Grantee Research Project Results
2007 Progress Report: Biomarkers Of Health Risks Associated With Environmental Exposure To Arsenic
EPA Grant Number: R832735Title: Biomarkers Of Health Risks Associated With Environmental Exposure To Arsenic
Investigators: Stýblo, Miroslav , García-Vargas, Gonzalo G , Del Razo, Luz M. , Lange, Ethan , Drobná, Zuzana
Current Investigators: Stýblo, Miroslav , García-Vargas, Gonzalo G , Del Razo, Luz M. , Loomis, Dana , Ethan, Lange , Drobna, Zuzana
Institution: University of North Carolina at Chapel Hill , Cinvestav-IPN , Juarez State University
Current Institution: University of North Carolina at Chapel Hill , University of Nevada - Reno
EPA Project Officer: Callan, Richard
Project Period: February 13, 2006 through February 12, 2009 (Extended to February 12, 2010)
Project Period Covered by this Report: February 18, 2007 through February 17,2008
Project Amount: $749,988
RFA: Early Indicators of Environmentally Induced Disease (2004) RFA Text | Recipients Lists
Research Category: Human Health
Objective:
1. To examine interindividual variations in urinary profiles of methylarsonite (MAsIII) and dimethylarsinite (DMAsIII), the toxic metabolites of arsenic (As), in individuals exposed to inorganic As (iAs) in drinking water.
2. To characterize the relationship between the urinary levels of MAsIII and DMAsIII and markers of carcinogenic and diabetogenic effects of iAs.
3. To examine the role of genetic polymorphism for arsenic (+3 oxidation state) methyltransferase (AS3MT) in modulation of the metabolism and adverse health effects of iAs.
4. To study the genotype-phenotype relationship using recombinant genetic variants of AS3MT.
Progress Summary:
The main part of this project is a population-based study carried out in two of the arseniasis endemic areas of Mexico, the Zimapan and Lagunera regions (Specific Aims 1-3). Work in Zimapan involving 149 subjects, 39 men (26.2%) and 110 women (73.8%) is nearly complete. All subjects underwent medical examinations and oral glucose tolerance tests (OGTT), providing blood samples for glucose and insulin analyses and for DNA isolation. The required analyses of blood, plasma, and urine samples collected in the Zimapan area have been completed in the subcontractor laboratory in CINVESTAV-IPN in Mexico. In Lagunera, recruitment was completed in summer 2007. Here, 113 subjects, 48 men (42.5%) and 65 women (57.5%), were recruited. All of these subjects underwent medical exams and OGTTs and provided blood samples for glucose and insulin analyses and for DNA isolation. All required analyses will be completed in 4 months. Aliquots of DNA from both study areas were sent to the PI's laboratory in UNC for genotyping.
The following is a brief description of results for each of the areas included in the population-based study and for laboratory research covered by Specific Aim 4.
a. Diagnosis of skin lesions: 27 percent of the study subjects were diagnosed with one or more types of skin lesions (Table 1).
|
Skin lesion
|
% subjects
|
|
Hypopigmentation
|
9.4
|
|
Hyperpigmentation
|
4.7
|
|
Hypo-hyperpigmentation
|
7.4
|
|
Hyperkeratosis palmar
|
19.6
|
|
Hyperkeratosis plantar
|
23.4
|
|
Skin cancer
|
2.1
|
|
Without skin lesion
|
73
|
Table 1: Skin lesions diagnosed in study subjects from the Zimapan area.
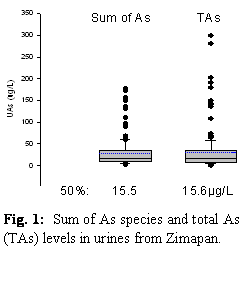
b. Analyses of total and speciated As in urine were completed for all subjects. The total As levels determined by hydride generation (HG)-atomic fluorescence spectrometry were in good agreement with the sum of As species determined by HG-atomic absorption spectrometry (AAS) (Fig. 1). These results suggest that most or all As in urine was associated with exposures to inorganic As.
c. Markers of diabetes: 8 percent of study subjects had elevated levels of glycated hemoglobin (HbA1c > 7%); 16.7 percent of subjects had fasting blood glucose (FBG) between 100 and 125 mg/dl; 4.7 percent had FBG > 126 mg/dl; 6.4 percent of subjects had 2HBG > 180 mg/dl. Fasting plasma insulin analyses were completed; evaluation is pending.
d. AST/ALT tests were completed; AST and ALT activities in plasma from all subjects were generally too low to suggest liver damage.
e. Exfoliated bladder epithelial cells (BEC) were collected from all 149 subjects in this area and TGF-α analysis in BEC was completed; evaluation is pending.
Lagunera Area:
a. Diagnosis of skin lesions: All of the participants in this study area were examined for skin lesions; evaluation is pending.
b. Analyses of total and speciated As: Analyses of total As and trivalent As species in urine were completed; pentavalent As species are currently being analyzed.
c. Markers of diabetes: Analyses were completed for HbA1c, FBG, and 2HBG levels; evaluation is pending.
d. Analyses to be completed: Insulin in fasting plasma and TGF-α in BEC.
Genotyping analysis:
Analysis of common functional genotypes for as3mt started in May 2008, using the TaqMan assay and custom-synthesized probes. A variable number of tandem repeats (VNTR) will be analyzed after PCR amplification of the corresponding 5’-UTR region by sequencing or gel electrophoresis.
Examination of genotype-phenotype relationship:
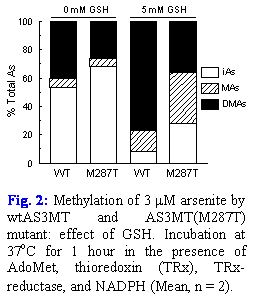
The main goal of this part of the project is to examine As-methylation activities of wild-type (wt) AS3MT and of common AS3MT variants. The kinetics of iAs methylation by recombinant AS3MT(M287T) mutant and wtAS3MT have recently been examined in collaboration with Dr. Thomas’s laboratory (EPA). Results show that in vitro methylation of iAsIII by recombinant AS3MT(M287T) in the presence of physiological concentrations of glutathione (GSH) yields significantly more methyl-As (MAs) and less dimethyl-As (DMAs) than methylation by wtAS3MT (Fig. 2). At this time, we do not know if MAs produced by AS3MT(M287T) is MAsV or highly toxic MAsIII. The oxidation state-specific analysis of iAs metabolites produced by recombinant AS3MT(M287T) and by other common AS3MT polymorphs will be carried out in the near future.
Other Results:
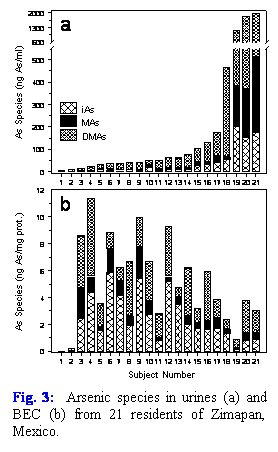
The original design for the population-based study in Mexico included an examination of the relationship between TGF-α levels in BEC and the concentration of As metabolites in the urine of subjects exposed to inorganic As. We have extended this design to include a pilot study of As metabolites in BEC that originate in the urinary bladder epithelium, one of the targets for carcinogenic effects of chronic exposures to inorganic As. Here, As was speciated by an optimized HG-AAS in BEC and in urine isolated from 21 study subjects in Zimapan. Inorganic As and its methylated metabolites, MAs and DMAs, were found in both urines and BEC (Fig. 3). Notably, we found no statistically significant correlations between the total As or As species in urine and BEC. Thus, speciation of As in BEC may provide a better marker of the risk of developing bladder cancer as a result of chronic exposure to As. Results of this work have been submitted for publication in Environmental Health Perspectives (see the list of publications below).
Future Activities:
1. All of the remaining analyses of urine, blood, and plasma will be completed in the next 4 months and all of the results will be provided to the PI’s team for preliminary analyses.
2. Genotyping will start in May 2008 and will be completed by September 2008.
3. The final data analyses will be carried out by the PI, Dr. Styblo, and Co-PIs, Drs. Del Razo and Garcia-Vargas in collaboration with Drs. Lange (biostatistician) and Loomis (epidemiologist).
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 19 publications | 9 publications in selected types | All 9 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Hernandez-Zavala A, Valenzuela OL, Matousek T, Drobna Z, Dedina J, Garcia-Vargas GG, Thomas DJ, Del Razo LM, Styblo M. Speciation of arsenic in exfoliated urinary bladder epithelial cells from individuals exposed to arsenic in drinking water. Environmental Health Perspectives 2008;116(12):1656-1660. |
R832735 (2007) R832735 (2008) R832735 (Final) |
Exit Exit |
|
|
Hernandez-Zavala A, Matousek T, Drobna Z, Paul DS, Walton F, Adair BM, Dedina J, Thomas DJ, Styblo M. Speciation analysis of arsenic in biological matrices by automated hydride generation-cryotrapping-atomic absorption spectrometry with multiple microflame quartz tube atomizer (multiatomizer). Journal of Analytical Atomic Spectrometry 2008;23(3):342-351. |
R832735 (2006) R832735 (2007) R832735 (2008) R832735 (Final) |
Exit |
|
|
Matousek T, Hernandez-Zavala A, Svoboda M, Langrova L, Adair BM, Drobna Z, Thomas DJ, Styblo M, Dedina J. Oxidation state specific generation of arsines from methylated arsenicals based on L-cysteine treatment in buffered media for speciation analysis by hydride generation-automated cryotrapping-gas chromatography-atomic absorption spectrometry with the multiatomizer. Spectrochimica Acta Part B: Atomic Spectroscopy 2008;63(3):396-406. |
R832735 (2007) R832735 (Final) |
Exit |
Supplemental Keywords:
RFA, Scientific Discipline, Water, POLLUTANTS/TOXICS, Health Risk Assessment, Arsenic, Biochemistry, Drinking Water, Genetics, Water Pollutants, bioavailability, health effects, cancer, drinking water contaminants, drinking water monitoring, genotype distributionProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
