Grantee Research Project Results
Final Report: PON1 as a Predictor of Differential Susceptibility of Children to Organophosphate Pesticides
EPA Grant Number: R832734Title: PON1 as a Predictor of Differential Susceptibility of Children to Organophosphate Pesticides
Investigators: Eskenazi, Brenda , Barr, Dana Boyd , Bradman, Asa , Holland, Nina T. , Harley, Kim , Hubbard, Alan
Institution: University of California - Berkeley
EPA Project Officer: Callan, Richard
Project Period: March 20, 2006 through February 28, 2009 (Extended to February 28, 2011)
Project Amount: $749,987
RFA: Early Indicators of Environmentally Induced Disease (2004) RFA Text | Recipients Lists
Research Category: Children's Health , Chemical Safety for Sustainability
Objective:
This research has focused on investigating whether PON1 genotype/activity in populations exposed to pesticides is an early indicator of environmentally-induced disease. The research results have the potential to inform future policy decisions regarding allowable pesticide exposure to children and pregnant women necessary for the implementation of the Food Quality Protection Act of 1996.
Recent studies demonstrate widespread pesticide exposures to pregnant women and children in the US. However, given the same level of pesticide exposure, some individuals may be more susceptible to the potential adverse health effects of pesticides depending on their genetic makeup and expression of genes encoding key metabolic enzymes. For example, the human enzyme paraoxonase (PON1) detoxifies various organophosphate (OP) pesticides with different efficiency depending on the main single nucleotide polymorphism (SNP) at position192 and other SNPs along promoter (-108) and coding regions. PON1 activity is very low in newborns, with the lowest levels in children with certain genotypes. Thus, PON1 genotype/activity may be a marker for the differential susceptibility of children to OP exposures and their potential adverse associations with neurodevelopment.
The CHAMACOS Study is a longitudinal birth cohort study of children living in the agricultural community of the Salinas Valley, CA and is the keystone project of the US EPA/NIEHS Center for Environmental Research and Children’s Health at UC Berkeley. This project utilized the bloods stored in the CHAMACOS Biorepository and the extensive longitudinal data on children’s neurodevelopment and OP exposures as assessed by the measurement of maternal and child urinary levels of diakylphosphate metabolites (DAPs). In this study, we investigated the role of PON1 in determining susceptible subpopulations. Initial data suggested that DAPs of the pregnant women in this study exceeded levels in national reference data (NHANES). This analysis benefited from the extensive information obtained over the years about this population, including data on chemical exposures beyond OPs (non-persistent endocrine disrupting pesticides, organochlorines, PCB’s, PBDE), physical environment (housing quality), diet, and participants’ psychosocial milieu (social support, depression, household structure, socioeconomic status, neighborhood characteristics,etc.). We considered many of these variables as potential confounders in the statistical analyses.
Summary/Accomplishments (Outputs/Outcomes):
Organophosphate pesticides (OPs) are widely used in agriculture and may have neurotoxic effects on people who are exposed to them. This research concerns effects of OP pesticides on children’s development and investigates whether individual children differ in their susceptibility to effects of OP exposure on the basis of their genetic makeup. Specifically, we investigate the effects of PON1 genes and the enzymes they code, which are related to the body’s ability to break down OPs.
We have conducted this research in a cohort of mothers and children residing in an agricultural community in California. Mothers were enrolled in the study while pregnant, and their children, born in 2000 and 2001, have been followed by the study ever since, with visits occurring at least every 2 years. We have measured OP breakdown products in urine collected from the mothers during their pregnancy and from children at multiple time points since birth. We have genotyped blood collected from mothers and children, and have measured the quantity and quality (i.e. efficiency in detoxifying OPs) of PON1 enzymes in blood samples collected at multiple timepoints. We have also formally assessed children’s learning, development, and behavior on an ongoing basis since infancy.
A key finding of our research is that the quantity and quality of PON1 enzymes in children’s blood do not reach adult levels until approximately age 9. This means that there is a long period of time (i.e. from conception through age 9) when children are likely more vulnerable than adults to effects of OPs. In addition, mothers and children with certain gene types habitually show relatively low levels of enzyme activity relative, suggesting that, at any age, susceptibility to potential toxic effects of OPs likely differs across individuals.
Another important finding is that mothers’ levels of OP breakdown products in urine as measured during pregnancy are associated with children’s mental development at age 2, attention problems at age 5, and IQ at age 7. Specifically, mothers with higher levels of OP exposure tended to have children with poorer mental development or IQ and more evidence of attention problems.
Our analyses provide some evidence that children with certain susceptible genotypes and/or lower quantity or quality of PON1 enzymes display poorer mental development/IQ and more attention problems than others. They also provide evidence, albeit limited, that the relationships between OP exposure and neurodevelopmental and behavioral outcomes differ according to the child’s genotype and/or level of enzymes.
In summary, our research indicates that prenatal OP exposure is associated with sustained (at least for ages 2-7) deficits in mental development and behavior, that the degree of effect may well differ between individuals based on their (or their mothers’) genetically-programmed ability to detoxify OPs, and that children’s ability to detoxify OPs does not reach adult levels of maturity until approximately 9 years of age, suggesting increased vulnerability to exposure in this time frame.
Summary of Findings:
Specific Aim 1: To determine PON1 genotype and enzyme activity levels in a cohort of 5- and 7-year-old Mexican-American children through analysis of cord and child blood samples.
Our findings with regards to this aim can be summarized as follows:
- We identified a significantly longer than expected period of lower PON1 enzyme expression (levels) and substratespecific activity in young children lasting through 7 years of age. Furthermore, increases of enzyme activity with age were modified by PON1 genotype (Figure 1) such that children with the PON192 RR genotype experienced much steeper increases of POase activity than PON1192 QQ (p<0.0001). In fact by age 7, mean POase acivity in QQ children were still lower than average POase activity in QR and RR children at age 2.
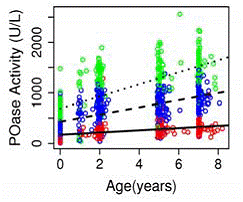
Figure 1. PON1 enzyme activity by age and PON1192 genotype (green RR, blue QR, red QQ)
- We determined that even at age 7, children’s PON1 activities were still slightly lower than those in mothers and this difference was statistically significant. These data suggest that the window of susceptibility to OPs lasts much longer than previously thought and children's protection from OPs via the PON1 enzyme remains somewhat lower than that in their mothers at least up to school age.
- We received ARRA Supplement funding for 2009-2010 to measure PON1 phenotype in 9-year-old children to establish the final range of ages when children's PON1 levels plateau. This work has recently been completed and a manuscript is in progress.
- As an extension of this work on PON1 genotypes and enzyme activity, we also sought to characterize genetic structure of the PON1 gene beyond a few commonly used single nucleotide polymorphisms (SNPs); no previous resequencing data and limited genotyping data were available in Mexican and other Hispanic populations. We sequenced the PON1 gene in 30 Mexican-American CHAMACOS subjects and identified 94 polymorphisms with a minor allele frequency > 5%, including several novel variants (6 SNPs,1 insertiion, 2 deletions). More than 40 variants of the PON1 gene were genotyped in CHAMACOS mothers and children and 12 were significantly associated with arylesterase activity (AREase, measure PON1 quantity), and 37 with paraoxonase activity (POase, measure of PON1 catalytic efficiency). However, most polymorphisms were in strong linkage disequilibrium with either PON1-108 or PON1192, SNPs with known effects on PON1 quantity and substrate-specific activity. We found that the general linkage disequilibrium structure of PON1 was similar between CHAMACOS Mexicans and Caucasians yet frequencies of many polymorphisms previously uncharacterized in Latino populations differed strikingly between populations. Additionally, the relative genetic contribution of PON1 SNPs toward enzyme activity differed between mothers and their children.
Specific Aim 2: To determine whether PON1 activity and genotype are associated with child neurodevelopment at ages 5 and 7 years.
With regards to direct associations of PON1 genotype and activity with neurodevelopment, we present our findings below by genotype and enzyme.
Genotype:
∙ PON and 2-year-old neurobehavioral development. We have published data demonstrating that child
PON1-108 genotype is associated with neurodevelopmental performance on the Bayley Scales of infant
Development. Children with the susceptible TT genotype performed more poorly on the Mental
Development Index (MDI) and Psychomotor Development Index (PDI) than CC individuals. Specifically,
compared to children with PON1-108CC genotype, those with PON1-108CT and PON1-108TT genotype
performed 3.8 points (p=0.005) and 5.7 points (p=0.001) lower on Bayley MDI and 1.4 (p=0.27) and 2.7
points (p=0.07) lower on Bayley PDI, respectively. PON1192 was not associated with MDI or PDI at age 2.
Enzyme Activity:
- PON enzymes and 2-year-old neurobehavioral development. We have noted no association between PON1 enzyme activity and Bayley MDI or PDI at age 2.
Specific Aim 3: To determine whether PON1 genotype/activity modifies the relationship of OP exposure and neurodevelopment at ages 5 and 7 years.
We have previously reported a positive association between mean maternal prenatal urinary DAP metabolite concentrations and attention problems in children and a negative association with IQ at age 7 years. Currently, we are testing the hypothesis that adverse associations of maternal DAPs would be most pronounced in children and/or offspring of mothers with susceptible PON1-108 and PON1-192 genotypes (i.e. TT and QQ, respectively) or with lower maternal or child levels of arylesterase, PO and CPO.
Implications for other researchers and policymakers:
- Studies of effects of OPs need to consider genetic susceptibility.
- Further research in this area should include determining whether common, current use OP pesticides can be measured in blood down to 1 pg/ml.
- Considerations for regulators:
▪ Not all individuals respond similarly to OPs
▪ Children are more susceptible than adults and much longer than we previously thought
▪ Children with the PON1 QQ/TT genotype seem to be the most susceptible for some neurodevelopmental outcomes.
Quality Assurance
Extensive QA/QC procedures were performed for measurement of PON1 genotype and activity. High throughput fluorogenic methods for PON1-108 genotyping were modified from the previously available version of the protocol, yielding much more consistent and robust results (>96% successful call rate). Additionally, an in-house RFLP method was established to validate results from fluorogenic methods. At least 5% of DNA specimens were genotyped using this QA/QC method, and the results show excellent rates of concordance with the high throughput genotyping.
New sets of QA/QC experiments to cross-validate PON1 level and activities measurements were conducted in collaboration with the laboratory of Dr. Furlong at the University of Washington, Seattle. The same reagents, Spectramax software, and protocols were applied to parallel samples of the CHAMACOS subjects independently analyzed in both labs. Enzyme activities for the same individuals assayed both at UC Berkeley and University of Washington were highly correlated. Significantly, we discovered activity levels were affected by the ambient temperature during the analysis. Adjustment for the temperature during the analyses was not previously employed by UW lab, but is now standard practice in both labs. The findings of this validation study were published in our 2009 paper, titled, “Validation of PON1 enzyme activity assays for longitudinal studies” (Huen et al, 2009).
Extensive quality assurance is also employed by CDC in the measurement of OP levels in blood. All OP analyses are conducted in the laboratory of Dr. Dana Barr. Dr. Barr uses stateof-the-art isotope dilution methods that allow sample-specific recovery adjustment, as well as stringent QA/QC that include the use of spikes, blanks, and blind external proficiency standards. The CDC laboratory is CLIA certified and follows Good Laboratory Practices. Plasma samples are stored in the UC Berkeley Biorepository at -80 degrees and shipped to CDC on dry ice.
Journal Articles on this Report : 20 Displayed | Download in RIS Format
| Other project views: | All 91 publications | 22 publications in selected types | All 22 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Bouchard MF, Chevrier J, Harley KG, Kogut K, Vedar M, Calderon N, Trujillo C, Johnson C, Bradman A, Barr DB, Eskenazi B. Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environmental Health Perspectives 2011;119(8):1189-1195. |
R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) R834513C001 (2012) |
|
|
|
Bradman A, Castorina R, Barr DB, Chevrier J, Harnly ME, Eisen EA, McKone TE, Harley K, Holland N, Eskenazi B. Determinants of organophosphorus pesticide urinary metabolite levels in young children living in an agricultural community. International Journal of Environmental Research and Public Health 2011;8(4):1061-1083. |
R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) R834513C002 (2010) R834513C002 (2011) R834513C002 (2012) R834513C003 (2010) R834513C003 (2011) R834513C003 (Final) |
Exit Exit Exit |
|
|
Coronado GD, Vigoren EM, Griffith WC, Faustman EM, Thompson B. Organophosphate pesticide exposure among pome and non-pome farmworkers:a subgroup analysis of a community randomized trial. Journal of Occupational and Environmental Medicine 2009;51(4):500-509. |
R832734 (Final) R831709 (2007) R831709 (Final) R834514 (2011) R834514 (Final) R834514C001 (Final) |
Exit |
|
|
Eskenazi B, Rosas LG, Marks AR, Bradman A, Harley K, Holland N, Johnson C, Fenster L, Barr DB. Pesticide toxicity and the developing brain. Basic & Clinical Pharmacology & Toxicology 2008;102(2):228-236. |
R832734 (2007) R832734 (Final) R831710 (2005) R831710 (Final) |
Exit Exit Exit |
|
|
Eskenazi B, Huen K, Marks A, Harley KG, Bradman A, Barr DB, Holland N. PON1 and neurodevelopment in children from the CHAMACOS study exposed to organophosphate pesticides in utero. Environmental Health Perspectives 2010;118(12):1775-1781. |
R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) R834513C003 (2010) R834513C003 (2011) |
Exit |
|
|
Eskenazi B, Kogut K, Huen K, Harley KG, Bouchard M, Bradman A, Boyd-Barr D, Johnson C, Holland N. Organophosphate pesticide exposure, PON1, and neurodevelopment in school-age children from the CHAMACOS study. Environmental Research 2014;134:149-157. |
R832734 (Final) R826709 (2002) R834513 (2014) R834513 (2015) R834513C001 (2014) R834513C001 (2015) |
Exit Exit Exit |
|
|
Harley KG, Huen K, Aguilar Schall R, Holland NT, Bradman A, Barr DB, Eskenazi B. Association of organophosphate pesticide exposure and paraoxonase with birth outcome in Mexican-American women. PLoS ONE 2011;6(8):e23923 (10 pp.). |
R832734 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) R834513C001 (2012) |
Exit Exit Exit |
|
|
Holland N, Furlong C, Bastaki M, Richter R, Bradman A, Huen K, Beckman K, Eskenazi B. Paraoxonase polymorphisms, haplotypes, and enzyme activity in Latino mothers and newborns. Environmental Health Perspectives 2006;114(7):985-991. |
R832734 (2006) R832734 (2007) R832734 (Final) R831709 (2005) R831709 (2006) R831709 (2007) R831709 (Final) R831709C002 (2006) R831710 (2004) R831710 (2005) R831710 (Final) R831710C001 (2007) R831710C003 (2006) |
|
|
|
Huen K, Richter R, Furlong C, Eskenazi B, Holland N. Validation of PON1 enzyme activity assays for longitudinal studies. Clinica Chimica Acta 2009;402(1-2):67-74. |
R832734 (2009) R832734 (Final) R831709 (2007) R831709 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (Final) |
Exit |
|
|
Huen K, Harley K, Brooks J, Hubbard A, Bradman A, Eskenazi B, Holland N. Developmental changes in PON1 enzyme activity in young children and effects of PON1 polymorphisms. Environmental Health Perspectives 2009;117(10):1632-1638. |
R832734 (2009) R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (Final) |
|
|
|
Huen K, Harley K, Bradman A, Eskenazi B, Holland N. Longitudinal changes in PON1 enzymatic activities in Mexican-American mothers and children with different genotypes and haplotypes. Toxicology and Applied Pharmacology 2010;244(2):181-189. |
R832734 (2009) R832734 (Final) R834513 (2010) R834513 (2011) R834513 (Final) |
Exit Exit Exit |
|
|
Huen K, Barcellos L, Beckman K, Rose S, Eskenazi B, Holland N. Effects of PON polymorphisms and haplotypes on molecular phenotype in Mexican-American mothers and children. Environmental and Molecular Mutagenesis 2011;52(2):105-116. |
R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) |
Exit Exit |
|
|
Huen K, Bradman A, Harley K, Yousefi P, Boyd Barr D, Eskenazi B, Holland N. Organophosphate pesticide levels in blood and urine of women and newborns living in an agricultural community. Environmental Research 2012;117:8-16. |
R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) R834513C002 (2010) R834513C002 (2012) R834513C002 (2013) R834513C003 (2010) R834513C003 (2011) R834513C003 (2012) |
Exit Exit Exit |
|
|
Marks AR, Harley K, Bradman A, Kogut K, Barr DB, Johnson C, Calderon N, Eskenazi B. Organophosphate pesticide exposure and attention in young Mexican-American children: the CHAMACOS Study. Environmental Health Perspectives 2010;118(12):1768-1774. |
R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) |
|
|
|
Neri M, Bonassi S, Knudsen LE, Sram RJ, Holland N, Ugolini D, Merlo DF. Children’s exposure to environmental pollutants and biomarkers of genetic damage:I. Overview and critical issues. Mutation Research-Reviews in Mutation Research 2006;612(1):1-13. |
R832734 (2006) R832734 (2007) R832734 (Final) R831710 (Final) R831710C003 (2006) |
Exit Exit Exit |
|
|
Neri M, Ugolini D, Bonassi S, Fucic A, Holland N, Knudsen LE, Sram RJ, Ceppi M, Bocchini V, Merlo DF. Children’s exposure to environmental pollutants and biomarkers of genetic damage: II. Results of a comprehensive literature search and meta-analysis. Mutation Research-Reviews in Mutation Research 2006;612(1):14-39. |
R832734 (2006) R832734 (Final) R831710 (Final) R831710C003 (2006) |
Exit Exit Exit |
|
|
Quiros-Alcala L, Alkon AD, Boyce WT, Lippert S, Davis NV, Bradman A, Barr DB, Eskenazi B. Maternal prenatal and child organophosphate pesticide exposures and children's autonomic function. NeuroToxicology 2011;32(5):646-655. |
R832734 (Final) R834513 (2010) R834513 (2011) R834513 (2012) R834513 (2013) R834513 (2015) R834513 (2016) R834513 (Final) R834513C001 (2010) R834513C001 (2011) R834513C001 (2012) |
Exit Exit Exit |
|
|
Sexton K, Adgate JL, Church TR, Hecht SS, Ramachandran G, Greaves IA, Fredrickson AL, Ryan AD, Carmella SG, Geisser MS. Children's exposure to environmental tobacco smoke:using diverse exposure metrics to document ethnic/racial differences. Environmental Health Perspectives 2004;112(3):392-397. |
R832734 (Final) R826789 (2002) R826789 (Final) |
|
|
|
Sly PD Eskenazi B, Pronczuk J, Sram R, Diaz-Barriga F, Machin DG, Carpenter DO, Surdu S, Meslin EM. Ethical issues in measuring biomarkers in children's environmental health. Environmental Health Perspectives 2009;117(8):1185-1190. |
R832734 (2009) R832734 (Final) R831710 (Final) R834513 (2010) R834513 (2011) R834513 (Final) |
|
|
|
Vose SC, Holland NT, Eskenazi B, Casida JE. Lysophosphatidylcholine hydrolases of human erythrocytes, lymphocytes, and brain:sensitive targets of conserved specificity for organophosphorus delayed neurotoxicants. Toxicology and Applied Pharmacology 2007;224(1):98-104. |
R832734 (2007) R832734 (Final) R831710 (2005) R831710 (Final) |
Exit Exit Exit |
Supplemental Keywords:
genetic predisposition, genetic polymorphisms, Health, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, ENVIRONMENTAL MANAGEMENT, Health Risk Assessment, Environmental Policy, Children's Health, Biochemistry, Genetics, Risk Assessment, Human Health Risk Assessment, pesticide exposure, genetic polymorphisms, susceptibility, human enzyme paraoxonase, children's vulnerablity, developmental neurotoxicity, assessment of exposure, farmworkers, gene-environment interaction, neurological developmentRelevant Websites:
The Center for Environmental Research and Children's Health (CERCH) Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
