Grantee Research Project Results
2016 Progress Report: High-Throughput Cellular Assays for Modeling Toxicity in the Fish Reproductive System
EPA Grant Number: R835167Title: High-Throughput Cellular Assays for Modeling Toxicity in the Fish Reproductive System
Investigators: Schultz, Irvin R. , Young, Graham , Nagler, James , Swanson, Penny
Current Investigators: Schultz, Irvin R.
Institution: University of Washington , University of Idaho , National Oceanic and Atmospheric Administration
Current Institution: University of Washington
EPA Project Officer: Aja, Hayley
Project Period: August 1, 2012 through September 30, 2015 (Extended to September 30, 2017)
Project Period Covered by this Report: August 1, 2015 through July 31,2016
Project Amount: $1,199,908
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The overall objective is to demonstrate that cellular in vitro assays can provide valid estimates of model parameters used in computational models of fish reproduction. Specific objectives include optimizing rainbow trout pituitary cell culture for assessing toxicant effects on follicle stimulating hormone (FSH) and luteinizing hormone (LH) synthesis. Other objectives are to optimize in vitro ovarian follicle incubations for assessing basal and FSH induced steroidogenesis, optimize rainbow trout primary hepatocyte culture for assessing toxicant effects on vitellogenin (Vg) expression and to incorporate in vitro derived estimates of effect into a computational model of the fish reproductive system.
Progress Summary:
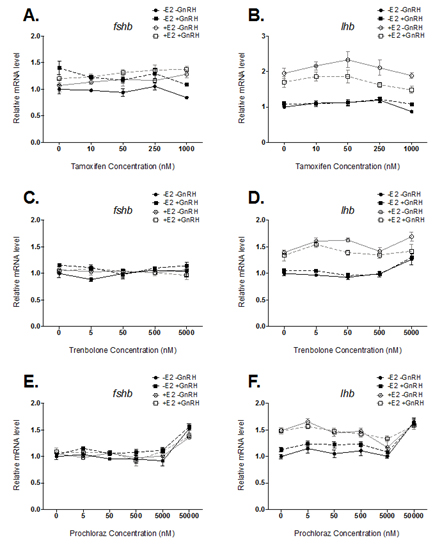
Objective 1: Pituitary cell culture. During the past year, trout primary pituitary cell cultures were exposed to several chemicals (tamoxifen [TAM], trenbolone [TREN], prochloraz [PRCHL], fluoxetine [FLX] and sertraline [SERT], citalopram [CIT]) and evaluated for effects on follicle stimulating hormone (FSH) and luteinizing hormone (LH) subunit gene expression. Pituitary cell cultures were prepared from maturing female rainbow trout and cells were exposed to varying concentrations of the test chemical for 48 hours. Gonadotropin-releasing hormone (GnRH; 10 nM) was then added to half of the cells for an additional 18 hours to investigate effects on basal versus GnRH-induced hormone secretion. In each experiment, half of the cells were exposed to 4 nM 17-beta estradiol (E2) for the duration of the exposure to test whether the chemical interferes with estrogen signaling in the cells. After 66 hours, cells and media were collected for RNA and protein analyses, respectively.
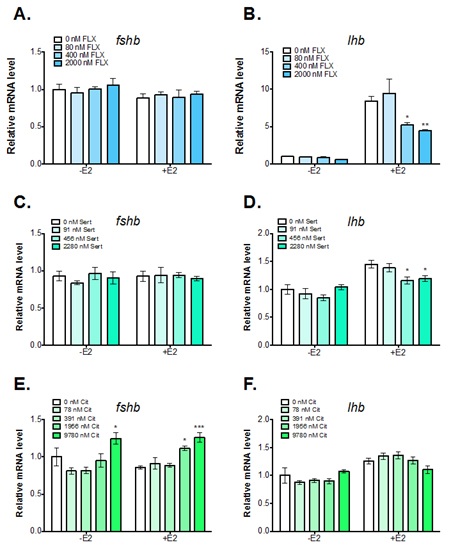
E2 treatment increased LH beta subunit (lhb) mRNA levels approximately 1.5 to 2-fold in each experiment (Figure 1B, 1D, and 1F). These results confirm our previous data that E2 stimulates lhb mRNA levels in vitro. Pituitary cells exposed to the highest doses of TREN (5000 nM) and PRCHL (50000 nM) had significantly higher levels of lhb and cells exposed to the highest dose of PRCHL also had increased FSH beta subunit (fshb) mRNA levels (Figure 1D, 1E, and 1F). However, there was no evidence of a consistent, dose-dependent effect of TAM, TREN, or PRCHL on FSH or LH subunit gene expression in primary pituitary cell cultures. These results indicate that TAM, TREN, and PRCHL have little effect on fshb or lhb mRNA levels. Although it was expected that the estrogen receptor antagonist TAM would decrease E2-induction of lhb, the lack of effect observed may be due to reduced metabolism of TAM into 4-hydroxy tamoxifen (4OH-TAM) within the pituitary cells. Future work examining the effects of 4OH-TAM on pituitary cells will be necessary to understand in-vivo impacts. The effects of TAM, TREN, and PRCHL on FSH and LH protein release are currently being analyzed. FLX treatment of 82 to 2040 nM caused a dose-dependent decrease in the E2-induction of lhb mRNA (Figure 2B). There was no effect of FLX on fshb mRNA or gonadotropin release at the examined doses. Pituitary cell death was observed in the 10,200 and 51,000 nM FLX treatments. Finally, primary pituitary cell cultures were also exposed to 91 nM to 57,000 nM SERT or 78 nM to 48,900 nM CIT for 66 hours with or without 4 nM E2. Similar to FLX, cells exposed to 456 or 2280 nM SERT had significantly lower E2-induction of lhb mRNA levels (Figure 2D). Additionally, visual inspection revealed cytotoxicity in cells exposed to 11,400 and 57,000 nM SERT. In contrast to FLX and SERT, cells exposed to 1956 nM and 9780 nM CIT showed increased levels of fshb mRNA levels and no inhibition of E2-induction of lhb mRNA levels. Further, cytotoxicity was only observed in cells exposed to 48,900 nM CIT.
Figure 1. Effect of tamoxifen (TAM), trenbolone (TREN), and procholoraz (PRCHL)
treatment for 66 hours on basal and E2-regulated fshb and lhb mRNA levels in
dispersed pituitary cells from female rainbow trout. mRNA levela were normalized
to ubiqitin, which was stably expressed across treatments. Significant differences
were analyzed by 2-way ANOVA followed by Dunnett's multiple comparison test.
Figure 2. Effect of fluoxetine (FLX), sertaline (SERT), and citalopram (CIT)
treatment on basal and E-2 regulated fshb and lhb mRNA levels in dispersed
pituitary cells from female rainbow trout. mRNA levels were normalized to
eukaryoitic elongation factor 1 (eefla), which was stably expressed across
treatments. Significant differences were analyzed by 2-way ANOVA followed
by Dunnett's multipl.e comparison test. Levels significantly different from
controls are indicated with an asterisk. (* = p<0.05, ** = p<0.01, *** = P<0.001)
These results demonstrate consistent upregulation of lhb mRNA levels in pituitary cell cultures exposed to E2. Treatment of cells with an estrogen receptor antagonist (TAM), a synthetic androgen (TREN), or an androgen-receptor antagonist (PRCHL) had little or no effect on basal or E2-regulated gonadotropin gene expression. However, effects of these chemicals on FSH and LH secretion cannot be ruled out. In addition, metabolites of the test chemicals (e.g. 4OH-TAM) may have different effects than the parent compounds. The effects of chemical metabolites on pituitary gonadotropin expression could have important implications in the context of whole animal effects and merits further study. In addition, results indicate selective serotonin reuptake inhibitors (SSRIs) such as FLX, SERT and CIT may affect basal and E2-regulation of pituitary gonadotropins. FLX and SERT are able to inhibit E2-induction of lhb in primary pituitary cell culture from female rainbow trout. In contrast, CIT treatment increased fshb mRNA levels in pituitary cells from female trout. These results suggest that various SSRIs may have differing effects on pituitary gonadotropin expression and should not be assumed to act via the same mechanism of action. In addition, all three SSRIs tested were shown to cause cytotoxicity to pituitary cells at high doses, possibly due to increased uptake into the cells. Additional studies should be conducted to confirm the effects of SSRIs on gonadotropin expression and to examine SSRI uptake into pituitary cells. In the coming year, our priorities will include drafting two papers for publication, and conducting experiments to test chemical metabolites (4OH-TAM) and further explore the effects of SSRIs on gonadotropin gene expression in vitro.
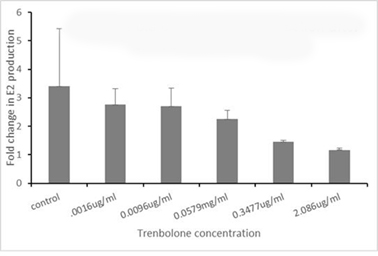
Objective 2: Ovarian Follicle. Testing of ovarian follicles (isolated from female trout in early vitellogenic stage of development) is continuing using our standard protocol: ovarian fragments (10 follicles/ml) are cultured in trout Ringers solution for either 18 or 40 hours with varying concentrations of the test chemical. Next, the Ringers solution is exchanged with a solution containing the same test concentrations with or without 500 ng/ml sGTH (a partially purified pituitary extract). At 6, 12 or 24 hours following the media change, 0.1 ml is collected for assessment of E2 levels by radioimmunoassay. In Figure 3, results from exposure to Trenbolone are shown. Note how increasing concentrations of Trenbolone decrease the GTH stimulated production of E2 after 6 hours. Testing is currently ongoing for two other chemicals with several more planned in early FY 17.
Figure 3. FOld change in E2 production after 6-hour sGTH stimulation.
Objective 3: Hepatocyte culture. Experiments this year exclusively used the approach of adding E2 prior to introduction of test compounds. This pre-treatment with E2 is intended to stimulate the hepatocyte system by turning on estrogen-responsive gene pathways (e.g., vitellogenin [VTG]) and to permit a better competitive assessment of ER-toxicant interaction(s). Our protocol adds E2 (1μg/ml) to the hepatocytes 24 hours before toxicant introduction and E2 (1μg/ml) is included with the test compounds when they are added for a further 24 hours. All chemicals (prochloraz, 4-hydroxytamoxifen, tamoxifen, trenbolone, and fluoxetine) have been exposed to hepatocytes across a dose response range (6 five-fold dilutions from the stocks provided) from three fish (i.e., three individual bioassays). Prochloraz is the only test compound that has been completely measured in the endpoint assay (RT-PCR for the VTG mRNA) in all three bioassays. The data shows no difference across any of the doses examined with respect to the E2 control. In other words, there is a positive response by the cells to E2, but the prochloraz treatments neither enhanced or inhibited the VTG mRNA response.
Objective 4: Computational modeling & in vitro – in vivo comparisons
During this reporting period major progress was made on modeling the reproductive axis in trout. Our complete computational model was published in PLoS Computational Biology in April 2016 (Gillies et al. 2016). Included in this paper was a demonstration of how the model can incorporate in vitro and in vivo data from contaminant exposures and then predict subsequent adverse reproductive outcomes in trout. We used trenbolone as the contaminant and demonstrated that multiple target sites (inhibition of ovarian E2 synthesis and direct effects on the liver) are needed to explain the relatively high potency of this contaminant on reproduction in fishes. We have subsequently used the model to predict the threshold when decreases in ovarian E2 synthesis, pituitary synthesis of FSH and LH and hepatic production of vitellogenin will begin to cause impacts on trout reproduction. An example of this process is shown in Figure 4. This figure shows model predicted oocyte growth during a trout reproductive cycle when there is normal E2 synthesis rates (black dashed line) and under progressively reduced rates of E2 synthesis. These simulations suggest decreases in E2 synthesis rates of 25-30% will cause reduction in oocyte size and delayed or failure in spawning. During FY17, we will use the model and in vitro test results from Objectives 1-3 to predict the effects on reproductive performance in trout.
Figure 4. Predicted average oocyte diameter in maturing female trout
with normal (dashed black lines) or reduced E2 formation and secretion
by the ovaries. Model simulations were performed using the Gillies et al.
2016 model. Colored lines show predicted effects on egg growth when the
rate of E2 formation is reduced up to 60% throughout the reproductive
cycle (Top) or restricted to the mid-vitellogenic phase of growth (stages
4,5 and 6 in the model; Bottom graph). These simulations suggest if
E2 production by the ovaries is reduced by 25% thorughout the cycle
(pink line) or 30% during the mid-vitellogenesis (yellow line), oocyte growth
will be reduced to the point where complete egg development may not occur.
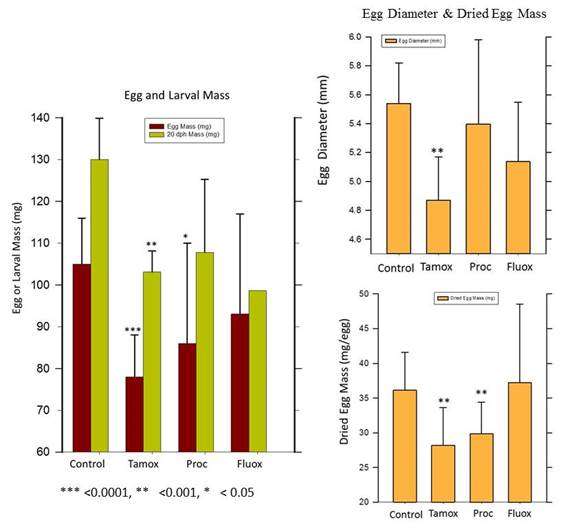
In addition to progress on model development and application, we also completed a year-long exposure of female rainbow trout to four test chemicals: trenbolone, tamoxifen, prochloraz and fluoxetine. This experiment was performed to provide in vivo experimental data for comparison with the in vitro test data and model predicted effects on reproduction. This will permit us in FY 17 to more rigorously evaluate the accuracy of our in vitro / modeling approach for predicting the effects of contaminant exposure on fish reproduction. Except for trenbolone, fish from all treatment groups were able to spawn after exposure. The primary effect of exposure was a decrease in egg mass or diameter, which is summarized in Figure 5. Note that for some chemicals such as the anti-estrogen tamoxifen, exposure caused egg mass at spawning to decrease by 30%. Exposure to trenbolone caused complete reproductive failure in all fish.
Figure 5. The effects of full reproductive cycle exposure to three tech chemicals, Tamoxifen (Tamox),
Procholraz (Proc) and Fluoxetine (Fluox) on egg size in rainbow trout. Mature female trout were
exposed to each test chemical beginning 3-weeks after their first spawn. The exposure continued
until time of the next (second) spawn, whch occured between 52-60 weeks later. At time of the second
spawning, all eggs were removed from each fish, counted, and a subset of 25-50 measured for
diameter and total mass. Dried egg mass was determined after 48 hours in a 70 oC oven. The mean
exposure level fo reach checmical was 501 ng/L (Tamox), 24 µg/L (Proc) and 2.5 µg/L (Fluox). Data
shown on figures are the mean ± SD of n=16 (controls), n=15 (Tamox), n=9 (Proc) and n=5 (Fluoxetine).
At completion of the experiment (14 months after previous spawn), all control trout had spawned while
83, 89 and 100% of the surviving Tamox, Proc and Fluox exposed trout, respectively, had spawned.
There were no mortalities int eh control ro Tamox groups, but 10 and 50% of the Proc and Fluox
exposed trout died during the experiment.
Future Activities:
Additional experiments will be performed with pituitary cells and ovarian follicles to complete chemical testing and to confirm the effects of SSRIs on gonadotropin expression and secretion. Experiments are also planned to determine chemical partitioning and macromolecular binding within the in vitro test systems. With regard to objective 4, sufficient project data will now be available to perform in vitro / in vivo comparisons and rigorous evaluation of the accuracy of our modeling approach for predicting the effects of contaminant exposure on fish reproduction. Other planned activities will be to draft manuscripts for peer-reviewed publication that summarize the in vitro testing and use of in vitro data with computational models for quantitative adverse outcome pathway modeling.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 24 publications | 6 publications in selected types | All 4 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Gillies K, Krone SM, Nagler JJ, Schultz IR. A computational model of the rainbow trout hypothalamus-pituitary-ovary-liver axis. PLoS Computational Biology 2016;20;12(4):e1004874 (27 pp.). |
R835167 (2015) R835167 (2016) R835167 (Final) |
Exit Exit Exit |
|
|
Jia Y, Cavileer TD, Nagler JJ. Acute hyperthermic responses of heat shock protein and estrogen receptor mRNAs in rainbow trout hepatocytes. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 2016;201:156-161. |
R835167 (2015) R835167 (2016) R835167 (Final) |
Exit Exit Exit |
|
|
Schultz IR, Nagler JJ, Swanson P, Wunschel D, Skillman AD, Burnett V, Smith D, Barry R. Toxicokinetic, toxicodynamic, and toxicoproteomic aspects of short-term exposure to trenbolone in female fish. Toxicological Sciences 2013;136(2):413-429. |
R835167 (2013) R835167 (2014) R835167 (2015) R835167 (2016) R835167 (Final) |
Exit Exit |
Supplemental Keywords:
recrudescence, maturation, gonadotropin, endocrine disruption, water;Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.