharnagl H, Schreiner PJ, Shah T, Shields DC, Shimbo D, Srinivasan SR, Stolk RP, Swerdlow DI, Taylor HA Jr, Topol EJ, Toskala E, van Pelt JL, van Setten J, Yusuf S, Whittaker JC, Zwinderman AH; LifeLines Cohort Study, Anand SS, Balmforth AJ, Berenson GS, Bezzina CR, Boehm BO, Boerwinkle E, Casas JP, Caulfield MJ, Clarke R, Connell JM, Cruickshanks KJ, Davidson KW, Day IN, de Bakker PI, Doevendans PA, Dominiczak AF, Hall AS, Hartman CA, Hengstenberg C, Hillege HL, Hofker MH, Humphries SE, Jarvik GP, Johnson JA, Kaess BM, Kathiresan S, Koenig W, Lawlor DA, März W, Melander O, Mitchell BD, Montgomery GW, Munroe PB, Murray SS, Newhouse SJ, Onland-Moret NC, Poulter N, Psaty B, Redline S, Rich SS, Rotter JI, Schunkert H, Sever P, Shuldiner AR, Silverstein RL, Stanton A, Thorand B, Trip MD, Tsai MY, van der Harst P, van der Schoot E, van der Schouw YT, Verschuren WM, Watkins H, Wilde AA, Wolffenbuttel BH, Whitfield JB, Hovingh GK, Ballantyne CM, Wijmenga C, Reilly MP, Martin NG, Wilson JG, Rader DJ, Samani NJ, Reiner AP, Hegele RA, Kastelein JJ, Hingorani AD, Talmud PJ, Hakonarson H, Elbers CC, Keating BJ, Drenos F. Large-scale gene-centric meta-analysis across 32 studies identifies multiple lipid loci. American Journal of Human Genetics 2012;91(5):823-838. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Asselbergs, F.W., Y. Guo, E.P. van Iperen, S. Sivapalaratnam, V. et al. Large-Scale Gene-Centric Meta-analysis across 32 Studies Identifies Multiple Lipid Loci. Am J Hum Genet, 2012. 91(5):p. 823-38. |
R834514C004 (Final) R834798C005 (Final) |
not available |
|
|
Baker PE, Cole TB, Cartwright M, Suzuki SM, Thummel KE, Lin YS, Co AL, Rettie AE, Kim JH, Furlong CE. Identifying safer anti-wear triaryl phosphate additives for jet engine lubricants. Chemico-Biological Interactions 2013;203(1):257-264. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit Exit |
|
|
Bal-Price AK, Coecke S, Costa L, Crofton KM, Fritsche E, Goldberg A, Grandjean P, Lein PJ, Li A, Lucchini R, Mundy WR, Padilla S, Persico AM, Seiler AE, Kreysa J. Advancing the science of developmental neurotoxicity (DNT): testing for better safety evaluation. ALTEX 2012;29(2):202-215. |
R834514 (2013) |
Exit Exit |
|
|
Balk SJ, Council on Environmental Health, Section on Dermatology. Technical report—ultraviolet radiation: a hazard to children and adolescents. Pediatrics 2011;127(3):e791-e817. |
R834514 (2011) R834514 (2012) R834514 (Final) |
Exit Exit |
|
|
Below JE, Earl DL, Shively KM, McMillin MJ, Smith JD, Turner EH, Stephan MJ, Al-Gazali LI, Hertecant JL, Chitayat D, Unger S, Cohn DH, Krakow D, Swanson JM, Faustman EM, Shendure J, Nickerson DA, Bamshad MJ, University of Washington Center for Mendelian Genomics. Whole-genome analysis reveals that mutations in inositol polyphosphate phosphatase-like 1 cause opsismodysplasia. American Journal of Human Genetics 2013;92(1):137-143. |
R834514 (2013) |
Exit Exit |
|
|
Besler C, Heinrich K, Rohrer L, Doerries C, Riwanto M, Shih DM, Chroni A, Yonekawa K, Stein S, Schaefer N, Mueller M, Akhmedov A, Daniil G, Manes C, Templin C, Wyss C, Maier W, Tanner FC, Matter CM, Corti R, Furlong C, Lusis AJ, von Eckardstein A, Fogelman AM, Luscher TF, Landmesser U. Mechanisms underlying adverse effects of HDL on eNOS-activating pathways in patients with coronary artery disease. The Journal of Clinical Investigation 2011;121(7):2693-2708. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Buka I, Osornio-Vargas A, Karr C. Melamine food contamination: relevance to Canadian children. Paediatrics and Child Health 2009;14(4):222-224. |
R834514 (2012) R834514 (2013) R834514 (Final) |
|
|
|
Chen J, Zhang XL, Kusumo H, Costa LG, Guizzetti M. 2013. Cholesterol efflux is differentially regulated in neurons and astrocytes:Implications for brain cholesterol homeostasis. Biochimica Et Biophysica Acta-Molecular and Cell Biology of Lipids. 1831(2):263-275.PMC3534809. |
R834514 (2013) |
Exit |
|
|
Clark NA, Demers PA, Karr CJ, Koehoorn M, Lencar C, Tamburic L, Brauer M. Effect of early life exposure to air pollution on development of childhood asthma. Environmental Health Perspectives 2010;118(2):284-290. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
|
|
|
Cole TB, Jansen K, Park S, Li W-F, Furlong CE, Costa LG. The toxicity of mixtures of specific organophosphate compounds is modulated by paraoxonase 1 status. Advances in Experimental Medicine and Biology 2010;660:47-60. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514C004 (Final) R831709 (2007) R831709 (Final) |
Exit Exit |
|
|
Cole TB, Beyer RP, Bammler TK, Park SS, Farin FM, Costa LG, Furlong CE. Repeated developmental exposure of mice to chlorpyrifos oxon is associated with paraoxonase 1 (PON1)-modulated effects on cerebellar gene expression. Toxicological Sciences 2011;123(1):155-169. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Cole TB, Giordano G, Co AL, Mohar I, Kavanagh TJ, Costa LG. Behavioral characterization of GCLM-knockout mice, a model for enhanced susceptibility to oxidative stress. Journal of Toxicology 2011;2011:157687 (7 pp.). |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Cole TB, Fisher JC, Burbacher TM, Costa LG, Furlong CE. Neurobehavioral assessment of mice following repeated postnatal exposure to chlorpyrifos-oxon. Neurotoxicology and Teratology. 2012;34(3):311-22. |
R834514 (Final) R834514C004 (Final) R834693 (2015) |
Exit Exit Exit |
|
|
Cole TB, Fisher JC, Burbacher TM, Costa LG, Furlong CE. Neurobehavioral assessment of mice following repeated postnatal exposure to chlorpyrifos-oxon. Neurotoxicology and Teratology 2012;34(3):311-322. |
R834514 (2011) R834514 (2012) R834514 (2013) R834693 (Final) |
Exit Exit Exit |
|
|
Committee on Environmental Health, Committee on Infectious Diseases, Rogan WJ, Brady MT. Drinking water from private wells and risks to children. Pediatrics 2009;123(6):1599-1605. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Committee on Environmental Health, Tester JM. The built environment: designing communities to promote physical activity in children. Pediatrics 2009;123(6):1591-1598. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Committee on Environmental Health, Committee on Substance Abuse, Committee on Adolescence, Committee on Native American Child Health. Tobacco use: a pediatric disease. Pediatrics 2009;124(5):1474-1484. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Coronado GD, Vigoren EM, Griffith WC, Faustman EM, Thompson B. Organophosphate pesticide exposure among pome and non-pome farmworkers:a subgroup analysis of a community randomized trial. Journal of Occupational and Environmental Medicine 2009;51(4):500-509. |
R834514 (2011) R834514 (Final) R834514C001 (Final) R831709 (2007) R831709 (Final) R832734 (Final) |
Exit |
|
|
Coronado GD, Griffith WC, Vigoren EM, Faustman EM, Thompson B. Where's the dust? Characterizing locations of azinphos-methyl residues in house and vehicle dust among farmworkers with young children. Journal of Occupational and Environmental Hygiene 2010;7(12):663-671. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C001 (Final) R831709 (Final) |
Exit |
|
|
Coronado GD, Holte S, Vigoren E, Griffith WC, Faustman E, Thompson B. Organophosphate pesticide exposure and residential proximity to nearby fields:evidence for the drift pathway. Journal of Occupational and Environmental Medicine 2011;53(8):884-891. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C001 (Final) R831709 (Final) |
Exit |
|
|
Coronado GD, Holte SE, Vigoren EM, Griffith WC, Barr DB, Faustman EM, Thompson B. Do workplace and home protective practices protect farm workers? Findings from the "For Healthy Kids" study. Journal of Occupational and Environmental Medicine 2012;54(9):1163-1169. |
R834514 (2013) R834514 (Final) R834514C001 (Final) |
Exit Exit |
|
|
Costa LG, Giordano G, Guizzetti M. In vitro models to study cell-cell interactions that influence developmental neurotoxicity. ALTEX 2010;27(Special Issue 1):303-308. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Costa LG, Giordano G, Furlong CE. Pharmacological and dietary modulators of paraxonase 1 (PON1) activity and expression: the hunt goes on. Biochemical Pharmacology 2011;81(3):337-344. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Costa LG, Giordano G, Cole TB, Marsillach J, Furlong CE. Paraoxonase 1 (PON1) as a genetic determinant of susceptibility to organophosphate toxicity. Toxicology 2013;307:115-122. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Costa LG. The birth and early years of INA, the International Neurotoxicology Association. Neurotoxicology 2013;36:89-103. |
R834514 (2013) |
Exit Exit |
|
|
Costa LG, Giordano G, Guizzetti M. Inhibition of cholinergic muscarinic signaling by ethanol: potential mechanism of developmental neurotoxicity and biological plausibility for the beneficial effects of choline supplementation. The International Journal of Alcohol and Drug Research 2013;2(3):17-25. |
R834514 (2013) |
Exit Exit |
|
|
Costa LG, Pellacani C, Dao K, Kavanagh TJ, Roque PJ. The brominated flame retardant BDE-47 causes oxidative stress and apoptotic cell death in vitro and in vivo in mice. NeuroToxicology 2015;48:68-76. |
R834514 (2015) R834514 (Final) R834514C003 (2015) R834514C003 (Final) |
Exit Exit Exit |
|
|
Council on Environmental Health, Section on Dermatology. Ultraviolet radiation: a hazard to children and adolescents. Pediatrics 2011;127(3):588-597. |
R834514 (2013) |
Exit Exit Exit |
|
|
Council on Environmental Health. Chemical-management policy: prioritizing children’s health. Pediatrics 2011;127(5):983-990. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Council on Environmental Health. Policy statement: pesticide exposure in children. Pediatrics 2012;130(6):e1757-e1763. |
R834514 (2012) |
Exit Exit |
|
|
Daniell WE, Van Tung L, Wallace RM, Havens DJ, Karr CJ, Bich Diep N, Croteau GA, Beaudet NJ, Duy Bao N. Childhood lead exposure from battery recyling in Vietnam. Biomed Research International 2015;2015:193715. |
R834514 (Final) |
Exit |
|
|
Furlong CE, Suzuki SM, Stevens RC, Marsillach J, Richter RJ, Jarvik GP, Checkoway H, Samii A, Costa LG, Griffith A, Roberts JW, Yearout D, Zabetian CP. Human PON1, a biomarker of risk of disease and exposure. Chemico-Biological Interactions 2010;187(1-3):355-361. |
R834514 (2011) R834514 (2012) R834514 (2013) R831709 (2007) R831709 (Final) |
Exit Exit Exit |
|
|
Furlong CE. Exposure to triaryl phosphates: metabolism and biomarkers of exposure. Journal of Biological Physics and Chemistry 2011;11(4):165-171. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Furlong CE, Marsillach J, Jarvik GP, Costa LG. Paraoxonases-1,-2 and-3: what are their functions? Chemico-Biological Interactions 2016;259(Pt B):51-62. |
R834514 (Final) |
Exit Exit Exit |
|
|
Garrick JM, Dao K, de Laat R, Elsworth J, Cole TB, Marsillach J, Furlong CE, Costa LG. Developmental expression of paraoxonase 2. Chemico-Biological Interactions 2016;259(Pt B):168-174. |
R834514 (Final) |
Exit Exit Exit |
|
|
Gibbs JL, Yost MG, Negrete M, Fenske RA. Passive sampling for indoor and outdoor exposures to chlorpyrifos, azinphos-methyl, and oxygen analogs in a rural agricultural community. Environmental Health Perspectives 2017;125(3):333-341. |
R834514 (Final) R834514C002 (Final) |
|
|
|
Giordano G, Guizzetti M, Dao K, Mattison HA, Costa LG. Ethanol impairs muscarinic receptor-induced neuritogenesis in rat hippocampal slices: role of astrocytes and extracellular matrix proteins. Biochemical Pharmacology 2011;82(11):1792-1799. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Giordano G, Cole TB, Furlong CE, Costa LG. Paraoxonase 2 (PON2) in the mouse central nervous system: a neuroprotective role? Toxicology and Applied Pharmacology 2011;256(3):369-378. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Giordano G, Costa LG. Developmental neurotoxicity: some old and new issues. ISRN Toxicology 2012;2012:814795. |
R834514 (2013) |
Exit Exit |
|
|
Giordano G, Kavanagh TJ, Faustman EM, White CC, Costa LG. Low-level domoic acid protects mouse cerebellar granule neurons from acute neurotoxicity: role of glutathione. Toxicological Sciences 2013;132(2):399-408. |
R834514 (2013) |
Exit Exit Exit |
|
|
Giordano G, Tait L, Furlong CE, Cole TB, Kavanagh TJ, Costa LG. Gender differences in brain susceptibility to oxidative stress are mediated by levels of paraoxonase-2 expression. Free Radical Biology and Medicine 2013;58:98-108. |
R834514 (2013) R834514C004 (Final) |
Exit Exit |
|
|
Griffith W, Curl CL, Fenske RA, Lu CA, Vigoren EM, Faustman EM. Organophosphate pesticide metabolite levels in pre-school children in an agricultural community:within-and between-child variability in a longitudinal study. Environmental Research 2011;111(6):751-756. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C002 (Final) R831709 (Final) |
Exit Exit Exit |
|
|
Guizzetti M, Moore NH, Giordano G, VanDeMark KL, Costa LG. Ethanol inhibits neuritogenesis induced by astrocyte muscarinic receptors. Glia 2010;58(12):1395-1406. |
R834514 (2011) R834514 (2012) R834514 (2013) R831709 (2007) R831709 (Final) |
Exit |
|
|
Guizzetti M, Moore NH, VanDeMark KL, Giordano G, Costa LG. Muscarinic receptor-activated signal transduction pathways involved in the neuritogenic effect of astrocytes in hippocampal neurons. European Journal of Pharmacology 2011;659(2-3):102-107. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Guo Y, Lanktree MB, Taylor KC, Hakonsarson H, Lange LA, Keating BJ, IBC 50K SNP Array BMI Consortium. Gene-centric meta-analyses of 108 912 individuals confirm known body mass index loci and reveal three novel signals. Human Molecular Genetics 2013;22(1):184-201. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Harris S, Hermsen SA, Yu X, Hong SW, Faustman EM. Comparison of toxicogenomic responses to phthalate ester exposure in an organotypic testis co-culture model and responses observed in vivo. Reproductive Toxicology 2015;58:149-159. |
R834514 (Final) R834514C003 (2015) R834514C003 (Final) R835738 (2016) R835738 (2017) R835738C004 (2015) R835738C004 (2017) |
Exit Exit Exit |
|
|
Harris S, Wegner S, Hong SW, Faustman EM. Phthalate metabolism and kinetics in an in vitro model of testis development. Toxicology in Vitro 2016;32:123-131. |
R834514 (Final) R834514C003 (Final) R833772 (2009) R835738 (2016) R835738 (2017) R835738C004 (2015) R835738C004 (2016) R835738C004 (2017) |
Exit Exit Exit |
|
|
Harris S, Shubin SP, Wegner S, Van Ness K, Green F, Hong SW, Faustman EM. The presence of macrophages and inflammatory responses in an in vitro testicular co-culture model of male reproductive development enhance relevance to in vivo conditions. Toxicology In Vitro 2016;36:210-215. |
R834514 (Final) R834514C003 (Final) R835738 (2016) R835738 (2017) R835738C004 (2016) R835738C004 (2017) |
Exit Exit |
|
|
Hofmann JN, Keifer MC, Furlong CE, De Roos AJ, Farin FM, Fenske RA, van Belle G, Checkoway H. Serum cholinesterase inhibition in relation to paraoxonase-1 (PON1) status among organophosphate-exposed agricultural pesticide handlers. Environmental Health Perspectives 2009;117(9):1402-1408. |
R834514 (2012) R834514 (2013) R831709 (Final) |
|
|
|
Hofmann JN, Keifer MC, De Roos AJ, Fenske RA, Furlong CE, van Belle G, Checkoway H. Occupational determinants of serum cholinesterase inhibition among organophosphate-exposed agricultural pesticide handlers in Washington State. Occupational and Environmental Medicine 2010;67(6):375-386. |
R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Hohl SD, Gonzalez C, Carosso E, Ibarra G, Thompson B. "I did it for us and I would do it again": perspectives of rural Latinos on providing biospecimens for research. American Journal of Public Health 2014;104(5):911-916. |
R834514 (Final) R834514C001 (Final) |
Exit Exit |
|
|
Holme F, Thompson B, Holte S, Vigoren EM, Espinoza N, Ulrich A, Griffith W, Faustman EM. The role of diet in children's exposure to organophosphate pesticides. Environmental Research 2016;147:133-140. |
R834514 (Final) R834514C001 (2015) R834514C001 (Final) |
Exit Exit Exit |
|
|
Inoue S, Becker AL, Kim J-H, Shu Z, Soelberg SD, Weigel KM, Hiraiwa M, Cairns A, Lee H-B, Furlong CE, Oh K, Lee K-H, Gao D, Chung J-H, Cangelosi GA. Semi-automated, occupationally safe immunofluorescence microtip sensor for rapid detection of mycobacterium cells in sputum. PLoS One 2014;9(1):e86018. |
R834514 (Final) R834514C004 (2015) |
Exit Exit |
|
|
Jackson JE, Yost MG, Karr CJ, Fitzpatrick C, Lamb BK, Chung S, Chen J, Avise J, Rosenblatt RA, Fenske RA. Public health impacts of climate change in Washington State: projected mortality risks due to heat events and air pollution. Climatic Change 2010;102(1-2):159-186. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C002 (Final) |
Exit |
|
|
Jansen KL, Cole TB, Park SS, Furlong CE, Costa LG. Paraoxonase 1 (PON1) modulates the toxicity of mixed organophosphorus compounds. Toxicology and Applied Pharmacology 2009;236(2):142-153. |
R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Karr CJ, Rudra CB, Miller KA, Gould TR, Larson T, Sathyanarayana S, Koenig JQ. Infant exposure to fine particulate matter and traffic and risk of hospitalization for RSV bronchiolitis in a region with lower ambient air pollution. Environmental Research 2009;109(3):321-327. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Karr CJ, Demers PA, Koehoorn MW, Lencar CC, Tamburic L, Brauer M. Influence of ambient air pollutant sources on clinical encounters for infant bronchiolitis. American Journal of Respiratory and Critical Care Medicine 2009;180(10):995-1001. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Karr CJ. Adding fuel to the fire: increasing evidence for developmental toxicity of indoor solid fuel combustion. Archives of Pediatric & Adolescent Medicine 2011;165(6):565-566. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Karr CJ. Children's environmental health in agricultural settings. Journal of Agromedicine 2012;17(2):127-139. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Karr C. Addressing environmental contaminants in pediatric practice. Pediatrics in Review 2011;32(5):190-200. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Kim DS, Burt AA, Ranchalis JE, Richter RJ, Marshall JK, Eintracht JF, Rosenthal EA, Furlong CE, Jarvik GP. Additional common polymorphisms in the PON gene cluster predict PON1 activity but not vascular disease. Journal of Lipids 2012;2012:476316 (11 pp.). |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Kim DS, Burt AA, Ranchalis JE, Richter RJ, Marshall JK, Nakayama KS, Jarvik ER, Eintracht JF, Rosenthal EA, Furlong CE, Jarvik JP. Dietary cholesterol increases paraoxonase 1 enzyme activity. Journal of Lipid Research 2012;53(11):2450-2458. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Kim DS, Burt AA, Crosslin DR, Robertson PD, Ranchalis JE, Boyko EJ, Nickerson DA, Furlong CE, Jarvik GP. Novel common and rare genetic determinants of paraoxonase activity: FTO, SERPINA12, and ITGAL. Journal of Lipid Research 2013;54(2):552-560. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Kim DS, Burt AA, Ranchalis JE, Jarvik ER, Rosenthal EA, Hatsukami TS, Furlong CE, Jarvik GP. Novel gene-by-environment interactions: APOB and NPC1L1 variants affect the relationship between dietary and total plasma cholesterol. Journal of Lipid Research 2013;54(5):1512-1520. |
R834514 (2013) |
Exit Exit Exit |
|
|
Kim DS, Marsillach J, Furlong CE, Jarvik GP. Pharmacogenetics of paraoxonase activity: elucidating the role of high-density lipoprotein in disease. Pharmacogenomics 2013;14(12):1495-1515. |
R834514 (2013) R834514C004 (Final) |
Exit |
|
|
Kim DS, Burt AA, Rosenthal EA, Ranchalis JE, Eintracht JF, Hatsukami TS, Furlong CE, Marcovina S, Albers JJ, Jarvik GP. HDL-3 is a superior predictor of carotid artery disease in a case-control cohort of 1725 participants. Journal of the American Heart Association 2014;3(3):e000902. |
R834514 (Final) R834514C004 (2015) |
Exit Exit |
|
|
Kim DS, Crosslin DR, Auer PL, Suzuki SM, Marsillach J, Burt AA, Gordon AS, Meschia JF, Nalls MA, Worrall BB, Longstreth Jr. WT, Gottesman RF, Furlong CE, Peters U, Rich SS, Nickerson DA, Jarvik GP. Rare coding variation in paraoxonase-1 is associated with ischemic stroke in the NHLBI Exome Sequencing Project. Journal of Lipid Research 2014;55(6):1173-1178. |
R834514 (Final) R834514C004 (2015) |
Exit Exit Exit |
|
|
Kim DS, Burt AA, Ranchalis JE, Jarvik LE, Eintracht JF, Furlong CE, Jarvik GP. Effects of dietary components on high-density lipoprotein measures in a cohort of 1,566 participants. Nutrition & Metabolism 2014;11(1):44 (9 pp.). |
R834514C004 (2015) |
Exit Exit |
|
|
Kim DS, Burt AA, Ranchalis JE, Vuletic S, Vaisar T, Li WF, Rosenthal EA, Dong W, Eintracht JF, Motulsky AG, Brunzell JD, Albers JJ, Furlong CE, Jarvik GP. PLTP activity inversely correlates with CAAD: effects of PON1 enzyme activity and genetic variants on PLTP activity. Journal of Lipid Research 2015;56(7):1351-1362. |
R834514 (Final) R834514C004 (2015) |
Exit Exit Exit |
|
|
Kim DS, Li YK, Bell GA, Burt AA, Vaisar T, Hutchins PM, Furlong CE, Otvos JD, Polak JF, Arnan MK, Kaufman JD, McClelland RL, Longstreth Jr WT, Jarvik GP. Concentration of smaller high-density lipoprotein particle (HDL-P) is inversely correlated with carotid intima media thickening after confounder adjustment: the Multi Ethnic Study of Atherosclerosis (MESA). Journal of the American Heart Association 2016;5(5):e002977. |
R834514 (Final) |
Exit Exit Exit |
|
|
Kim HY, Wegner SH, Van Ness KP, Park JJ, Pacheco SE, Workman T, Hong S, Griffith W, Faustman EM. Differential epigenetic effects of chlorpyrifos and arsenic in proliferating and differentiating human neural progenitor cells. Reproductive Toxicology 2016;65:212-223. |
R834514 (Final) R834514C003 (2015) R834514C003 (Final) |
Exit Exit Exit |
|
|
Kim JH, Stevens RC, MacCoss MJ, Goodlett DR, Scherl A, Richter RJ, Suzuki SM, Furlong CE. Identification and characterization of biomarkers of organophosphorus exposures in humans. Advances in Experimental Medicine and Biology 2010;660:61-71. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit |
|
|
Kim JH, Yeo WH, Shu Z, Soelberg SD, Inoue S, Kalyanasundaram D, Ludwig J, Furlong CE, Riley JJ, Weigel KM, Cangelosi GA, Oh K, Lee KH, Gao D, Chung JH. Immunosensor towards low-cost, rapid diagnosis of tuberculosis. Lab on a Chip 2012;12(8):1437-1440. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Kim NJ, Vasquez VB, Torres E, Nicola RM, Karr C. Breaking the silence: sexual harassment of Mexican women farmworkers. Journal of Agromedicine 2016;21(2):154-162. |
R834514 (Final) |
Exit |
|
|
Kullman SW, Hamm JT, Hinton DE. Identification and characterization of a cDNA encoding cytochrome P450 3A from the fresh water teleost medaka (Oryzias latipes). Archives of Biochemistry and Biophysics 2000;380(1):29-38. |
R834514 (2011) R834514 (Final) R825298 (Final) |
Exit Exit |
|
|
Lanktree MB, Guo Y, Murtaza M, Glessner JT, Bailey SD, Onland-Moret NC, Lettre G, Ongen H, Rajagopalan R, Johnson T, Shen H, Nelson CP, Klopp N, Baumert J, Padmanabhan S, Pankratz N, Pankow JS, Shah S, Taylor K, Barnard J, Peters BJ, Maloney CM, Lobmeyer MT, Stanton A, Zafarmand MH, Romaine SP, Mehta A, van Iperen EP, Gong Y, Price TS, Smith EN, Kim CE, Li YR, Asselbergs FW, Atwood LD, Bailey KM, Bhatt D, Bauer F, Behr ER, Bhangale T, Boer JM, Boehm BO, Bradfield JP, Brown M, Braund PS, et al. Meta-analysis of dense genecentric association studies reveals common and uncommon variants associated with height. American Journal of Human Genetics 2011;88(1):6-18. |
R834514 (2011) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Liyasova M, Li B, Schopfer LM, Nachon F, Masson P, Furlong CE, Lockridge O. Exposure to tri-o-cresyl phosphate detected in jet airplane passengers. Toxicology and Applied Pharmacology 2011;256(3):337-347. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Loftus C, Yost M, Sampson P, Arias G, Torres E, Vasquez VB, Bhatti P, Karr C. Regional PM2.5 and asthma morbidity in an agricultural community: a panel study. Environmental Research 2015;136:505-512. |
R834514 (2015) R834514 (Final) R834514C002 (2015) R834514C002 (Final) |
Exit Exit Exit |
|
|
Loftus C, Yost M, Sampson P, Torres E, Arias G, Breckwich Vasquez V, Hartin K, Armstrong J, Tchong French M, Vedal S, Bhatti P, Karr C. Ambient ammonia exposures in an agricultural community and pediatric asthma morbidity. Epidemiology 2015;26(6):794-801. |
R834514 (2015) |
Exit |
|
|
MacIntyre EA, Karr CJ, Koehoorn M, Demers P, Tamburic L, Lencar C, Brauer M. Otitis media incidence and risk factors in a population-based birth cohort. Paediatrics and Child Health 2010;15(7):437-442. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
MacIntyre EA, Karr CJ, Koehoorn M, Demers PA, Tamburic L, Lencar C, Brauer M. Residential air pollution and otitis media during the first two years of life. Epidemiology 2011;22(1):81-89. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Mackenzie-Ross SJ, Brewin CR, Curran HV, Furlong CE, Abraham-Smith KM, Harrison V. Neuropsychological and psychiatric functioning in sheep farmers exposed to low levels of organophosphate pesticides. Neurotoxicology and Teratology 2010;32(4):452-459. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Malen R, Knerr S, Delgado F, Fullerton SM, Thompson B. Rural Mexican-Americans’ perceptions of family health history, genetics, and disease risk: implications for disparities-focused research dissemination. Journal of Community Genetics 2016;7(1):91-96. |
R834514 (Final) R834514C001 (Final) |
Exit Exit |
|
|
Marsillach J, Richter RJ, Kim JH, Stevens RC, MacCoss MJ, Tomazela D, Suzuki SM, Schopfer LM, Lockridge O, Furlong CE. Biomarkers of organophosphorus (OP) exposures in humans. NeuroToxicology 2011;32(5):656-660. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Marsillach J, Hsieh EJ, Richter RJ, MacCoss MJ, Furlong CE. Proteomic analysis of adducted butyrylcholinesterase for biomonitoring organophosphorus exposures. Chemico-Biological Interactions 2013;203(1):85-90. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Marsillach J, Costa LG, Furlong CE. Protein adducts as biomarkers of exposure to organophosphorus compounds. Toxicology 2013;307:46-54. |
R834514 (2013) R834514C004 (Final) |
Exit Exit |
|
|
Marsillach J, Suzuki SM, Richter RJ, McDonald MG, Rademacher PM, MacCoss MJ, Hsieh EJ, Rettie AE, Furlong CE. Human valacyclovir hydrolase/biphenyl hydrolase-like protein is a highly efficient homocysteine thiolactonase. PLoS One 2014;9(10):e110054. |
R834514 (Final) R834514C004 (2015) |
Exit Exit |
|
|
Marsillach J, Becker JO, Vaisar T, Hahn BH, Brunzell JD, Furlong CE, deBoer IH, McMahon MA, Hoofnagle AN, DCCT/EDIC Research Group. Paraoxonase-3 is depleted from the high-density lipoproteins of autoimmune disease patients with subclinical atherosclerosis. Journal of Proteome Research 2015;14(5):2046-2054. |
R834514 (Final) R834514C004 (2015) |
Exit Exit Exit |
|
|
Marsillach J, Costa LG, Furlong CE. Paraoxonase-1 and early-life environmental exposures. Annals of Global Health 2016;82(1):100-110. |
R834514 (Final) |
Exit Exit Exit |
|
|
McMillin MJ, Below JE, Shively KM, Beck AE, Gildersleeve HI, Pinner J, Gogola GR, Hecht JT, Grange DK, Harris DJ, Earl DL, Jagadeesh S, Mehta SG, Robertson SP, Swanson JM, Faustman EM, Mefford HC, Shendure J, Nickerson DA, Bamshad MJ, University of Washington Center for Mendelian Genomics. Mutations in ECEL1 cause distal arthrogryposis type 5D. American Journal of Human Genetics 2013;92(1):150-156. |
R834514 (2013) |
Exit Exit |
|
|
Mehta AJ, Zanobetti A, Bind MA, Kloog I, Koutrakis P, Sparrow D, Vokonas PS, Schwartz JD. Long-term exposure to ambient fine particulate matter and renal function in older men: the Veterans Administration Normative Aging Study. Environmental Health Perspectives 2016;124(9):1353-1360. |
R834514C004 (Final) R834798 (Final) R834798C002 (Final) |
|
|
|
Moreira EG, Yu X., Robinson JF, Griffith W, Hong SW, Beyer RP, Bammler TK, Faustman EM. Toxicogenomic profiling in maternal and fetal rodent brains following gestational exposure to chlorpyrifos. Toxicology and Applied Pharmacology 2010;245(3):310-325. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C003 (Final) R831709 (2007) R831709 (Final) |
Exit Exit Exit |
|
|
Nonnenmann MW, Coronado G, Thompson B, Griffith WC, Hanson JD, Vesper S, Faustman EM. Utilizing pyrosequencing and quantitative PCR to characterize fungal populations among house dust samples. Journal of Environmental Monitoring 2012;14(8):2038-2043. |
R834514 (2013) R834514 (Final) |
Exit |
|
|
O’Toole TE, Conklin DJ, Bhatnagar A. Environmental risk factors for heart disease. Reviews on Environmental Health 2008;23(3):167-202. |
R834514 (2011) R834514 (Final) EM833367 (2009) EM833367 (Final) |
Exit |
|
|
Paulson JA, Karr CJ, Seltzer JM, Cherry DC, Sheffield PE, Cifuentes E, Buka I, Amler RW. Development of the pediatric environmental health specialty unit network in North America. American Journal of Public Health 2009;99(Suppl 3):S511-S516. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Perkins A, Walters F, Sievert J, Rhodes B, Morrissey B, Karr CJ. Home use of a pyrethroid-containing pesticide and facial paresthesia in a toddler:a case report. International Journal of Environmental Research and Public Health 2016;13(8):829 (7 pp.). |
R834514 (Final) |
Exit Exit Exit |
|
|
Perla ME, Rue T, Cheadle A, Krieger J, Karr CJ. Population-based comparison of biomarker concentrations for chemicals of concern among Latino-American and non-Hispanic white children. Journal of Immigrant and Minority Health 2015;17(3):802-819. |
R834514 (2015) |
Exit |
|
|
Perla ME, Iman E, Campos L, Perkins A, Liebman AK, Miller ME, Beaudet NJ, Karr CJ. Agricultural occupational health and safety perspectives among Latino-American youth. Journal of Agromedicine 2015;20(2):167-177. |
R834514 (2015) |
|
|
|
Perla ME, Rue T, Cheadle A, Krieger J, Karr CJ. Biomarkers of insecticide exposure and asthma in children: a National Health and Nutrition Examination Survey (NHANES) 1999-2008 analysis. Archives of Environmental & Occupational Health 2015;70(6):309-322. |
R834514 (2015) |
Exit |
|
|
Pizzurro DM, Dao K, Costa LG. Astrocytes protect against diazinon- and diazoxon-induced inhibition of neurite outgrowth by regulating neuronal glutathione. Toxicology 2014;318:59-68. |
R834514C003 (Final) |
Exit Exit Exit |
|
|
Pizzurro DM, Dao K, Costa LG. Diazinon and diazoxon impair the ability of astrocytes to foster neurite outgrowth in primary hippocampal neurons. Toxicology and Applied Pharmacology 2014;274(3):372-382. |
R834514C003 (Final) |
Exit Exit Exit |
|
|
Plascak JJ, Molina Y, Wu-Georges S, Idris A, Thompson B. Latino residential segregation and self-rated health among Latinos: Washington State Behavioral Risk Factor Surveillance System, 2012-2014. Social Science & Medicine 2016;159:38-47. |
R834514 (Final) R834514C001 (Final) |
Exit Exit |
|
|
Postma J, Karr C, Kieckhefer G. Community health workers and environmental interventions for children with asthma: a systematic review. Journal of Asthma 2009;46(6):564-576. |
R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Postma J, Younglove LR, Odom-Maryon T, Brooks K, Burbacher TM, Butterfield PW, Butterfield PG, Cederblom N, Grant K, Beresford SAA, Faustman EM. Hispanic representation in a longitudinal birth cohort study. Journal of Health Disparities Research and Practice 2016;9(2):66-77. |
R834514 (Final) |
Exit Exit |
|
|
Raanan R, Gunier RB, Balmes JR, Beltran AJ, Harley KG, Bradman A, Eskenazi B. Elemental sulfur use and associations with pediatric lung function and respiratory symptoms in an agricultural community (California, USA). Environmental Health Perspectives 2017;125(8):087007 (8 pp.). |
R834514 (Final) R826709 (2002) R831710 (Final) R834513 (Final) |
|
|
|
Ramaprasad J, Tsai MG, Fenske RA, Faustman EM, Griffith WC, Felsot AS, Elgethun K, Weppner S, Yost MG. Children's inhalation exposure to methamidophos from sprayed potato fields in Washington State:exploring the use of probabilistic modeling of meteorological data in exposure assessment. Journal of Exposure Science and Environmental Epidemiology 2009;19(6):613-623. |
R834514 (Final) R834514C002 (Final) R831709 (2007) R831709 (Final) |
Exit Exit |
|
|
Richter RJ, Jarvik GP, Furlong CE. Paraoxonase 1 (PON1) status and substrate hydrolysis. Toxicology and Applied Pharmacology 2009;235(1):1-9. |
R834514 (2012) R834514 (2013) R834514 (Final) R831709 (2007) R831709 (Final) |
Exit Exit Exit |
|
|
Richter RJ, Jarvik GP, Furlong CE. Paraoxonase 1 status as a risk factor for disease or exposure. Advances in Experimental Medicine and Biology 2010;660:29-35. |
R834514 (2011) R834514 (2013) |
Exit |
|
|
Roberts JR, Karr CJ, Council on Environmental Health. Pesticide exposure in children. Pediatrics 2012;130(6):e1765-e1788. |
R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Roberts JR, Karr CJ, de Ybarrondo L, McCurdy LE, Freeland KD, Husley TC, Forman J. Improving pediatrician knowledge about environmental triggers of asthma. Clinical Pediatrics 2013;52(6):527-533. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Roberts JR, Newman N, McCurdy LE, Chang JS, Salas MA, Eskridge B, De Ybarrondo L, Sandel M, Mazur L, Karr CJ. Integrating environmental management of asthma into pediatric health care: what worked and what still needs improvement? Clinical Pediatrics 2016;55(14):1271-1278. |
R834514 (Final) |
Exit Exit |
|
|
Robinson JF, Guerrette Z, Yu X, Hong S, Faustman EM. A systems-based approach to investigate dose-and time-dependent methylmercury-induced gene expression response in C57BL/6 mouse embryos undergoing neurulation. Birth Defects Research. Part B: Developmental and Reproductive Toxicology 2010;89(3):188-200. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C003 (Final) R831709 (Final) |
Exit |
|
|
Robinson JF, Yu X, Hong S, Zhou C, Kim N, DeMasi D, Faustman EM. Embryonic toxicokinetic and dynamic differences underlying strain sensitivity to cadmium during neurulation. Reproductive Toxicology 2010;29(3):279-285. |
R834514 (2011) R834514 (2012) R834514 (2013) R831709 (2007) R831709 (Final) |
Exit Exit Exit |
|
|
Robinson JF, Port JA, Yu X, Faustman EM. Integrating genetic and toxicogenomic information for determining underlying susceptibility to developmental disorders. Birth Defects Research. Part A--Clinical and Molecular Teratology 2010;88(10):920-930. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit |
|
|
Robinson JF, Griffith WC, Yu X, Hong S, Kim E, Faustman EM. Methylmercury induced toxicogenomic response in C57 and SWV mouse embryos undergoing neural tube closure. Reproductive Toxicology 2010;30(2):284-291. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C003 (Final) R831709 (Final) |
Exit Exit Exit |
|
|
Robinson JF, Yu X, Moreira EG, Hong S, Faustman EM. Arsenic-and cadmium-induced toxicogenomic response in mouse embryos undergoing neurulation. Toxicology and Applied Pharmacology 2011;250(2):117-129. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C003 (Final) R831709 (Final) |
Exit Exit Exit |
|
|
Robinson JF, Theunissen PT, van Dartel DA, Pennings JL, Faustman EM, Piersma AH. Comparison of MeHg-induced toxicogenomic responses across in vivo and in vitro models used in developmental toxicology. Reproductive Toxicology 2011;32(2):180-188. |
R834514 (Final) R834514C003 (Final) |
Exit Exit Exit |
|
|
Sathyanarayana S, Basso O, Karr CJ, Lozano P, Alavanja M, Sandler DP, Hoppin JA. Maternal pesticide use and birth weight in the Agricultural Health Study. Journal of Agromedicine 2010;15(2):127-136. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) |
Exit |
|
|
Sathyanarayana S, Zhou C, Rudra CB, Gould T, Larson T, Koenig J, Karr CJ. Prenatal ambient air pollution exposure and small for gestational age birth in the Puget Sound Air Basin. Air Quality, Atmosphere & Health 2013;6(2):455-463. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Saxena R, Elbers CC, Guo Y, Peter I, Gaunt TR, Mega JL, Lanktree MB, Tare A, Castillo BA, Li YR, Johnson T, Bruinenberg M, Gilbert-Diamond D, Rajagopalan R, Voight BF, Balasubramanyam A, Barnard J, Bauer F, Baumert J, Bhangale T, Bohm BO, Braund PS, Burton PR, Chandrupatla HR, Clarke R, Cooper-DeHoff RM, Crook ED, Davey-Smith G, Day IN, de Boer A, de Groot MC, Drenos F, Ferguson J, Fox CS, Furlong CE, Gibson Q, Gieger C, Gilhuijs-Pederson LA, Glessner JT, Goel A, Gong Y, Grant SF, Grobbee DE, Hastie C, Humphries SE, Kim CE, Kivimaki M, Kleber M, Meisinger C, Kumari M, Langaee TY, Lawlor DA, Li M, Lobmeyer MT, Maitland-van der Zee AH, Meijs MF, Molony CM, Morrow DA, Murugesan G, Musani SK, Nelson CP, Newhouse SJ, O'Connell JR, Padmanabhan S, Palmen J, Patel SR, Pepine CJ, Pettinger M, Price TS, Rafelt S, Ranchalis J, Rasheed A, Rosenthal E, Ruczinski I, Shah S, Shen H, Silbernagel G, Smith EN, Spijkerman AW, Stanton A, Steffes MW, Thorand B, Trip M, van der Harst P, van der A DL, van Iperen EP, van Setten J, van Vliet-Ostaptchouk JV, Verweij N, Wolffenbuttel BH, Young T, Zafarmand MH, Zmuda JM; Look AHEAD Research Group; DIAGRAM consortium, Boehnke M, Altshuler D, McCarthy M, Kao WH, Pankow JS, Cappola TP, Sever P, Poulter N, Caulfield M, Dominiczak A, Shields DC, Bhatt DL, Zhang L, Curtis SP, Danesh J, Casas JP, van der Schouw YT, Onland-Moret NC, Doevendans PA, Dorn GW 2nd, Farrall M, FitzGerald GA, Hamsten A, Hegele R, Hingorani AD, Hofker MH, Huggins GS, Illig T, Jarvik GP, Johnson JA, Klungel OH, Knowler WC, Koenig W, März W, Meigs JB, Melander O, Munroe PB, Mitchell BD, Bielinski SJ, Rader DJ, Reilly MP, Rich SS, Rotter JI, Saleheen D, Samani NJ, Schadt EE, Shuldiner AR, Silverstein R, Kottke-Marchant K, Talmud PJ, Watkins H, Asselbergs FW, de Bakker PI, McCaffery J, Wijmenga C, Sabatine MS, Wilson JG, Reiner A, Bowden DW, Hakonarson H, Siscovick DS, Keating BJ. Large-scale gene-centric meta-analysis across 39 studies identifies type 2 diabetes loci. American Journal of Human Genetics 2012;90(3):410-425. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Schopfer LM, Furlong CE, Lockridge O. Development of diagnostics in the search of an explanation of aerotoxic syndrome. Analytical Biochemistry 2010;404(1):64-74. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Serrano SE, Karr CJ, Seixas NS, Nguyen RH, Barrett ES, Janssen S, Redmon B, Swan SH, Sathyanarayana S. Dietary phthalate exposure in pregnant women and the impact of consumer practices. International Journal of Environmental Research and Public Health 2014;11(6):6193-6215. PMID:24927036. |
R834514 (2015) |
Exit Exit |
|
|
Shepherd-Banigan M, Ulrich A, Thompson B. Macro-activity patterns of farmworker and non-farmworker children living in an agricultural community. Environmental Research 2014;132:176-181. |
R834514 (Final) R834514C001 (Final) |
Exit Exit Exit |
|
|
Shepherd-Banigan M, Hohl SD, Vaughan C, Ibarra G, Carosso E, Thompson B. "The promotora explained everything": participant experiences during a household-level diabetes education program. The Diabetes Educator 2014;40(4):507-515. |
R834514C001 (2015) |
Exit |
|
|
Smith MN, Griffith WC, Beresford SA, Vredevoogd M, Vigoren EM, Faustman EM. Using a biokinetic model to quantify and optimize cortisol measurements for acute and chronic environmental stress exposure during pregnancy. Journal of Exposure Science and Environmental Epidemiology 2014;24(5):510-516. |
R834514 (2013) R834514 (Final) |
Exit |
|
|
Smith MN, Wilder CS, Griffith WC, Workman T, Thompson B, Dills R, Onstad G, Vredevoogd M, Vigoren EM, Faustman EM. Seasonal variation in cortisol biomarkers in Hispanic mothers living in an agricultural region. Biomarkers 2015;20(5):299-305. |
R834514 (2015) R834514 (Final) R834514C001 (Final) |
Exit Exit |
|
|
Smith MN, Grice J, Cullen A, Faustman EM. A toxicological framework for the prioritization of Children’s Safe Product Act data. International Journal of Environmental Research and Public Health 2016;13(4):431 (24 pp.). |
R834514 (Final) R835738 (2016) R835738 (2017) R835738C005 (2015) R835738C005 (2016) R835738C005 (2017) |
Exit Exit Exit |
|
|
Smith MN, Workman T, McDonald KM, Vredevoogd MA, Vigoren EM, Griffith WC, Thompson B, Coronado GD, Barr D, Faustman EM. Seasonal and occupational trends of five organophosphate pesticides in house dust. Journal of Exposure Science and Environmental Epidemiology 2017;27(4):372-378. |
R834514 (Final) |
Exit |
|
|
Smith M, Hubal E, Faustman E. A Case study on the utility of predictive toxicology tools in alternatives assessments for hazardous chemicals in children's consumer products. JOURNAL OF EXPOSURE SCIENCE AND ENVIRONMENTAL EPIDEMIOLOGY 2020;30(1):160-170. |
R834514 (Final) |
Exit |
|
|
Souza-Nogueira A, Camargo AE, Remondi FA, Paoliello MM, Richter RJ, Furlong CE, Barbosa DS, Maes M, Moreira EG. Paraoxonase 1 (PON1) Q192R genotypes and their interaction with smoking strongly increase atherogenicity and the Framingham risk score. Archives of Endocrinology and Metabolism 2016;60(5):426-435. |
R834514 (Final) |
Exit Exit |
|
|
Sridharan G, Wamalwa D, John-Stewart G, Tapia K, Langat A, Moraa Okinyi H, Adhiambo J, Chebet D, Maleche-Obimbo E, Karr CJ, Benki-Nugent S. High viremia and wasting before antiretroviral therapy are associated with pneumonia in early-treated HIV-infected Kenyan infants. Journal of the Pediatric Infectious Diseases Society 2017;6(3):245-252. |
R834514 (Final) |
Exit Exit |
|
|
Stanaway IB, Wallace JC, Shojaie A, Griffith WC, Hong S, Wilder CS, Green FH, Tsai J, Knight M, Workman T, Vigoren EM, McLean JS, Thompson B, Faustman EM. Human oral buccal microbiomes are associated with farmworker status and azinphos-methyl agricultural pesticide exposure. Applied and Environmental Microbiology 2017;83(2):e02149-16. |
R834514 (Final) |
Exit Exit Exit |
|
|
Stanaway I, Wallace J, Hong S, WIlder C, Green F, Tsai J, Knight M, Workman T, Vigoren E, Smith M, Griffith W, Thompson B, Shojaie A, Faustman E. Alteration of oral microbiome composition in children living with pesticide-exposed farm workers. INTERNATIONAL JOURNAL OF HYGIENE AND ENVIRONMENTAL HEALTH 2023;248(114090). |
R834514 (Final) R832733 (Final) |
Exit |
|
|
Strong LL, Thompson B, Koepsell TD, Meischke H, Coronado GD. Reducing the take-home pathway of pesticide exposure:behavioral outcomes from the Para Niños Saludables study. Journal of Occupational and Environmental Medicine 2009;51(8):922-933. |
R834514 (Final) R834514C001 (Final) R831709 (2007) R831709 (Final) |
Exit |
|
|
Strong LL, Starks HE, Meischke H, Thompson B. Perspectives of mothers in farmworker households on reducing the take-home pathway of pesticide exposure. Health Education & Behavior 2009;36(5):915-929. |
R834514 (Final) R834514C001 (Final) |
Exit Exit Exit |
|
|
Suzuki SM, Stevens RC, Richter RJ, Cole TB, Park S, Otto TC, Cerasoli DM, Lenz DE, Furlong CE. Engineering human PON1 in an E. coli expression system. Advances in Experimental Medicine and Biology 2010;660:37-45. |
R834514 (2011) R834514 (2012) R834514 (2013) R831709 (2007) R831709 (Final) |
Exit |
|
|
Thompson B, Griffith WC, Barr DB, Coronado GD, Vigoren EM, Faustman EM. Variability in the take-home pathway: farmworkers and non-farmworkers and their children. Journal of Exposure Science & Environmental Epidemiology 2014;24(5):522-531. |
R834514 (Final) R834514C001 (Final) |
Exit Exit |
|
|
Ticozzi N, LeClerc AL, Keagle PJ, Glass JD, Wills AM, van Blitterswijk M, Bosco DA, Rodriguez-Levva I, Gellera C, Ratti A, Taroni F, McKenna-Yasek D, Sapp PC, Silani V, Furlong CE, Brown Jr. RH, Landers JE. Paraoxonase gene mutations in amyotrophic lateral sclerosis. Annals of Neurology 2010;68(1):102-107. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit |
|
|
Wegner SH, Yu X, Pacheco Shubin S, Griffith WC, Faustman EM. Stage-specific signaling pathways during murine testis development and spermatogenesis: a pathway-based analysis to quantify developmental dynamics. Reproductive Toxicology 2015;51:31-39. |
R834514 (2015) R834514 (Final) R834514C003 (2015) R834514C003 (Final) R835738 (2016) R835738 (2017) R835738C004 (2015) R835738C004 (2017) |
Exit Exit Exit |
|
|
Wegner S, Hong S, Yu X, Faustman EM. Preparation of rodent testis co-cultures. Current Protocols in Toxicology 2013;Chapter 16:Unit 16.10. |
R834514 (2013) |
Exit |
|
|
Wegner S, Yu X, Kim HY, Harris S, Griffith WC, Hong S, Faustman EM. Effect of dipentyl phthalate in 3-dimensional in vitro testis co-culture is attenuated by cyclooxygenase-2 inhibition. Journal of Toxicology and Environmental Health Sciences 2014;6(8):161-169. |
R834514 (2015) R834514 (Final) R834514C003 (2015) R834514C003 (Final) |
Exit Exit |
|
|
Wegner S, Park J, Workman T, Workman T, Hermsan S, Wallace J, Stanaway I, Kim H, Griffith W, Hong S, Faustman E. Anchoring a dynamic in vitro model of human neuronal differentiation to key processes of early brain development in vivo. Reproductive Toxicology 2020;91:116-130. |
R834514 (Final) R835738 (2018) |
Exit Exit |
|
|
Wegner S, Workman T, Park J, Harris S, Wallace J, Stanaway I, Hong S, Hansen B, Griffith W, Faustman E. A dynamic in vitro developing testis model reflects structures and functions of testicular development in vivo. REPRODUCTIVE TOXICOLOGY 2023;118(108362) |
R834514 (Final) R835738 (Final) |
Exit |
|
|
Weiner MW, Meyerhoff DJ, Neylan TC, Hlavin J, Ramage ER, McCoy D, Studholme C, Cardenas V, Marmar C, Truran D, Chu PW, Kornak J, Furlong CE, McCarthy C. The relationship between Gulf War war illness, brain N-acetylaspartate, and post-traumatic stress disorder. Military Medicine 2011;176(8):896-902. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit |
|
|
Weldon BA, Shubin SP, Smith MN, Workman T, Artemenko A, Griffith WC, Thompson B, Faustman EM. Urinary microRNAs as potential biomarkers of pesticide exposure. Toxicology and Applied Pharmacology 2016;312:19-25. |
R834514 (Final) R834514C001 (Final) |
Exit Exit Exit |
|
|
Whitely Binder LC, Barcelos JK, Booth DB, Darzen M, Elsner MM, Fenske R, Graham TF, Hamlet AF, Hodges-Howell J, Jackson JE, Karr C, Keys PW, Littell JS, Mantua N, Marlow J, McKenzie D, Robinson-Dorn M, Rosenberg EA, Stockle CO, Vano JA. Preparing for climate change in Washington State. Climatic Change 2010;102(1-2):351-376. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C002 (Final) |
Exit Exit |
|
|
Woodruff TJ, Parker JD, Darrow LA, Slama R, Bell ML, Choi H, Glinianaia S, Hoggatt KJ, Karr CJ, Lobdell DT, Wilhelm M. Methodological issues in studies of air pollution and reproductive health. Environmental Research 2009;109(3):311-320. |
R834514 (2012) R834514 (2013) R834514 (Final) R833863 (2009) |
Exit Exit Exit |
|
|
Yu X, Robinson JF, Sidhu JS, Hong S, Faustman EM. A system-based comparison of gene expression reveals alterations in oxidative stress, disruption of ubiquitin-proteasome system and altered cell cycle regulation after exposure to cadmium and ethylmercury in mouse embryonic fibroblast. Toxicological Sciences 2010;114(2):356-377. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R834514C003 (Final) R831709 (2007) R831709 (Final) |
Exit Exit |
|
|
Yu X, Sidhu JS, Hong S, Robinson JF, Ponce RA, Faustman EM. Cadmium induced p53-dependent activation of stress signaling, accumulation of ubiquitinated proteins, and apoptosis in mouse embryonic fibroblast cells. Toxicological Sciences 2011;120(2):403-412. |
R834514 (2011) R834514 (2012) R834514 (2013) R834514 (Final) R831709 (Final) |
Exit Exit |
|
|
Zachek CM, Karr CJ, Daniell W, Sweeney C, Miller MD. A network of Pediatric Environmental Health Specialty Units (PEHSUs): filling a critical gap in the health care system. Medycyna Środowiskowa--Environmental Medicine 2012;15(3):9-20. |
R834514 (2012) R834514 (2013) R834514 (Final) |
Exit Exit |
|
|
Zhou C, Chen J, Zhang X, Costa LG, Guizzetti M. Prenatal ethanol exposure up-regulates the cholesterol transporters ATP-binding cassette A1 and G1 and reduces cholesterol levels in the developing rat brain. Alcohol and Alcoholism 2014;49(6):626-634. |
R834514 (2015) R834514 (Final) R834514C003 (2015) R834514C003 (Final) |
Exit Exit Exit |
|
|
Marsillach, J., E.J. Hsieh, R.J. Richter, M.J. Maccoss, and C.E. Furlong, Proteomic analysis of adducted butyrylcholinesterase for biomonitoring organophosphorus exposures. Chem Biol Interact, 2012. |
R834514C004 (Final) |
not available |
|
|
Kim, DS, Crosslin, DR, Auer, PL, Suzuki, SM, Marsillach, J, Burt, AA, Gordon, AS, Meschia, JF, Nalls, MA, Worrall, BB, Longstreth, WT, Jr., Gottesman, RF, Furlong, CE, Peters, U, Rich, SS, Nickerson, DA and Jarvik, GP. 2014. Rare coding variation in paraoxonase-1 is associated with ischemic stroke in the NHLBI Exome Sequencing Project. J Lipid Res. 55(6):1173-1178. 4031948. |
R834514C004 (Final) |
not available |
|
|
Guizzetti, M., N.H. Moore, G. Giordano, K.L. VanDeMark, and L.G. Costa, Ethanol inhibits neuritogenesis induced by astrocyte muscarinic receptors. Glia, 2010. 58(12):p. 1395-406. PMCID2925144. |
R834514C004 (Final) |
not available |
|
|
Richter RJ, Jarvik GP, Furlong CE. 2010. Paraoxonase 1 (PON1) Status as a Risk Factor for Disease or Exposure. Adv Exp Med Biol 660:29-35. PMID 20221868. DOI 10.1007/978-1-60761-350-3_. NIHMSID #193805. |
R834514C004 (Final) |
not available |
|
|
Kim, DS, Burt, A, Ranchalis, J, Richter, RJ, Marshall, E, Rosenthal, E, Furlong, C and Jarvik, GP. 2012. Additional common polymorphisms in the PON gene cluster predict PON1 activity but not vascular disease. J Lipids 2012:476316. PMCID:PMC3364586. |
R834514C004 (Final) |
not available |
|
|
Kim, DS, Burt, AA, Ranchalis, JE, Richter, RJ, Marshall, JK, Nakayama, KS, Jarvik, ER, Eintracht, JF, Rosenthal, EA, Furlong, CE and Jarvik, GP. 2012. Dietary cholesterol increases paraoxonase 1 enzyme activity. J Lipid Res. 53(11):2450-8. PMCID:PMC3466014. |
R834514C004 (Final) |
not available |
|
|
Kim, DS, Burt, AA, Crosslin, DR, Robertson, PD, Ranchalis, JE, Boyko, EJ, Nickerson, DA, Furlong, CE and Jarvik, GP. 2013. Novel common and rare genetic determinants of paraoxonase activity:FTO, SERPINA12, and ITGAL. J Lipid Res. 54(2):552-60. PMCID:PMC3588879. |
R834514C004 (Final) |
not available |
|
|
Costa, LG, de Laat, R, Dao, K, Pellacani, C, Cole, TB and Furlong, CE. 2013. Paraoxonase-2 (PON2) in brain and its potential role in neuroprotection. Neurotoxicology. 3942372. |
R834514C004 (Final) |
not available |
|
|
Marsillach, J, Hsieh, EJ, Richter, RJ, MacCoss, MJ, Furlong, CE. 2013. Proteomic analysis of adducted butyrylcholinesterase for biomonitoring organophosphorus exposures. Chem Biol Interact 203(1):85-90. |
R834514C004 (Final) |
not available |
|
|
Costa, LG, Tait, L, de Laat, R, Dao, K, Giordano, G, Pellacani, C, Cole, TB and Furlong, CE. 2013. Modulation of paraoxonase 2 (PON2) in mouse brain by the polyphenol quercetin:a mechanism of neuroprotection? Neurochem Res. 38(9):1809-18. 3735620. |
R834514C004 (Final) |
not available |
|
|
Costa, LG, Giordano, G, Cole, TB, Marsillach, J and Furlong, CE. 2013. Paraoxonase 1 (PON1) as a genetic determinant of susceptibility to organophosphate toxicity. Toxicology. 307(10):115-122. PMCID:PMC3516631. |
R834514C004 (Final) |
not available |
|
|
Cole, TB, Li, WF, Co, AL, Hay, AM, MacDonald, JW, Bammler, TK, Farin, FM, Costa, LG and Furlong, CE. 2014. Repeated Gestational Exposure of Mice to Chlorpyrifos Oxon is Associated with Paraoxonase 1 (PON1)-Modulated Effects in Maternal and Fetal Tissues. Toxicol Sci. |
R834514C004 (Final) |
not available |
|
|
Furlong CE, Marsillach J, Jarvik GP, Costa LG. 2016. Paraoxonases-1,-2 and-3:What are their functions? Chem Biol Interact. doi:10.1016/j.cbi.2016.05.036. PMID:27238723. |
R834514C004 (Final) R834513C003 (2016) |
not available |
|
|
Garrick JM, Dao K, de Laat R, Elsworth J, Cole TB, Marsillach J, Furlong CE, Costa LG. 2016. Developmental expression of paraoxonase 2. Chem Biol Interact. pii:S0009-2797(16)30116-8. doi:10.1016/j.cbi.2016.04.001. PMID:27062895. |
R834514C004 (Final) |
not available |
|
|
Marsillach J, Costa LG, Furlong CE. 2016. Paraoxonase-1 and Early-Life Environmental Exposures. Ann Glob Health. 82(1):100-10. PMID:27325068; PMCID:PMC4916371. |
R834514C004 (Final) |
not available |
|
|
Cole, T.B., J.C. Fisher, T.M. Burbacher, L.G. Costa, and C.E. Furlong, Neurobehavioral assessment of mice following repeated postnatal exposure to chlorpyrifos-oxon. Neurotoxicol Teratol, 2012. 34(3):p. 311-322. |
R834514C004 (Final) R834798C003 (Final) |
not available |
|
|
Burton C, Marsillach J, Zhang YS, Zhao W, Zheng X. Bioanalysis. 2015;7(13):1667-73. doi:10.4155/bio.15.90. Epub 2015 Jul 7** Judit Marsillach selected as a finalist for the Young Investigator Award to identify and reward promising early career researchers in the community. |
R834514C004 (Final) |
not available |
|
|
Furlong, C.E., R.J. Richter, L.G. Costa, and G.P. Jarvik, Paraoxonase 1 (PON1) Status in Risk Assessment for Organophosphate Exposure and Pharmacokinetics, in Chapter 9:Parameters for Pesticide QSAR and PBPK/PD Models for Human Risk Assessment. 2012, American Chemical Society:Available at:http://pubs.acs.org/doi/abs/10.1021/bk-2012-1099.ch009. p. 133-147. |
R834514C004 (Final) |
not available |
|
|
Costa, L.G., G. Giordano, and C.E. Furlong, Pharmacological and dietary modulators of paraxonase 1 (PON1) activity and expression:the hunt goes on. Biochem Pharmacol, 2011. 81(3):p. 337-344. PMCID3077125. |
R834514C004 (Final) |
not available |
|
|
Kim, D.S., A.A. Burt, J.E. Ranchalis, R.J. Richter, J.K. Marshall, K.S. Nakayama, E.R. Jarvik, J.F. Eintracht, E.A. Rosenthal, C.E. Furlong, and G.P. Jarvik, Dietary cholesterol increases paraoxonase 1 enzyme activity. J Lipid Res, 2012. 53(11):p. 2450-8. |
R834514C004 (Final) |
not available |
|
|
Kim, D.S., A. Burt, J. Ranchalis, R.J. Richter, E. Marshall, E. Rosenthal, C. Furlong, and G.P. Jarvik, Additional common polymorphisms in the PON gene cluster predict PON1 activity but not vascular disease. J Lipids, 2012. 2012(http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3364586/pdf/JL2012-476316.pdf). |
R834514C004 (Final) |
not available |
Supplemental Keywords:
Children’s health, environmental health, pesticide, environmental exposure, risk assessment, risk management, risk communication, community-based participatory research, CBPR, intervention, Washington State, outreach and translation, mechanisms, susceptibility, genetics, gene-interactions, modeling, biostatistics, exposure assessment, exposure pathway, Health, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, ENVIRONMENTAL MANAGEMENT, Environmental Policy, Biology, Children's Health, Biochemistry, Risk Assessment, biological markers, pesticides, age-related differences, pesticide exposure, agricultural community, children's vulnerablityProgress and Final Reports:
Original Abstract Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834514C001 Community-Based Participatory Research
R834514C002 Pesticide Exposure Pathways
R834514C003 Molecular Mechanisms
R834514C004 Genetic Susceptibility
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2015 Progress Report
- 2014
- 2013 Progress Report
- 2012 Progress Report
- 2010
- Original Abstract
178 journal articles for this center