Grantee Research Project Results
2013 Progress Report: High-Throughput Cellular Assays for Modeling Toxicity in the Fish Reproductive System
EPA Grant Number: R835167Title: High-Throughput Cellular Assays for Modeling Toxicity in the Fish Reproductive System
Investigators: Schultz, Irvin R.
Institution: Pacific Northwest National Laboratory
Current Institution: University of Washington
EPA Project Officer: Aja, Hayley
Project Period: August 1, 2012 through September 30, 2015 (Extended to September 30, 2017)
Project Period Covered by this Report: September 1, 2012 through August 31,2013
Project Amount: $1,199,908
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The overall objective is to demonstrate that cellular in vitro assays can provide valid estimates of model parameters used in computational models of fish reproduction. Specific objectives include optimizing rainbow trout pituitary cell culture for assessing toxicant effects on follicle stimulating hormone (FSH) and luteinizing hormone (LH) synthesis and determine whether trout pituitary cell lines are an acceptable alternative to primary pituitary cells. Other objectives are to optimize in vitro ovarian follicle incubations for assessing basal and FSH induced steroidogenesis, optimize rainbow trout primary hepatocyte culture for assessing toxicant effects on vitellogenin and estrogen receptor- α1 expression and to incorporate in vitro derived estimates of effect into a computational model of the fish reproductive system.
Progress Summary:
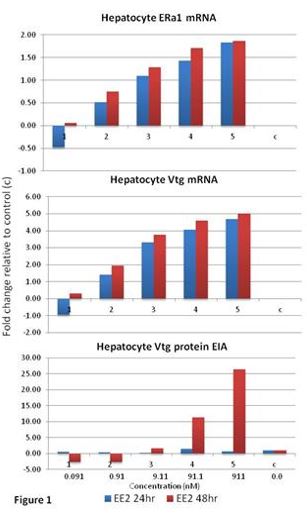
During this project period, progress was made in all aspects of the project. Specific accomplishments were the establishment of a chemical repository for the project, which involved acquisition of test chemicals and preparation of stock solutions. Chemical stability tests in cell culture media are currently underway. A series of isolated hepatocyte preparations were performed in order to optimize cell isolation methods, culturing conditions and evaluate cryopreservation techniques. Freshly prepared hepatocytes consistently have viability in excess of 90%. Our initial attempts at cryopreservation of trout hepatocytes resulted in 30% viability after freezing, thus continued work is planned to improve viability to > 50%. Test chemical exposure of hepatocytes was started with ethynylestradiol (EE2), which was incubated with cells for 24 and 48 hrs. Endpoints measured included vitellogenin (VG) mRNA expression (measured by Q RT-PCR), VG protein (measured by VG ELISA), and estrogen receptor alpha-1 (ERα1) mRNA (measured by Q RT-PCR). A summary of these results are presented in figure 1. A robust, dose response for both of the mRNAs was observed at each time point. The VG protein endpoint was only apparent in the medium at the two higher exposure levels after 48 hours. A series of isolated pituitary cell preparations were made from several fishes to optimize culturing conditions and establish responsiveness of FSHβ and LHβ gene expression towards added estrogen (E2) and androgen (11-ketotestosterone). Initial results demonstrated LHβ expression was stimulated by E2 at concentrations of 3.7 pM or greater. In addition, a graduate student supported by the project Lousia Harding, traveled to Dr. Thomas Chen’s laboratory to gain hands on experience with the culturing and maintenance of trout pituitary cell lines that will be used on the project. A representative photograph of these cultures is shown in Figure 2 (mag. 100x). With regard to computational modeling, a series of experiments was performed to characterize E2 elimination and plasma protein binding in female trout. The focus was to establish whether maturational changes (ovarian growth) cause a shift in binding or clearance. The results, which were incorporated into a manuscript now in press with Toxicological Sciences, indicated no significant changes were occurring and constant values could be used in models.
Future Activities:
Planned activities for the second project year are to expand in vitro testing with 2-4 test chemicals using isolated pituitary cells, hepatocyes and ovarian follicles. Recruiting is underway for a post-doctoral scientist to assist the PI with computational modeling and reverse toxicokinetics.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 24 publications | 6 publications in selected types | All 4 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Schultz IR, Nagler JJ, Swanson P, Wunschel D, Skillman AD, Burnett V, Smith D, Barry R. Toxicokinetic, toxicodynamic, and toxicoproteomic aspects of short-term exposure to trenbolone in female fish. Toxicological Sciences 2013;136(2):413-429. |
R835167 (2013) R835167 (2014) R835167 (2015) R835167 (2016) R835167 (Final) |
Exit Exit |
Supplemental Keywords:
recrudescence, maturation, gonadotropin, disruption, waterProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.