Grantee Research Project Results
Final Report: Biomarkers Of Health Risks Associated With Environmental Exposure To Arsenic
EPA Grant Number: R832735Title: Biomarkers Of Health Risks Associated With Environmental Exposure To Arsenic
Investigators: Stýblo, Miroslav , García-Vargas, Gonzalo G , Del Razo, Luz M. , Loomis, Dana , Ethan, Lange , Drobna, Zuzana
Institution: University of North Carolina at Chapel Hill , University of Nevada - Reno
EPA Project Officer: Callan, Richard
Project Period: February 13, 2006 through February 12, 2009 (Extended to February 12, 2010)
Project Amount: $749,988
RFA: Early Indicators of Environmentally Induced Disease (2004) RFA Text | Recipients Lists
Research Category: Human Health
Objective:
The main goals of research included in this project were (A) to characterize the role of metabolism and specific metabolites of inorganic arsenic (iAs) in adverse effects of iAs exposure and (B) to identify biomarkers of individual susceptibility to these effects.
The specific aims of this research were:
- To examine interindividual variations in urinary profiles of methylarsonite (MAsIII) and dimethylarsinite (DMAsIII) in individuals exposed to iAs in drinking water.
- To characterize the relationship between urinary levels of MAsIII and DMAsIII and markers of carcinogenic and diabetogenic effects of iAs.
- To examine the role of genetic polymorphism for arsenic (+3 oxidation state) methyltransferase (AS3MT) in modulation of the metabolism and adverse health effects of iAs.
- To study the genotype-phenotype relationship using recombinant genetic variants of AS3MT.
The main part of this project (aims 1, 2 and 3) was a population-based study carried out in two arsenicosis endemic areas of Mexico: Zimapan and La Laguna (Lagunera) regions. Laboratory studies examining the metabolic phenotypes of AS3MT variants found in the Mexican populations (aim 4) represented only a small component of the project.
Summary/Accomplishments (Outputs/Outcomes):
METHODS
Study Subjects and Medical Exam
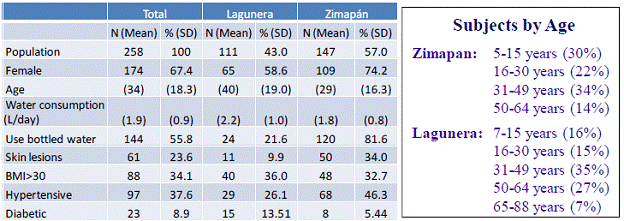
147 Zimapan and 111 Lagunera (genetically unrelated) residents, both females (109 and 65, respectively) and males, adults and children (5-year old or older) were recruited for this project. Basic characteristics of the study population are provided in Table 1. A questionnaire focusing on exposure to iAs, sources of drinking water, and history of disease was administered to all consenting study subjects. All subjects were examined by expert physicians for skin lesions that are known to result from chronic exposures to iAs, including hypo- or hyper-pigmentation, keratosis, Bowen’s disease, and skin cancer. Oral glucose tolerance test (OGTT) was administered to all subjects, except those who were previously diagnosed with diabetes.
Table 1: Basic characteristics of subjects recruited for the population study (Specific aims 1-3).
Samples and Analyses
- Samples of drinking water provided by subjects were analyzed for total arsenic (tAs) content by hydride generation (HG)-atomic fluorescence spectrometry (AFS).
- HG-atomic absorption spectrometry (AAS) with a cryotrap (CT) for capture and separation of hydrides was used for analysis of iAs metabolites in spot urine. To avoid degradation of unstable MAsIII and DMAsIII, the trivalent forms of As were analyzed in freshly collected urines in Del Razo’s and Garcia-Vargas’ laboratories that are located in close proximities to the Zimapan and Lagunera study areas, respectively.
- tAs was analyzed in frozen urine aliquots by HG-AFS after digestion in hydrochloric, nitric, perchloric, and sulfuric acids.
- Glucose concentrations were determined by a glucometer in fasting blood (FBG) and 2-hour blood (2HBG) collected before and during OGTT.
- Concentration of glycated hemoglobin (HbA1c) was measured in fasting blood.
- Fasting blood insulin (FBI) was measured by ELISA.
- Activities of alanine and aspartate aminotransferases, the markers of liver injury, were measured in fasting blood.
Indicators of iAs Exposure and Metabolism
The following indicators were used to determine individual exposure to iAs in drinking water:
- The concentration of tAs in water from the participants’ households.
- The concentration of tAs in urines collected during medical exams.
- The cumulative exposure was calculated using historical data on levels of arsenic in the local water supplies and data on each subject’s length of residence (provided in the questionnaire).
The metabolism of iAs was characterized by the relative amount of the metabolites of iAs in urine (% of tAs in urine) and by the ratios of DMAs/MAs, MAs/iAs, and (DMAs+MAs)/iAs.
Diagnosis of Diabetes
Diabetic individuals were identified by FBG ≥126 mg/dl, 2HBG ≥200 mg/dl, existing diagnosis of diabetes, or use of anti-diabetic medication (indicated in questionnaires). FPI, HbA1c, and HOMA-IR (FPI [µU/mL] x FBG [mmol/L]/22.5) were used to further characterize the glucose homeostasis and insulin resistance for each of the study subjects.
Genotyping
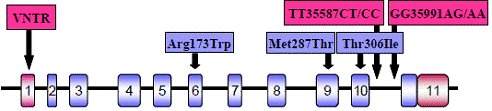
Genotyping focused on AS3MT, the key enzyme in the pathway for metabolism of iAs. The TaqMan assay was used for detection of the functional single nucleotide polymorphisms (SNPs), i.e., SNPs that have been linked by previous studies to specific patterns of iAs metabolism or to differences in susceptibility to iAs toxicity (Fig. 1). Variable number of tandem repeats (VNTR) that is thought to affect AS3MT expression was analyzed by sequencing of the AS3MT 5’-UTR region amplified by PCR.
Fig. 1: Location of the functional SNPs and VNTR in AS3MT gene.
Statistical Analysis
Standard descriptive analyses were carried out using means and standard errors for continuous variables and frequencies for categorical variables. The distributions of continuous variables were examined graphically using histograms and normal probability plots. The concentration of tAs in water and concentrations of tAs and iAs metabolites in urine were approximately log-normally distributed, so log transformations were used for analyses that assume normality.
Associations of iAs and urinary metabolites of iAs with diabetes status and the presence of skin lesions were examined using logistic regression to estimate odds ratios (ORs) and 95% confidence intervals (CIs). Diabetes was classified by the combination of either FBG≥126 or 2HBG≥200 and report of doctor-diagnosed diabetes or use of diabetes medication. tAs concentration in drinking water and urine and urinary concentrations of iAs and its metabolites were entered both as continuous variables and in categorical form. To control for potential confounding, age, sex, hypertension (SBP≥140, DBP≥90 or reported use of anti-hypertensive drugs) and obesity (BMI>30) were included as covariates in models for diabetes, while age and sex alone were included in models for skin lesions. Age was entered as a continuous variable in all models. Concentrations of tAs and urinary metabolites of iAs were not normalized to creatinine concentration because of evidence that diabetes may affect creatine production. However, analyses of urinary metabolites of iAs were conducted both with and without urinary creatinine concentration as a covariate to assess possible effects of variation in urine volume.
Associations of continuous variables for 2HBG, FBG, FPI, and HOMA-IR with log-transformed measures of iAs and iAs metabolites were analyzed by linear regression. Age, sex, hypertension and obesity, classified as described above, were included as covariates in these models.
RESULTS
Exposure to iAs
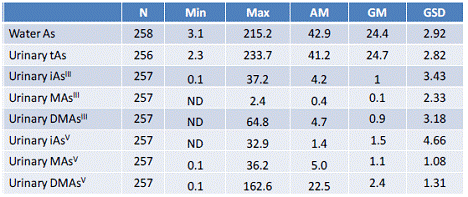
The concentrations of As in the samples of drinking water provided by study subjects ranged from 3 to 215 μg/L. Urines contained 2 to 234 ng tAs/mL (5 to 1512 ng tAs/mg creatinine) (Table 2). The tAs levels determined by HG-AFS in the digested urines correlated strongly with the sum of tri- and pentavalent metabolites of iAs (iAs + MAs + DMAs) determined in urines by HG-CT-AAS (R = 0.975; p < 0.0001). This correlation suggests that iAs metabolites accounted for all or most of the arsenic in urines and that urines did not contain thioarsenicals or complex organic As species that are not products of iAs metabolism (e.g., arsenobetaine or arsenocholine) and cannot be detected by HG-CT-AAS. A weak, but statistically significant correlation was found between the level of As in drinking water and tAs level in urine (data not shown). This correlation was stronger for the logarithmically transformed levels of As in water and tAs in urine, both before and after normalization for creatinine: R = 0.42 (p < 0.0001) and R = 0.36 (p < 0.0001), respectively.
Urinary Metabolites of iAs
Most of the urine samples collected in Zimapan and Lagunera regions contained all known metabolites of iAs, including iAsIII, iAsV, MAsIII, MAsV, DMAsIII, and DMAsV. Together, DMAsIII and DMAsV were the most prevalent forms of As in urines, regardless the exposure level, age, or gender of the study subjects (Table 2). MAsIII was detected in 67% urines collected in Zimapan and 100% urines collected in Lagunera; DMAsIII was detected in 75% and 100% urines collected in Zimapan and Lagunera, respectively. On average, MAsIII accounted for a larger portion of tAs (1.7%) in urines of young subjects (≤18-year old) as compared to subjects older than 18 years (1.3%, p = 0.0187) or older than 50 years (1.0%, p = 0.0005). The DMAs/MAs ratio was also lower in urines of the young subjects (4.7) as compared to the other age groups: 5.9 (p = 0.0067) and 6.1 (p = 0.004), respectively.
Table 2: Indicators of the exposure to iAs and of iAs metabolism (ng As/mL).
Associations Between Skin Lesions and iAs Exposure or Metabolism
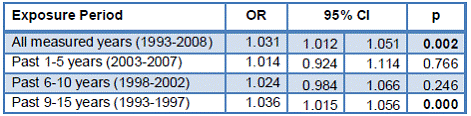
In Zimapan, that had historically higher levels of As in drinking water supplies, 34% subjects were diagnosed with at least one type of skin lesion as compared to 18% subjects in Lagunera. Surprisingly, the current exposure to iAs, represented by either As in drinking water or tAs in urine, was negatively associated with risk of skin lesions (Table 3). Knowing that recent interventions lowered As levels in drinking water supplies in both study areas, we have also examined associations between skin lesion and the cumulative exposure to iAs. We found that the cumulative exposure to iAs over the past 15 years (from 1993 to 2008) significantly increased risk of skin lesions (Table 4). Notably, the highest and statistically significant odds ratio (OR) was found for the chronologically earliest segment of 1993-1997. This data indicate that the past exposure to high, unregulated levels of iAs in drinking water played a decisive role in the development of skin lesions among our study subjects.
Table 3: Associations between skin lesions and the current exposure to iAs and the urinary metabolites of iAs.
Table 4: Associations between skin lesions and the cumulative exposure to iAs in drinking water.
Associations Between Diabetes and iAs Exposure or Metabolism
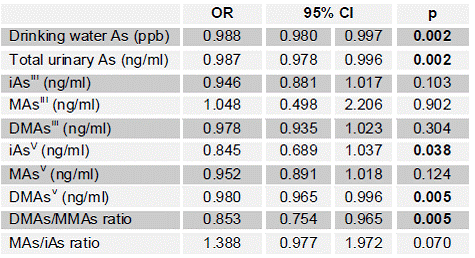
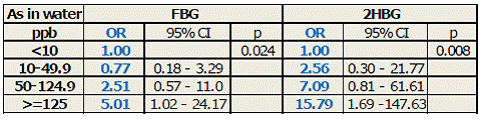
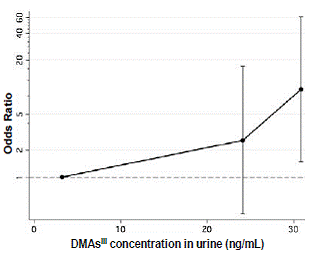
FBG, 2HBG, previous diagnosis, and reported use of anti-diabetic medication were used to identify diabetic subjects. Notably, strong positive correlations (R > 0.78; p < 0.0001) were found between FBG and 2HBG and between FBG and HbA1c values. Odds of diabetes by either FBG (≥126 mg/dl) or 2HBG (≥200 mg/dl) increase proportionally to iAs concentration in drinking water (Table 5). Odds of diabetes by either measure increased about 1% per ppb of As in drinking water (p = 0.002). Increasing levels of HBA1c in blood were also associated with increasing concentration of iAs in drinking water, both with and without log-transformation of both variables (p = 0.03 and p = 0.003, respectively) after adjustment for age, sex, hypertension and obesity. Diabetes was not significantly associated with urinary tAs concentration, or with concentrations of tri- or pentavalent iAs in urine. However, the odds of diabetes measured by FBG, previous diagnosis or medication increased approximately 5% per ng/ml of urinary DMAsIII (p = 0.05) (Fig. 2). The odds of diabetes by either FBG or 2HBG increased by about 6% per ng/ml of urinary DMAsIII after adjustment for creatinine and these associations were statistically significant (p ~ 0.02). In spite of a high OR value, MAsIII was not significantly associated with diabetes. Notably, we found a negative association between HOMA-IR and exposure to iAs represented either by As level in drinking water (p < 0.0001) or urinary tAs (p = 0.003). A similar negative association (p < 0.001) was found between FPI and As in water (adjusted for age, sex, hypertensions and obesity). Negative associations were also found for log-transformed FPI value and both log-transformed As level in water (p < 0.001) and log-transformed urinary tAs (p < 0.001). Taken together, these data indicate that (i) the lower HOMA-IR values associated with iAs exposure are due to decreased FPI and that (ii) the increase in FBG is not due to insulin resistance, but could be a results of an insufficient insulin production by pancreatic β-cells.
Table 5: Risk of diabetes as a function of iAs concentration in drinking water; OR, odds ratio; 95% CI, 95% confidence interval.
Fig.2: Risk of diabetes as a function of urinary DMAsIII (OR and 95% CI are shown).
AS3MT genotypes
We found only one of the three previously described exonic SNPs, Met287Thr, including 28 heterozygotes and 3 homozygotes. Both functional intronic SNPs, T35587C and G35991A, were found with frequencies similar to those previously reported by other laboratories. Only two of the 3 previously described VNTR structures were detected: AB and A2B with the respective frequencies of 83.8% and 16.2% in Zimapan and 56.6% and 43.4% in Lagunera.
Association Between AS3MT Polymorphism and Metabolisms of iAs
The following are statistically significant associations for each of the AS3MT genotypes:
- Met287Thr. The CC or TC (Thr/Thr or Met/Thr) genotypes were associated with higher urinary concentrations of MAsIII (p = 0.018) and DMAsIII (p = 0.022) as compared to the wild type, TT (Met/Met) genotype.
- T35587C. The T/C genotype was associated with a higher MAs/iAs ratio (p = 0.031) and higher (DMAs+MAs)/iAs ratio (p = 0.001) as compared to T/T genotype. These differences were statistically significant also for the logarithmically transformed values.
- G35991A. The A/A or G/A genotypes were associated with a higher MAs/iAs ratio (p = 0.002) and higher (DMAs+MAs)/iAs ratio (p = 0.002) as compared to G/G genotype. These differences were statistically significant also for the logarithmically transformed values.
- VNTR. VNTR structure in the 5’UTR region of AS3MT was significantly associated only with the DMAs/MAs ratio. The carriers of A2B structure had higher DMAs/MAs ratio in urine as compared to AB carriers (p = 0.016).
Association Between AS3MT Polymorphism and Diabetes
Among all AS3MT polymorphisms examined, Met287Thr showed the strongest association with diabetes. The AS3MT/287Thr variant [T(C)/C)] was associated with a higher FBG (114 ±59 mg/dl vs. 89.85 ±35 for T/T; p = 0.001) and with a marginally increased 2HBG (135 ±98 vs. 103 ± 46; p = 0.091). T(C)/C carriers were more likely to have FBG ≥126 mg/dl (odds ratio 2.36) and 2HBG >200 mg/dl (odds ratio 2.86), as compared to T/T carriers, although neither association was statistically significant. The average HbA1c level was also marginally higher in the blood of the T(C)/C carriers: 7 ±1.87% vs. 6.3 ±1.15% for the T/T carriers (p = 0.070). The associations of FBG and HbA1c with Met287Thr genotype were statistically significant after logarithmic transformation.
Specific Aim 4
Because only one AS3MT polymorph was identified among residents of Zimapan and Lagunera (Met287Thr), the work in this aim focused entirely on comparison of the catalytic properties of the wild-type (wt) AS3MT and the AS3MT/287Thr variant.
METHODS
Generation of Recombinant AS3MT Variants
Recombinant proteins were produced after expression in E. coli, using RSETa expression vectors containing human wtAS3MT and AS3MT/287Thr cDNA (gift from Dr. David J. Thomas, US EPA).
In Vitro Methylation Assay
The in vitro reaction mixture contained 100 mM TRIS-HCl buffer (pH 7.4), 1 mM tris(2carboxyethyl)phosphine, 1 mM S-adenosylmethionine, 5 μg AS3MT protein, and sodium arsenite (0.1 to 10 μM iAsIII) as a substrate. The reaction was carried out at 37oC. Products of the enzymatic methylation of iAs were determined by the oxidation state specific HG-CT-AAS.
RESULTS
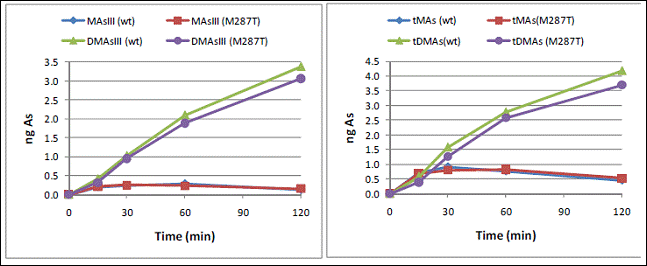
The methylation of iAsIII by either AS3MT variant yielded both tri- and pentavalent MAs and DMAs; no TMAsO was detected. Consistent with previous reports, MAsIII and MAsV were the intermediary, and DMAsIII and DMAsV were the final products of iAs methylation for both proteins. Notably, DMAsIII accounted for 75-90% of total DMAs produced over a 2-hour reaction period at the range of substrate concentrations of 0.1 to 10 μM. We found no significant differences between the rates of iAsIII methylation by the two AS3MT variants; yields of the methylated tri- and pentavalent metabolites were also similar in in vitro reaction systems containing micromolar or sub-micromolar iAsIII (Fig. 3). No statistically significant differences were found between wtAS3MT- and AS3MT/287Thr-catalyzed reactions at the range of 0.1 to 10 μM iAsIII. However, the DMAs/MAs ratios were consistently higher for the reactions catalyzed by AS3MT/287Thr, suggesting that this AS3MT polymorph may be more efficient than wtAS3MT in converting MAs to DMAs.
Fig. 3: Methylation of iAsIII (1 μM) by recombinant wtAS3MT and AS3MT/287Thr: The production of trivalent MAsIII and DMAsIII (left panel) and the total MAs (tMAs) and total DMAs (tDMAs) yields (right panel) over a 2-hour reaction period. (n = 2 for each time point).
Previous reports from Thomas’ laboratory showed that glutathione (GSH), the most abundant intracellular reductant, modulates in vitro methylation of iAs by recombinant rat As3mt. In our study, we examined effects of a physiological concentration of GSH (5 mM) on the rate and pattern of iAsIII methylation by recombinant human wtAS3MT and the AS3MT/287Thr variant. The presence of GSH markedly increased the rates and yields of iAsIII methylation with similar effects for both enzymes (data not shown). In the assay mixtures containing 1 μM iAsIII, GSH stimulated DMAs production, increasing the DMAs/MAs ratio. In contrast, in the assay mixtures containing 10 μM iAsIII, the presence of GSH resulted in a several-fold increase in MAs production and dramatically decreased DMAs/MAs ratios.
In summary, results of the in vitro study using recombinant AS3MT variants are in disagreement with results of the population study in Zimapan and Lagunera which found the AS3MT/287Thr variant to be associated with higher concentrations of MAsIII and DMAsIII in urine of Zimapan and Lagunera residents exposed to iAs in drinking water.
Conclusions:
- Results of our project confirmed a previously reported, but frequently questioned, association between chronic exposure to iAs in drinking water and diabetes mellitus. Using three diabetes indicators (FBG, 2HBG, and HbA1c), we were able to show that the risk of developing diabetes significantly increases with increasing concentration of iAs in drinking water. Notably, the risk of diabetes in our study was linked to current, rather than historical or cumulative exposure to iAs. In contrast, skin lesions, a typical sign of chronic iAs toxicity, was significantly associated with the historical/cumulative, but not with current exposure to iAs in drinking water. These findings suggest that while development of skin lesions may require a prolong exposure to iAs, diabetes may develop already after a short exposure and may be reversed after the exposure decreases or ends.
- The negative associations we found between iAs exposure and HOMA-IR or FPI indicate that mechanisms by which iAs exposure produces diabetes may differ from those underlying common type-2 diabetes, which is typically characterized by chronic insulinemia and insulin resistance.
- We found that risk of diabetes significantly increases with increasing concentration of DMAsIII in urine and that carriers of the AS3MT/287Thr variant had, generally, higher FBG, 2HFBG and HbA1c levels in blood. Importantly, this genotype was also associated with higher concentrations of MAsIII and DMAsIII in urine. These data strongly suggest that the AS3MT/287Thr variant is associated with an increased risk of developing diabetes, possibly, due to a greater capacity to convert iAs to MAsIII and, particularly, to DMAsIII. It should be noted, however, that we found no differences in the rates and patterns of in vitro methylation of iAs (including MAsIII and DMAsIII production) by recombinant wtAS3MT and the AS3MT/287Thr variant. Thus, more studies will be needed to further characterize and explain the association between the Met287Thr polymorphism, iAs metabolism, and risk of diabetes associated with iAs exposure.
In summary, the results of our study show (i) that FBG, 2HBG and HbA1c, but not FPI or HOMA-IR, are reliable indicators of the diabetogenic effects of chronic exposure to iAs and (ii) that the concentration of DMAsIII in urine, alone or in combination with Met287Thr polymorphism for AS3MT, can be used to identify individuals with increased susceptibility to these effects.
Journal Articles on this Report : 8 Displayed | Download in RIS Format
| Other project views: | All 19 publications | 9 publications in selected types | All 9 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Del Razo LM, Garcia-Vargas GG, Valenzuela OL, Castellanos EH, Sanchez-Pena LC, Currier JM, Drobna Z, Loomis D, Styblo M. Exposure to arsenic in drinking water is associated with increased prevalence of diabetes: a cross-sectional study in the Zimapan and Lagunera regions in Mexico. Environmental Health 2011;10:73. |
R832735 (Final) |
Exit Exit Exit |
|
|
Drobna Z, Del Razo LM, Garcia-Vargas GG, Sanchez-Pena LC, Barrera-Hernandez A, Styblo M, Loomis D. Environmental exposure to arsenic, AS3MT polymorphism and prevalence of diabetes in Mexico. Journal of Exposure Science & Environmental Epidemiology 2013;23(2):151-155. |
R832735 (Final) |
Exit Exit |
|
|
Drobná Z, Del Razo LM, Garcia-Vargas G, Sánchez-Ramírez B, González-Horta C, Ballinas-Casarrubias L, Loomis D, Stýblo M. Identification of the GST-T1 and GST-M1 Null Genotypes Using High Resolution Melting Analysis. Chemical Research in Toxicology 2012;25(1):216-224. |
R832735 (Final) |
Exit |
|
|
Hernandez-Zavala A, Valenzuela OL, Matousek T, Drobna Z, Dedina J, Garcia-Vargas GG, Thomas DJ, Del Razo LM, Styblo M. Speciation of arsenic in exfoliated urinary bladder epithelial cells from individuals exposed to arsenic in drinking water. Environmental Health Perspectives 2008;116(12):1656-1660. |
R832735 (2007) R832735 (2008) R832735 (Final) |
Exit Exit |
|
|
Hernandez-Zavala A, Matousek T, Drobna Z, Paul DS, Walton F, Adair BM, Dedina J, Thomas DJ, Styblo M. Speciation analysis of arsenic in biological matrices by automated hydride generation-cryotrapping-atomic absorption spectrometry with multiple microflame quartz tube atomizer (multiatomizer). Journal of Analytical Atomic Spectrometry 2008;23(3):342-351. |
R832735 (2006) R832735 (2007) R832735 (2008) R832735 (Final) |
Exit |
|
|
Matousek T, Hernandez-Zavala A, Svoboda M, Langrova L, Adair BM, Drobna Z, Thomas DJ, Styblo M, Dedina J. Oxidation state specific generation of arsines from methylated arsenicals based on L-cysteine treatment in buffered media for speciation analysis by hydride generation-automated cryotrapping-gas chromatography-atomic absorption spectrometry with the multiatomizer. Spectrochimica Acta Part B: Atomic Spectroscopy 2008;63(3):396-406. |
R832735 (2007) R832735 (Final) |
Exit |
|
|
Smeester L, Rager JE, Bailey KA, Guan X, Smith N, García-Vargas G, Del Razo L-M, Drobná Z, Kelkar H, Stýblo M, Fry RC. Epigenetic Changes in Individuals with Arsenicosis. Chemical Research in Toxicology 2011;24(2):165-167. |
R832735 (Final) |
Exit Exit |
|
|
Valenzuela OL, Drobna Z, Hernandez-Castellanos E, Sanchez-Pena LC, Garcia-Vargas GG, Borja-Aburto VH, Styblo M, Del Razo LM. Association of AS3MT polymorphisms and the risk of premalignant arsenic skin lesions. Toxicology and Applied Pharmacology 2009;239(2):200-207. |
R832735 (Final) |
Exit |
Supplemental Keywords:
arsenic, drinking water, chronic exposure, cumulative exposure, skin lesions, diabetes mellitus, arsenic methyltransferase, arsenic metabolites, urine, biomarkers of exposure and disease, genetic polymorphism, susceptibility to arsenic toxicity, mechanism of arsenic-induced diabetes, RFA, Scientific Discipline, Water, POLLUTANTS/TOXICS, Health Risk Assessment, Arsenic, Biochemistry, Drinking Water, Genetics, Water Pollutants, bioavailability, health effects, cancer, drinking water contaminants, drinking water monitoring, genotype distributionProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.