Grantee Research Project Results
2014 Progress Report: High-Throughput Cellular Assays for Modeling Toxicity in the Fish Reproductive System
EPA Grant Number: R835167Title: High-Throughput Cellular Assays for Modeling Toxicity in the Fish Reproductive System
Investigators: Schultz, Irvin R.
Institution: Pacific Northwest National Laboratory
Current Institution: University of Washington
EPA Project Officer: Aja, Hayley
Project Period: August 1, 2012 through September 30, 2015 (Extended to September 30, 2017)
Project Period Covered by this Report: August 1, 2013 through August 31,2014
Project Amount: $1,199,908
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The overall objective is to demonstrate that cellular in vitro assays can provide valid estimates of model parameters used in computational models of fish reproduction. Specific objectives include optimizing rainbow trout pituitary cell culture for assessing toxicant effects on follicle stimulating hormone (FSH) and luteinizing hormone (LH) synthesis and determine whether trout pituitary cell lines are an acceptable alternative to primary pituitary cells. Other objectives are to optimize in vitro ovarian follicle incubations for assessing basal and FSH induced steroidogenesis, optimize rainbow trout primary hepatocyte culture for assessing toxicant effects on vitellogenin (Vg) and estrogen receptor- α1 expression and to incorporate in vitro derived estimates of effect into a computational model of the fish reproductive system.
Progress Summary:
During this project period, progress was made in all aspects of the project. Specific accomplishments for the hepatocyte studies were the use of a juvenile male rainbow trout hepatocyte culture system to expand upon the number of test compounds examined from year 1. Besides ethynylestradiol (EE2), which is being used as a positive control, a range of five concentrations of tamoxifen (Tam), fluoxetine, and flutamide were tested. Similar to past results, EE2 gives a robust induction of the two mRNAs (ERα1 and Vg) and the protein (Vg) endpoints. However, of the three new compounds tested only Tam induced the Vg protein in a dose response manner in hepatocytes isolated from 2 of the 3 fish tested. Subsequent experiments are co-incubating test chemicals with estradiol (E2), which is added as a chemical competitor and to stimulate a basal Vg synthesis comparable to mid-vitellogenic female trout. We evaluated different doses of E2, timing of toxicant introduction, and cell harvesting. An E2 dose of 1μg/ml was selected to add to the cells 24 hours before toxicant introduction; further incubation for 48 hours indicates a sufficient time interval for a significant Vg response. Most recently, we have been conducting experiments with a specific ER inhibitor, 4-hydroxytamoxifen (4-HTam), the major metabolite of tamoxifen. The highest dose of 4-HTam (450 nM) shows a significant reduction (by 50%) in ERa1 mRNA (the only endpoint tested presently), compared to controls. Other, lower doses of 4-HTam are not different from the control.
For the ovarian follicle studies, initial experiments were conducted using vitellogenic rainbow trout follicles ranging in size from approximately 1-3 mm diameter. Comparison of basal and time-course production of estradiol-17β (E2) production was evaluated in response to a range of concentrations of partially purified salmon gonadotropin (sGTH), using three different types of incubation media. On the basis of these experiments, the following protocol was adopted: ovarian fragments (10 follicles/ml) are cultured in trout Ringers for 18 hours at 14 C with concentrations of a test chemical varying from solubility limits to environmentally relevant concentrations. At 18 hours, the media is completely changed and replaced with media containing the same test concentrations with or without 500 ng/ml sGTH. At 6 hours, 12 hours, and 24 hours following the media change, 0.1 ml is collected for assessment of E2 levels by radioimmunoassay. The concentration of E2 in media is compared across treatments and time points. Depending on follicle size, sGTH caused an increase in E2 production of between approximately 2-2.5 fold. The following four chemicals were then tested: Fluoxetine At 50 µM exposure, a 15% inhibition of sGTH-stimulated E2 production was observed, but there was no effect on either basal or sGTH-stimulated E2 production at lower concentrations of fluoxetine.
Trenbolone A 14% inhibition of sGTH-stimulated E2 production occurred at a concentration of 8.0 nM, with a shallow dose response curve resulting in 31% inhibition of sGTH-stimulated E2 production at 50 µM of the chemical. There was no effect on basal E2 production. Flutamide A 46% inhibition of basal E2 production and a 37% inhibition of sGTH-stimulated E2 production at 100 µM occurred but there was no effect on either basal or sGTH-stimulated E2 production at lower concentrations. EE2 An approximately 17% inhibition of sGTH-stimulated E2 production occurred at 0.1 nM, 1.0 nM and 10.0 nM EE2, with a strong concentration-related decrease of 66% at 100 nM and 80% at 1000 nM EE2. EE2 had no significant effects on basal production.
Summary: We are confident with the reproducibility of the protocol adopted on the basis of extensive pilot experiments. EE2 was the only chemical tested that exhibited inhibition of GTH-stimulated E2 production at low, near environmentally-relevant concentrations. As far as we are aware, this is the first study to show that EE2 directly downregulates the ability of follicles to respond to sGTH.
For the pituitary cell culture system, focus was on 1) evaluating effects of natural and synthetic estrogens on female trout pituitary FSH and LH subunit gene expression and secretion and 2) to determine if the responses varied between pre-vitellogenic versus vitellogenic stages since it is well established that steroid feedback effects on gonadotropins vary during the reproductive cycle. This was important to establish in the strains of rainbow trout that we are using to establish the cell based screening assays. For our studies we are using Troutlodge rainbow trout strains that have been selected to spawn in February, May, August or November. The advantage is that we can simultaneously test effects of test chemicals/hormones on pituitary cells prepared from previtellogenic and vitellogenic fish in a single experiment.
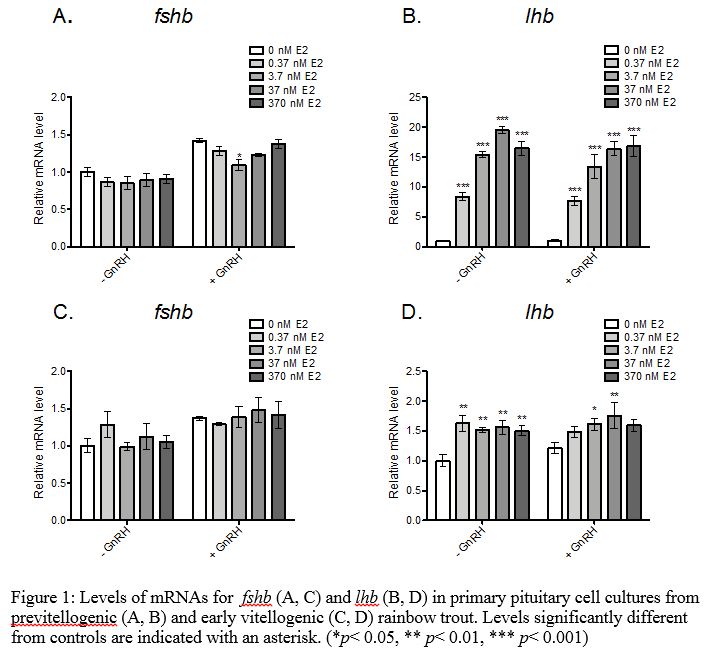
Stage Dependent Effects of estradiol-17beta (E2) on FSH and LH: Cell cultures were prepared from previtellogenic rainbow trout (9 months pre-spawning) and from early vitellogenic rainbow trout (5 months pre-spawning. Cells were exposed to 0.37 nM to 367nM E2 for 48 hours and then gonadotropin-releasing hormone (GnRH) was added to half of the cells for an additional 18 hours to investigate effects on basal versus GnRH-induced hormone secretion. Luteinizing hormone beta subunit (lhb) was upregulated 8- to 20-fold in pituitary cells from previtellogenic trout at all concentrations tested with maximal induction at 37 nM E2 (Figure 1). E2 also increased GnRH-induced secretion of luteinizing hormone (LH) in cells from previtellogenic trout (data not shown). In contrast, E2 had no effect while GnRH had a slight positive effect on fshb mRNAs. However, when GnRH was combined with E2, the positive effect on fshb was not observed. In pituitary cells from early vitellogenic females, lhb was upregulated 1.5-fold at all concentrations tested, but this effect was not concentration dependent. There was no effect of E2 on basal or GnRH-induced LH secretion. Due to the greater inducibility of lhb and LH secretion by E2 in previtellogenic rainbow trout, subsequence tests were conducted with pituitary cells from previtellogenic trout.
Effects of ethinylestradiol (EE2) on FSH and LH
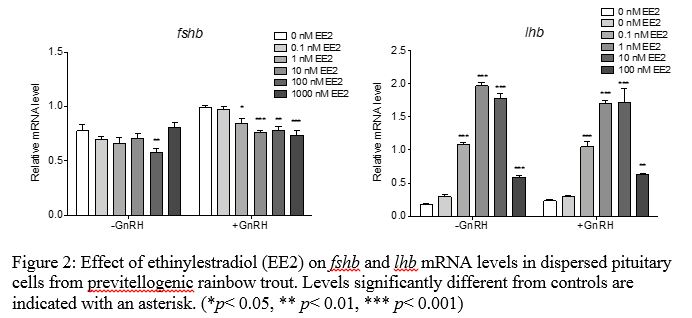
A series of primary pituitary cell culture experiments were conducted to investigate the effects of EE2 and FLX on gonadotropin synthesis and secretion. For each chemical, dispersed pituitary cells from previtellogenic rainbow trout were exposed to a range of doses in the culture media for 48 hours followed by 18 hours with or without 10 nM GnRH for a total exposure of 66 hours. At the termination, cells and media were collected for RNA isolation and gonadotropin immunoassays, respectively. Pituitary cell cultures exposed to 0.1 – 1000 nM EE2 with or without GnRH showed altered gonadotropin gene expression. Fshb mRNA levels were significantly depressed by 100 nM EE2 alone, or 1 – 1000 nM EE2 in the presence of GnRH. On the other hand, lhb mRNA levels were significantly elevated by 1 to 1000 nM EE2, with maximal induction at 10 nM EE2 (Figure 2). This confirms that estrogen receptor agonists can directly induce lhb mRNA levels in rainbow trout pituitary glands.
Effects of fluoxetine (FLX) and E2 on FSH and LH
The effects of one SSRI, FLX, on E2-regulated gonadotropin gene expression were examined in previtellogenic fish. Dispersed pituitary cells were exposed to 82 to 51,000 nM FLX with or without 3.7 nM E2 and 10 nM GnRH. In these cells, E2 elevated lhb mRNA levels 4- to 7-fold, as expected. FLX treatment of 82 to 2040 nM caused a dose-dependent decrease in the E2-induction of lhb mRNA (Figure 3). There was no effect of FLX on fshb mRNA or gonadotropin release at the examined doses. Pituitary cell death was observed in the 10,200 and 51,000 nM FLX treatments, and this was also reflected in poor RNA yields from cells isolated from these higher doses.
Summary and next steps
Pituitary cells from the previtellogenic stage fish were more sensitive and had a greater magnitude of response to estrogens (EE2, E2) in terms of lhb gene expression and GnRH-induced LH secretion than early vitellogenic stage fish. GnRH had slight stimulatory effects on fshb, but this was inhibited in the presence of either E2 or EE2 suggesting estrogens may modulate FSH synthesis in a negative feedback manner through effects on GnRH signaling. Fluoxetine suppressed E2-induced lhb gene expression and at high concentrations is toxic to the pituitary cells.

Future Activities:
In the coming year, experiments will focus on testing other SSRIs, estrogen receptor antagonists and androgens. Additionally, we will test the trout pituitary cell lines for responses to estrogens.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 24 publications | 6 publications in selected types | All 4 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Schultz IR, Nagler JJ, Swanson P, Wunschel D, Skillman AD, Burnett V, Smith D, Barry R. Toxicokinetic, toxicodynamic, and toxicoproteomic aspects of short-term exposure to trenbolone in female fish. Toxicological Sciences 2013;136(2):413-429. |
R835167 (2013) R835167 (2014) R835167 (2015) R835167 (2016) R835167 (Final) |
Exit Exit |
Supplemental Keywords:
recrudescence, maturation, gonadotropin, disruption, waterProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
