Grantee Research Project Results
2006 Progress Report: Biomarkers Of Health Risks Associated With Environmental Exposure To Arsenic
EPA Grant Number: R832735Title: Biomarkers Of Health Risks Associated With Environmental Exposure To Arsenic
Investigators: Stýblo, Miroslav , García-Vargas, Gonzalo G , Del Razo, Luz M. , Lange, Ethan
Current Investigators: Stýblo, Miroslav , García-Vargas, Gonzalo G , Del Razo, Luz M. , Loomis, Dana , Ethan, Lange , Drobna, Zuzana
Institution: University of North Carolina at Chapel Hill , Cinvestav-IPN , Juarez State University
Current Institution: University of North Carolina at Chapel Hill , University of Nevada - Reno
EPA Project Officer: Callan, Richard
Project Period: February 13, 2006 through February 12, 2009 (Extended to February 12, 2010)
Project Period Covered by this Report: February 18, 2006 through February 17,2007
Project Amount: $749,988
RFA: Early Indicators of Environmentally Induced Disease (2004) RFA Text | Recipients Lists
Research Category: Human Health
Objective:
The objectives are to:
- Examine interindividual variations in urinary profiles of methylarsonite (MAsIII) and dimethylarsinite (DMAsIII), the toxic metabolites of arsenic (As), in individuals exposed to inorganic As (iAs) in drinking water.
- Characterize the relationship between the urinary levels of MAsIII and DMAsIII and markers of carcinogenic and diabetogenic effects of iAs.
- Examine the role of genetic polymorphism for arsenic (+3 oxidation state) methyltransferase (AS3MT) in modulation of the metabolism and adverse health effects of iAs.
- Study the genotype-phenotype relationship using recombinant genetic variants of AS3MT.
Approach:
Study participants will be recruited among the residents (adults and children) of Zimapan and Lagunera regions (Mexico) who are exposed to a wide range of iAs concentrations in drinking water. Metabolites of iAs, including MAsIII and DMAsIII, will be analyzed in freshly collected urines. Markers of (pre)carcinogenic and diabetogenic effects of iAs will be examined, including skin lesions, concentration of transforming growth factor-α (TGF-α) in the urine and in exfoliated bladder epithelial cells, fasting glucose and insulin concentrations and hemoglobin A1c levels in the blood, and results of oral glucose tolerance test. AS3MT genotypes will be analyzed, using blood RNA. The genetic variants of AS3MT found in the study participants will then be cloned, and expressed. Purified recombinant AS3MT variants will be used in an in vitro assay system to further examine links between the specific genotypes and the pattern of iAs metabolism. Finally, relationships between the markers of adverse health effects, the AS3MT genotypes, and the in vivo and in vitro metabolic patterns for iAs will be characterized.
Progress Summary:
This project is carried out in two of the arseniasis-endemic areas of Mexico, Zimapan, and Lagunera regions. To date, 196 study subjects (53 men, 143 women; ranging from 5 to 66 years of age) were recruited in the Zimapan region. Concentrations of total As in urine of these individuals range from 2 to 533 μg/L: 123 subjects (29 men) have urinary levels of As below 50 μg/L; 36 subjects (14 men) have As above 50, but below 100 μg/L; 19 subjects (3 men) have As above 100, but below 150 ppb; and 18 subjects (7 men) have As levels higher than 150 μg/L. These urinary As levels represent exposures to iAs in drinking water at levels ranging from 1 to 762 μg/L.
During the medical examination, severe cases of skin lesions, including skin cancer, were diagnosed in 15 study subjects exposed to high levels of iAs in drinking water. These individuals are now undergoing medical treatments in specialized clinical facilities in Zimapan and Mexico City. Thus, our work helped to identify serious health risks which would otherwise be overlooked or left without a proper treatment.
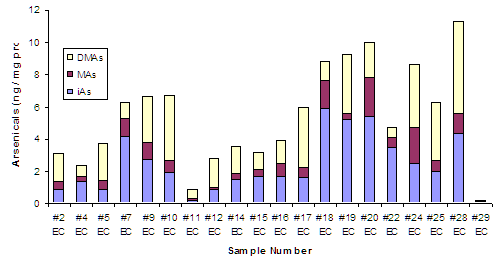
We have been able to further optimize the hydride generation-atomic absorption spectrometry (HG-AAS) procedure and instrumentation used for the speciation analysis of As in urine. The optimized instrumentation includes a newly developed multiatomizer for As detection, resulting in a dramatic improvement of detection limits (DL) for all As species of interest. The current DL values range from 8 to 20 pg. Using the optimized HG-AAS design, we were able to detect and quantify iAs metabolites not only in urine, but also in exfoliated cells isolated from urine of the study participants. To our best knowledge, this is the first successful attempt to speciate As in exfoliated human bladder cells. All relevant iAs metabolites were detected in these cells (Figure 1). Notably, the concentrations of individual metabolites retained in cells did not correlate with the levels of these metabolites in urine. These results indicate that speciation of As in exfoliated cells may be more reliable than As speciation in urine as a biomarker of the adverse effects of As in human urinary bladder.
Figure 1. Speciation of As in Exfoliated Cells Isolated from Urine of Individuals Exposed to iAs in Drinking Water in Zimapan Region: iAs: Inorganic As; MAs: Methyl-As; DMAs: Dimethyl-As.
The analysis of DNA samples collected in the study area has identified two new AS3MT polymorphisms. Two independent high-frequency intronic deletions were detected between exons 2 and 3, including a CTC deletion at the position 5523–5525 and a CTC deletion at the position 5526–5528. We are currently examining relationships between these polymorphisms and urinary profiles of iAs metabolites. If a significant association is found, these polymorphisms will be evaluated (along with the other previously identified functional polymorphisms) with respect to the urinary profiles of iAs metabolites and markers of adverse effects of iAs exposure in both study areas.
Expected Results:
Given the results of preliminary studies, we expect to find positive correlations between the urinary levels of MAsIII and/or DMAsIII and markers of adverse health effects in exposed individuals. We also expect to link specific metabolic profiles for iAs to specific AS3MT genotypes. Ultimately, this project will provide a set of metabolic, biochemical, and genetic biomarkers that, taken together, may significantly improve the assessment of health risks associated with chronic exposures to iAs and identification of individuals with increased susceptibility to toxic and cancer promoting effects associated with these exposures.
Future Activities:
- Sample collection will be completed in the Zimapan region. Recruitment of study participants will be completed, and sample collection will start in the Lagunera region.
- Analysis of unstable As species (trivalent arsenicals) will be carried out in freshly collected urine samples in Zimapan and Lagunera regions. In Zimapan, this analysis will be completed during the next year and followed by analysis of stable pentavalent arsenicals in frozen aliquots of urine.
- All participants in Zimapan and some in Lagunera will undergo an oral glucose tolerance test; results of this test will be continuously evaluated.
- DNA and RNA samples isolated in Zimapan will be aliquoted, and aliquots will be shipped to the University of North Carolina at Chapel Hill (UNC) for genotyping.
- Exfoliated bladder cells will be analyzed for the content of transforming growth factor (TGF)-α.
- The original study design included examination of the relationship between TGF-α in exfoliated bladder cells and the concentration of iAs metabolites in urine. We would like to extend this design to include analysis of iAs metabolites in exfoliated bladder cells, using the optimized HG-AAS. Evaluation of associations between the retention of these metabolites and TGF-α levels in the exfoliated cells may provide novel biomarkers of adverse effects of As in human bladder.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 19 publications | 9 publications in selected types | All 9 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Hernandez-Zavala A, Matousek T, Drobna Z, Paul DS, Walton F, Adair BM, Dedina J, Thomas DJ, Styblo M. Speciation analysis of arsenic in biological matrices by automated hydride generation-cryotrapping-atomic absorption spectrometry with multiple microflame quartz tube atomizer (multiatomizer). Journal of Analytical Atomic Spectrometry 2008;23(3):342-351. |
R832735 (2006) R832735 (2007) R832735 (2008) R832735 (Final) |
Exit |
Supplemental Keywords:
Human health, sensitive populations, dose-response, arsenic, biomarkers, carcinogen, chemicals, toxics, metals, metalloids, trace elements, gene, epidemiology, toxicology, medicine, analytical chemistry, biochemistry, analytical, spectrometry,, RFA, Scientific Discipline, Water, POLLUTANTS/TOXICS, Health Risk Assessment, Arsenic, Biochemistry, Drinking Water, Genetics, Water Pollutants, bioavailability, health effects, cancer, drinking water contaminants, drinking water monitoring, genotype distributionProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.