Grantee Research Project Results
Final Report: Center for the Study of Childhood Asthma in the Urban Environment (CCAUE)
EPA Grant Number: R836152Center: Center for the Study of Childhood Asthma in the Urban Environment
Center Director: Hansel, Nadia
Title: Center for the Study of Childhood Asthma in the Urban Environment (CCAUE)
Investigators: Hansel, Nadia , Diette, Greg , McCormack, Meredith , Koehler, Kirsten , Polotsky, Seva
Institution: The Johns Hopkins University
EPA Project Officer: Callan, Richard
Project Period: September 1, 2015 through August 31, 2019 (Extended to August 31, 2021)
Project Amount: $1,200,000
RFA: Children's Environmental Health and Disease Prevention Research Centers (2014) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
Project 1:
Through both an observational and intervention study, this project aims to identify why obese children with asthma have increased susceptibility to air pollution compared to lean counterparts. We will examine the leading candidate mediators of the increased susceptibility to PM among overweight children, with asthma who are more susceptible to indoor particulate matter compared to normal weight children with asthma using an experimental study design with an air purifier intervention that targets indoor PM reduction.
Specific aims of the study include:
- To determine if overweight inner-city children, compared to lean inner-city children, have greater improvement in asthma with an air purifier intervention aimed at reducing indoor PM
- To determine whether increases in tidal volume and thereby increases in doses of inhaled particles mediate increased susceptibility to indoor PM among overweight versus lean children with asthma
- To determine whether increases in inflammatory and oxidative stress responses mediate increased susceptibility to indoor PM among overweight versus lean children with asthma.
- To determine whether increases in glucocorticoid resistance mediate increased susceptibility to indoor PM among overweight versus lean children with asthma.
- To determine whether differences in sleep disordered breathing mediate increased susceptibility to indoor PM among overweight versus lean children with asthma.
Project 2
The goal of this project is to determine which potentially modifiable factors of fine particulate matter (PM2.5), including ultrafine particles, micro-environmental and peak exposures that are associated with asthma symptoms in children. A second, related objective is to evaluate the effects of these fine PM factors on overweight children with asthma. Together, such evidence will allow us to build individualized environmental intervention strategies that target this susceptible population.
Specific Aim 1: Evaluate the relationship between ultrafine particles (UFP) and fine particles and asthma symptoms, lung function, and inflammatory biomarkers among asthmatic children. A backpack containing air pollution monitoring equipment for ultrafine particle number concentration (PNC) and fine particulates (PM2.5 mass) and a GPS receiver will be provided to each child for this repeated-measures panel study. Concurrent with the personal exposure assessment, we will collect information on participant symptoms, lung function, urinary leukotriene E4 (uLTE4) and exhaled NO daily.
Specific Aim 2: Evaluate the contribution of peak exposure to cumulative exposure and estimate its effects in inner-city children with asthma. We will calculate the contribution of peak exposures (PNC and PM2.5 mass) to 24-hour cumulative exposure. We will ask children and parents about activities during each 24-hour period to assess the role of sources on peak exposures (e.g. cooking).
Specific Aim 3: Evaluate the role of weight as a susceptibility factor for the impact of UFP and peak PM exposure on asthma outcomes. Approximately half the children in this study will have BMI > 85th percentile and half will be lean.
Project 3
Studies in animal models (Project 3) have advanced our understanding of obesity as a driver of asthma morbidity. Experiments in C57BL/6J mice showed that diet-induced obesity leads to low grade pulmonary inflammation with overexpression of interleukin 1β and airway hyperresponsiveness, which was attenuated after caloric restriction and the IL-1β receptor blockade.
Summary/Accomplishments (Outputs/Outcomes):
Project 1
As of April 30th, 2021, Project 1 contacted 1478 potential participants, scheduled 380 clinic visit 1 (screening visits), 197 children enrolled (consented), completed 162 clinic visit 2 (randomized pre-intervention), completed 148 clinic visit 3 (post-intervention), and completed 135 sleep visits.
Project 1 completed the collection of clinical data, respiratory physiologic data, and biomarkers. Biomarkers include complete blood count with differential, hemoglobin A1C, peripheral blood mononuclear cells, serum stored for cytokines, oxidative stress markers, fatty acids from the blood, nasal epithelial lining fluid, urine, skin, oral and nasal sampling for microbiota. Home environmental sampling was completed for most participants at two one-week periods, which excludes the last participants due to COVID-19 restrictions. Indoor dust samples were analyzed for allergens.
Additionally, the AIRWEIGHS Study received additional funding through the Bloomberg American Health Initiative Launchpad grant to study the impact of COVID 19 and COVID 19 pandemic conditions on study participants. Approximately 70 participants were followed for 12 additional weeks during 2020-2021, where data was collected on asthma symptoms, school experience, COVID-19 symptoms, and psychosocial factors within the family unit.
The AIRWEIGHS study supported the career development of early investigators with projects nested within Project 1. Early investigators included:
- Dr. Nima AfsharMojaher was a Postdoctoral Fellow who led a paper with other trainees (Wu, Brigham) entitled “Obesity, tidal volume, and pulmonary deposition of fine particulate matter in children with asthma” which was published in the European Respiratory Journal in 2021.
- Dr. Emily Brigham has received a K23 application with NIEHS and her project was embedded in the AIRWEIGHS study. She proposed to study the effect of ultrafine particles in the participant homes and modifying effects of diet, obesity, and metabolism.
- Dr. Parisa Kaviany was a pediatric pulmonology fellow from July 2018 through June 2021 and joined faculty at DC Children's. During her fellowship received the Bauernschmidt Research Fellowship Award for her work in the AIRWEIGHS study.
- Amanda McCormack is a PhD candidate who is working with Dr. Meghan Davis. She is completing her thesis work using data from AIRWEIGHS to investigate biologic impacts of Staphylococcus Aureus exposure on children with asthma. In 2021, she was awarded the Fisher Center Discovery Award as PI to study immunologic markers associated with environmental exposure to enterotoxinproducing staphylococcus aureus.
- Dr. Lesliam Quiros Alcala has received an NHLBI K01 that investigated the effect of parabens and troclosan in personal care products on respiratory health among children with asthma.
- Dr. Vikram Tejwani was a postdoctoral fellow who received a CHEST Foundation Research Grant in Severe Asthma, “Peripheral Immune Cell Landscape in Obese Children with Severe Asthma” in 2019 with a project embedded in the AIRWEIGHS study.
- Dr. David Wu was a postdoctoral fellow who received an F32 NRSA to study the effects of secondhand smoke on childhood asthma.
The AIRWEIGHS study provided support for the training grants listed above. In addition, there have been several awards that build on the findings from AIRWEIGHS and the Obese Asthma Program.
The AIRWEIGHS study has provided preliminary data for the NIH R01 HL 152419 (PI Dr. McCormack) to study the effect of environmental exposures and sleep in Baltimore City children with asthma.
The AIRWEIGHS Study has provided preliminary data for an NIH UG3 HL154273 (PI Eakin and McCormack) to study the implementation of a multi-level school-based asthma intervention.
The AIRWEIGHS Study received additional funding through the Bloomberg American Health Initiative Launchpad grant to study the impact of COVID 19 and COVID 19 pandemic conditions on study participants. Approximately 70 participants were followed for 12 additional weeks during 2020-2021, where data was collected on asthma symptoms, school experience, COVID-19 symptoms, and psychosocial factors within the family unit.
AIRWEIGHS participant characteristics are presented in Table 1. Of note, we had initially excluded children who were underweight. However, in reviewing characteristics of children who were screen failing due to this reason, we found that this often was in the context of being otherwise healthy and sometimes recent growth spurts played a role in this finding. We therefore changed our inclusion criteria to include underweight children who did not have any chronic medical problems that would otherwise exclude them.
Table 1
| Age (years) | 11.00 ± 2.44 | 197 |
| Female | 82 (41.6%) | 197 |
| Black/African American | 164 (83.2%) | 197 |
| Caregiver Education (some college) | 84 (42.6%) | 197 |
| Household Income |
| 197 |
| less than $15,000 | 52 (26.4%) |
|
| $15,000 - $34,999 | 67 (34.0%) |
|
| $35,000 - $49,999 | 25 (12.7%) |
|
| $50,000 or more | 24 (12.2%) |
|
| Refuse/Don't Know | 29 (14.7%) |
|
| Insurance |
| 197 |
| Private | 28 (14.2%) |
|
| Public | 167 (84.8%) |
|
| Self-pay (no insurance) | 2 (1.0%) |
|
| Pulmonary Function/Breathing Test (in liters) |
| 189 |
| Pre FEV1-best | 2.10 ± 0.69 |
|
| Pre FVC-best | 2.59 ± 0.88 |
|
| Pre FEV1 % predicted | 94.84 ± 15.80 |
|
| Post FEV1 best | 2.28 ± 0.73 |
|
| Post FVC best | 2.69 ± 0.88 |
|
| Post FEV1 % predicted | 102.86 ± 14.27 |
|
| BMI Categories |
| 164 |
| Underweight | 2 (1.2%) |
|
| Normal | 59 (36.0%) |
|
| Overweight | 25 (15.2%) |
|
| Obese | 78 (47.6%) |
|
| ATAQ | 2.45 ± 1.94 | 161 |
| Indoor Environmental Monitoring |
|
|
| PM2.5 | 23.96 ± 26.44 | 158 |
| PM10 | 33.00 ± 32.18 | 159 |
| NO2 | 16.71 ± 17.77 | 162 |
| Air Nicotine | 0.67 ± 2.07 | 160 |
| AHI (Apnea Hypopnea Index) | 4.99 ± 12.60 | 135 |
| Data as of October 8, 2021 |
Project 2
Collaborations and Developing Materials: We continue to closely collaborate with the Project 1 (AIRWEIGHS: Investigating Obesity as a Susceptibility Factor for Air Pollution in Childhood Asthma) study team in different aspects of the project. Bimonthly center meetings are held where all the lead investigators as well as the research coordinators and field and clinic staff for from both PEAK and AIRWEIGHS projects take part. One of the outcomes of this close collaboration with the AIRWEIGHS team has been the development and review of the necessary Standards of Practice (SOPs) to be used in the PEAK study. All SOPs are complete and being followed by all field and laboratory staff. Study questionnaires have also been developed for the participating children and parents, which will ask about asthma health, medications, diet, and physical activity as part of their enrollment in the study. Questionnaires have also been developed to ask about daily exposures and activities that may contribute to asthma health. These questionnaires have been used throughout the study.
Approvals: We have obtained IRB approval and renewal for the PEAK study.
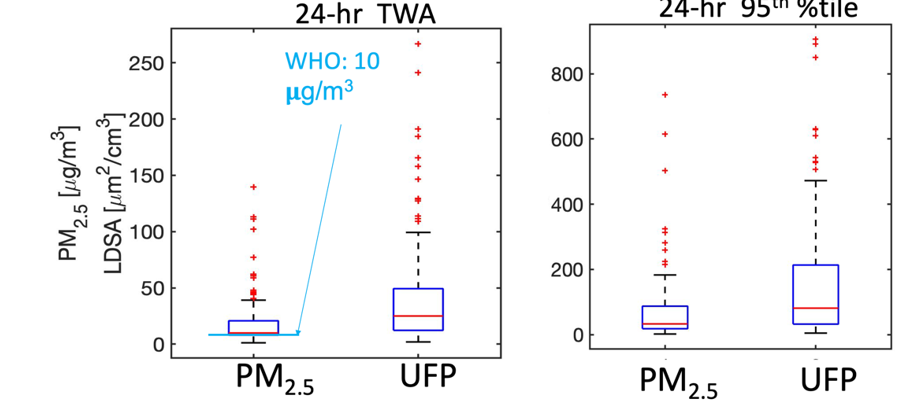
Results to Date: Last year we completed enrollment of all 50 children with a targeted four days of sampling per child for the PEAK study. Enrollment and data collection is complete. We also conducted a pilot study at the beginning of the study in which we enrolled 5 children to examine the feasibility of the backpack sampling and obtaining symptom data via text message. They were asked to fill out a set of questionnaires including daily activity and asthma symptoms, their experience with carrying the backpack and being involved in a research project. This feedback was reviewed and taken into account for main PEAK study. This year we have focused on continuing data quality assurance and analysis. We developed an interactive software to use the GPS, accelerometry, and light data to assign time periods of exposure to different microenviornments. Preliminarily, our analyses focus on the 50 children from the main study who had a mean age of 11 with a range from 8-17 years. Forty-six percent of the children were female and 80% were Black race. Our preliminary analyses show that the children were exposed to levels of PM mass often exceeding WHO recommendations for PM2.5 in indoor air (24-hr mean (N=154): 19.5 μg/m3; range: 3.1-178.8 μg/m3; see Figure 1). Ultrafine particle (UFP) exposures (expressed as lung deposited surface area [LDSA], 24-hr mean (N=152): 41 μm2/cm3; range: 4-262 μm2/cm3) typically varied over 2 orders of magnitude within 24-hours and showed relatively low correlation with PM2.5 (median Pearson coefficient of 1-minute measurements across 134 sampling days with valid PM2.5 and UFP data was 0.61 with a range of -0.2 to 0.99). Exposures to fine and ultrafine particles tended to be highest at home (mean PM2.5 was 24 μg/m3 and mean LDSA was 53 μm2/cm3) during exposure to secondhand smoke and cooking events. Exposures in school were the lowest, on average (PM2.5= 6.1 μg/m3; LDSA=12 μm2/cm3) and were somewhat higher in transit (PM2.5= 10.4 μg/m3; LDSA= 21 μm2/cm3). Daily averaged exposures to PM2.5 and UFP were more highly correlated, with a Pearson coefficient of 0.70.
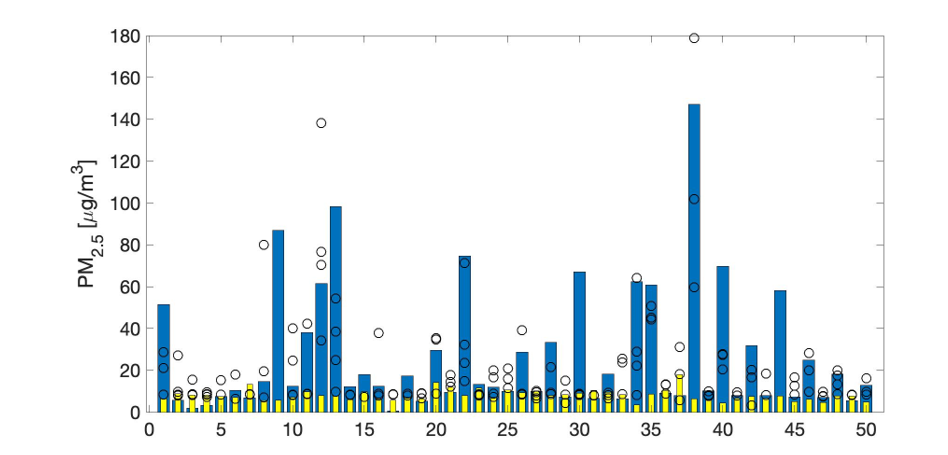
We compared personal exposures from children wearing the backpack in PEAK (circles in Figure 2; participants in PEAK are a subset of participants in AIRWEIGHS) with weekly-average home PM2.5 concentrations measured in the AIRWEIGHS study (baseline only; blue bars) and weekly-average ambient PM2.5 measured by the Maryland Department of the Environment centrally in Baltimore city (yellow bars). We found that for most participants, home exposures greatly exceeded ambient concentrations, and in some cases personal exposures were even higher.
We have also deployed a text messaging interface for children or parents to provide information on child symptoms twice daily. This method has been very successful with over 85% completion of symptom information returned by text message to study staff. These children were symptomatic, with cough, wheeze, rescue inhaler use, or awakened with symptoms reported on 45%, 21%, 20%, and 18% of sampling days, respectively. All urine samples have been sent for uLTE4 analysis and show both within- and between- child variability in biomarker levels. Epidemiologic analyses are ongoing.
Figure 1: Boxplots of daily average exposures and 95th percentile exposures to PM2.5 and UFP (expressed as lung deposited surface area, LDSA).
Figure 2: Comparison of personal (circles), weekly-average home concentrations (blue bars) and weekly-average ambient concentrations (yellow bars) of PM2.5 for 50 children (numbered on x-axis) in PEAK.
Project 3
Specific Aim 1
Experiments performed during Years 1-4 of the award have demonstrated that diet-induced obesity (DIO) induced AHR in association with low grade inflammation in lung tissue and, specifically, up-regulation of interleukin 1 β (IL-1β) expression. Experiments of Year 4 examined the role of IL-1β in DIO-induced AHR.
Our results showed that the IL-1β receptor blockade prevented both obesity-induced AHR and pulmonary inflammation, supporting the concept that up-regulation of IL-1β gene expression in the lung could be a mechanism linking obesity and asthma. Given that obesity increases AHR early in the time course and that anakinra was administered only during last two weeks of the 8-week experiment, our data suggest that IL-1β receptor blockers not merely prevent, but also reverse obese asthma. In our study, anakinra lowered fasting serum insulin levels without a significant change in fasting glucose levels indicating increased insulin sensitivity. Our finding suggests that anakinra could contribute to improvement in AHR due to its off-target effects by improving glucose metabolism.
Hypothesis: Lowering insulin resistance in mice DIO will have an independent effect on AHR
Methods: C57BL/6J mice were fed with a high fat diet (HFD) ad libitum for 8 weeks and treated with metformine at 300 mg/mouse/day added receptor blocker (anakinra) or placebo during the last 2 weeks of the experiment (n = 10 per group). Upon completion of the experiment, mice were anesthetized with ketamine/xylazine i.p., tracheostomized and the total respiratory resistance (Rrs) was measured by forced oscillation (Flexivent, SCIREQ Québec, Canada). at baseline and after methacholine aerosol challenge at 3 and 30 mg/mL.
Results: Mice in the Meformin and Control groups were of the age and body weight. They consumed the same amount of water and food. Metformin did not affect fasting blood glucose levels. Plasma fasting insulin, leptin and adiponectin levels are pending. Metformin did not affect Rrs at baseline and at 3 mg/ml of methacholine, but there was a nearly two-fold decrease in total respiratory system resistance at 30 mg/ml of methacholine. Surprisingly, metformin increased cellularity of the bronchoalveolar lavage fluid. There was an increase in the percentage of lymphocytes with a corresponding decrease in the percentage of macrophages. There was no evidence of systemic inflammation on CBC (Table 3). IL-1β, TNF-α, IL-6 in the lungs and plasma cytokine levels are pending.
Conclusions: Metformin significantly decreased the airway response to methacholine, which may suggest the role of insulin resistance in increased airway reactivity in obese asthma.
Specific Aim 2.
During Years 1-3 of the award we have developed a mouse model of sleep disordered breathing by expressing inhibitory designer receptor exclusively activated by designer drugs (DREADD) in hypoglossal motoneurons 3. During Year 4 we examine the effect of sleep disordered breathing on AHR. Methods: DIO C57BL/6J mice (n = 18) were treated with an inhibitory DREADD harbored by a retrograde adeno-associated virus, AAV9-HA- hM4D-mCherry (Gi). AAV9-DREADD was administered to the genioglossal (GG) muscle of the tongue bilaterally (7 x 1010 vg/10 μl in total). Four weeks after viral infection mice (n = 9) underwent daily injection of a novel DREADD ligand (JHU37160 dihydrochloride, 0.1mg/kg in 250 µl saline i.p.) vs saline (n = 9) for 2 weeks. Upon completion of treatment, GG EMG was recorded followed by sleep study and AHR measurements.
Results: J60 injections did not affect mouse body weight, food intake or blood glucose level.
There was a significant decrease of GG muscle EMG activity, which strongly suggests the development of sleep apnea (as we have demonstrated previously. However, J60 administration was not associated with an increase in AHR.
Conclusion: Obstruction of upper airway in our mouse model of sleep disordered breathing was not associated with AHR. This data suggests that OSA does not cause or exacerbate asthma. The application was focused on obesity-induced non-eosinophilic model of AHR and sleep apnea in diet-induced C57BL/6J. Unfortunately this model appeared to be resistant to PM. We have attempted multiple models of PM exposure including nasal administration, intratracheal administration, exposure in sealed chambers, nebulization, etc. with varying PM2.5 concentration up to very high (New Delhi ) – none of them increased AHR in obese C57BL/6J mice. Mouse strains susceptible to PM (Balb/c for instance) are resistant to diet-induced obesity.
Journal Articles: 49 Displayed | Download in RIS Format
| Other center views: | All 74 publications | 49 publications in selected types | All 49 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Belli AJ, Bose S, Aggarwal N, DaSilva C, Thapa S, Grammer L, Paulin LM, Hansel NN. Indoor particulate matter exposure is associated with increased black carbon content in airway macrophages of former smokers with COPD. Environmental Research 2016;150:398-402. |
R836152 (2019) R836152 (2020) R836150 (2017) R836150 (2019) R836150 (2020) |
Exit |
|
|
Berger S, Pho H, Fleury-Curado T, Bevans-Fonti S, Younas H, Shin M, Jun J, Anokye-Danso F, ahima R, Enquist L, Mendelowitz D, Schwartz A, Polotsky V. Intranasal Leptin Relieves Sleep-disordered Breathing in Mice with Diet-induced Obesity. AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE 2019;199(6):773-783. |
R836152 (Final) R834510 (Final) |
Exit Exit |
|
|
Berman JD, McCormack MC, Koehler KA, Connolly F, Clemons-Erby D, Davis MF, Gummerson C, Leaf PJ, Jones TD, Curriero FC. School environmental conditions and links to academic performance and absenteeism in urban, mid-Atlantic public schools. International Journal of Hygiene and Environmental Health 2018;221(5):800-808. |
R836152 (Final) R835639 (2016) R835639 (2017) R835639 (2018) |
Exit Exit Exit |
|
|
Bhavnani D, Lilley T, Rathouz P, Beaudenon-Huibregste S, Davis M, Mccormack M, Keet C, Balcer-Whaley S, Newman M, Matsui E. Indoor allergen exposure and its association to upper respiratory infections and pulmonary outcomes among children with asthma. JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY 2024;154(6):1434-1441 |
R836152 (Final) |
Exit |
|
|
Brigham EP, Matsui EC, Appel LJ, Bull DA, Curtin-Brosnan J, Zhai S, White K, Charleston JB, Hansel NN, Diette GB, McCormack MC. A pilot feeding study for adults with asthma: the healthy eating better breathing trial. PLoS One 2017;12(7):e0180068 (14 pp.). |
R836152 (2018) R836152 (2020) |
Exit Exit Exit |
|
|
Brigham EP, Steffen LM, London SJ, Boyce D, Diette GB, Hansel NN, Rice J, McCormack MC. Diet pattern and respiratory morbidity in the Atherosclerosis Risk in Communities Study. Annals of the American Thoracic Society 2018;15(6):675-682. |
R836152 (2018) R836152 (2020) |
Exit Exit |
|
|
Brigham E, Woo H, McCormack M, Rice J, Koehler K, Vulcain T, Wu T, Koch A, Sharma S, Kolandooz F, Bose S, Hanson C, Romero K, Diette G, Hansel N. Omega-3 and Omega-6 Intake Modifies Asthma Severity and Response to Indoor Air Pollution in Children. AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE 2019;199(12):1478-1486. |
R836152 (Final) R834510 (Final) |
Exit Exit |
|
|
Caballero-Eraso C, Shin M, Pho H, Kim L, Pichard L, Wu Z, Gu C, Berger S, Pham L, Yeung H, Shirahata M, Schwartz A, Tang W, Sham J, Polotsky V. Leptin acts in the carotid bodies to increase minute ventilation during wakefulness and sleep and augment the hypoxic ventilatory response. JOURNAL OF PHYSIOLOGY-LONDON 2019;597(1):151-172. |
R836152 (Final) R834510 (Final) |
Exit Exit |
|
|
Davis MF, Ludwig S, Brigham EP, McCormack MC, Matsui EC. Effect of home exposure to Staphylococcus aureus on asthma in adolescents. Journal of Allergy and Clinical Immunology 2018;141(1):402-405.e10. |
R836152 (2018) R836152 (2020) R836152 (Final) R834510 (Final) |
Exit Exit Exit |
|
|
Fleury Curado T, Fishbein K, Pho H, Brennick M, Dergacheva O, Sennes LU, Pham LV, Ladenheim EE, Spencer R, Mendelowitz D, Schwartz AR, Polotsky VY. Chemogenetic stimulation of the hypoglossal neurons improves upper airway patency. Scientific Reports 2017;7:44392. |
R836152 (2017) R836152 (2020) |
Exit Exit Exit |
|
|
Fleury Curado T, Pho H, Berger S, Caballero-Eraso C, Shin MK, Sennes LU, Pham L, Schwartz AR, Polotsky VY. Sleep-disordered breathing in C57BL/6J mice with diet-induced obesity. Sleep 2018;41(8).zsy089 (9 pp.). |
R836152 (2018) R836152 (2020) |
Exit Exit |
|
|
Fricke K, Vieira M, Younas H, Shin MK, Bevans-Fonti S, Berger S, Lee R, D’Alessio FR, Zhong Q, Nelson A, Loube J. Sanchez I, Hansel NN, Mitzner W, Polotsky VY. High fat diet induces airway hyperresponsiveness in mice. Scientific Reports 2018;8(1):6404 (6 pp.). |
R836152 (2018) R836152 (2020) R834510 (Final) |
Exit Exit Exit |
|
|
Galiatsatos P, Kineza C, Hwang S, Pietri J, Brigham E, Putcha N, Rand CS, McCormack M, Hansel NN. Neighbourhood characteristics and health outcomes:evaluating the association between socioeconomic status, tobacco store density and health outcomes in Baltimore City. Tobacco Control 2018;27(e1):e19-e24. |
R836152 (2018) R836152 (2019) R836152 (2020) R836150 (2018) R836150 (2020) |
Exit |
|
|
Grant T, McCormack M, Peng R, Keet C, Davis M, Newman M, Balcer-Whaley S, Matsui E. Comprehensive home environmental intervention did not reduce allergen concentrations or controller medication requirements among children in Baltimore. JOURNAL OF ASTHMA 2022;1-10 |
R836152 (Final) |
Exit |
|
|
Grant T, LIlley T, McCormack M, Rathouz P, Pent R, Keeo C, Rule A, Davis M, Balcer-Whaley S, Newman M, Matsui E. Indoor environmental exposures and obstructive lung disease phenotypes among children with asthma living in poor urban neighborhoods. JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY 2023;151(3):716. |
R836152 (Final) |
Exit |
|
|
Gu C, Loube J, Lee R, Bevans-Fonti S, Wu T, Barmine J, Jun J, McCormack M, Hansel N, Mitzner W, Polotsky V. Metformin Alleviates Airway Hyperresponsiveness in a Moe Model of Diet-Induced Obesity. FRONTIERS IN PHYSIOLOGY 2022;13(883275). |
R836152 (Final) R834510 (Final) |
Exit |
|
|
Javaheri S, Barbe F, Campos-Rodriguez F, Dempsey JA, Khayat R, Javaheri S, Malhotra A, Martinez-Garcia MA, Mehra R, Pack AI, Polotsky VY, Redline S, Somers VK. Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. Journal of the American College of Cardiology 2017;69(7):841-858. |
R836152 (2017) R836152 (2020) |
Exit Exit Exit |
|
|
Kaviany P, Brighham E, Collaco J, Rice J, Woo H, Wood M, Koehl R, Wu T, Eakin M, koehler K, Hansel N, McCormack M. Patterns and predictors of air purifier adherence in children with asthma living in low-income, urban Households. JOURNAL OF ASTHMA 2022;59(5):946-955 |
R836152 (Final) |
Exit |
|
|
Lambert AA, Putcha N, Drummond MB, Boriek AM, Hanania NA, Kim V, Kinney GL, McDonald MN, Brigham EP, Wise RA, McCormack MC, Hansel NN, COPDGene Investigators. Obesity is associated with increased morbidity in moderate to severe COPD. Chest 2017;151(1):68-77. |
R836152 (2019) R836152 (2020) R836150 (2017) R836150 (2019) R836150 (2020) |
Exit |
|
|
Lemoine S, Brigham E, Woo H, Hanson C, McCormack M, Koch A, Putcha N, hansel N. Omega-3 fatty acid intake and prevalent respiratory symptoms among adults with COPD. BMC PULMONARY MEDICINE 2019;19. |
R836152 (Final) R834510 (Final) |
Exit Exit |
|
|
Levy JI, Quiros-Alcala L, Fabian MP, Basra K, Hansel NN. Established and emerging environmental contributors to disparities in asthma and chronic obstructive pulmonary disease. Current Epidemiology Reports 2018;5(2):114-124. |
R836152 (2018) R836152 (2019) R836152 (2020) R836150 (2019) R836150 (2020) R836156 (2018) R836156 (2019) R836156 (2020) |
Exit |
|
|
Lu KD, Phipatanakul W, Perzanowski MS, Balcer-Whaley S, Matsui EC. Atopy, but not obesity is associated with asthma severity among children with persistent asthma. Journal of Asthma 2016;53(10):1033-1044. |
R836152 (2017) R836152 (2020) |
Exit Exit |
|
|
McCormack MC, Belli AJ, Waugh D, Matsui EC, Peng RD, Williams DL, Paulin L, Saha A, Aloe CM, Diette GB, Breysse PN, Hansel NN. Respiratory effects of indoor heat and the interaction with air pollution in chronic obstructive pulmonary disease. Annals of the American Thoracic Society 2016;13(12):2125-2131. |
R836152 (2018) R836152 (2019) R836152 (2020) R836150 (2019) R836150 (2020) |
Exit Exit Exit |
|
|
McCormack MC, Paulin LM, Gummerson CE, Peng RD, Diette GB, Hansel NN. Colder temperature is associated with increased COPD morbidity. European Respiratory Journal 2017;49(6):1601501. |
R836152 (2019) R836152 (2020) R836150 (2018) R836150 (2019) R836150 (2020) |
Exit |
|
|
McCormack M, Balsubramanian A, Wise R, Keet C, Matsui E, Peng R. Reply by McCormack et al. to Townsend and Cowl, and to Miller et al. AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE 2022;206(6):795-796. |
R836152 (Final) |
Exit Exit |
|
|
Mesarwi OA, Shin MK, Bevans-Fonti S, Schlesinger C, Shaw J, Polotsky VY. Hepatocyte hypoxia inducible factor-1 mediates the development of liver fibrosis in a mouse model of nonalcoholic fatty liver disease. PLoS One 2016;11(12):e0168572. |
R836152 (2017) R836152 (2020) |
Exit Exit Exit |
|
|
Nnodum BN, McCormack MC, Putcha N, Hwang S, Paulin LM, Brigham EP, Fawzy A, Romero K, Diette GB, Hansel NN. Impact of physical activity on reporting of childhood asthma symptoms. Lung 2017;195(6):693-698. |
R836152 (2018) R836152 (2020) |
Exit Exit Exit |
|
|
Paulin LM, Williams DA, Peng R, Diette GB, McCormack MC, Breysse P, Hansel NN. 24-h Nitrogen dioxide concentration is associated with cooking behaviors and an increase in rescue medication use in children with asthma. Environmental Research 2017;159:118-123. |
R836152 (2018) R836152 (2020) |
Exit Exit Exit |
|
|
Peters KO, Williams AL, Abubaker S, Curtin-Brosnan J, McCormack MC, Peng R, Breysse PN, Matsui EC, Hansel NN, Diette GB, Strickland PT. Predictors of polycyclic aromatic hydrocarbon exposure and internal dose in inner city Baltimore children. Journal of Exposure Science and Environmental Epidemiology 2017;27(3):290-298. |
R836152 (2017) R836152 (2020) |
Exit Exit |
|
|
Pham LV, Polotsky VY. Genome-wide association studies in obstructive sleep apnea. Will we catch a black cat in a dark room? American Journal of Respiratory and Critical Care Medicine 2016;194(7):789-791. |
R836152 (2017) R836152 (2020) |
Exit |
|
|
Pham LV, Miele CH, Schwartz NG, Arias RS, Rattner A, Gilman RH, Miranda JJ, Polotsky VY, Checkley W, Schwartz AR. Cardiometabolic correlates of sleep disordered breathing in Andean highlanders. European Respiratory Journal 2017;49(6):1601705 (20 pp.). |
R836152 (2018) R836152 (2020) |
Exit |
|
|
Pham LV, Meinzen C, Arias RS, Schwartz NG, Rattner A, Miele CH, Smith PL, Schneider H, Miranda JJ, Gilman RH, Polotsky VY, Checkley W, Schwartz AR. Cross-sectional comparison of sleep-disordered breathing in native Peruvian highlanders and lowlanders. High Altitude Medicine & Biology 2017;18(1):11-19. |
R836152 (2018) R836152 (2020) |
Exit Exit |
|
|
Rice JL, Brigham E, Dineen R, Muqueeth S, O'Keefe G, Regenold S, Koehler K, Rule A, McCormack M, Hansel NN, Diette GB. The feasibility of an air purifier and secondhand smoke education intervention in homes of inner city pregnant women and infants living with a smoker. Environmental Research 2018;160:524-530. |
R836152 (2018) R836152 (2020) R834510 (Final) |
Exit Exit Exit |
|
|
Shin M, Eraso C, Mu Y, Gu C, Yeung B, Kim L, Liu X, Wu Z, Paudel O, Prchard L, Shirahata M, Tang W, Sham J, Polotsky V. Leptin Induces Hypertension Acting on Transient Receptor Potential Melastatin 7 Channel in the Carotid Body. CIRCULATION RESEARCH 2019;125(11):989-1002. |
R836152 (Final) R834510 (Final) |
Exit Exit |
|
|
Shin M-K, Han W, Joo H, Bevans-Fonti S, Shiota M, Stefanovski D, Polotsky VY. Effect of adrenal medullectomy on metabolic responses to chronic intermittent hypoxia in the frequently sampled intravenous glucose tolerance test. Journal of Applied Physiology 2017;122(4):767-774. |
R836152 (2017) R836152 (2020) |
Exit Exit |
|
|
Stauber CE, Ortiz GM, Loomis DP, Sobsey MD. A randomized controlled trial of the concrete biosand filter and its impact on diarrheal disease in Bonao, Dominican Republic. American Journal of Tropical Medicine and Hygiene 2009;80(2):286-293. |
R836152 (2017) R836152 (2020) SU832463 (Final) |
Exit Exit Exit |
|
|
Tejwani V, McCormack A, Suresh K, Woo H, Xu N, Davis M, Brigham E, Hansel N, McCormack M, D'Alessio F. Dexamethasone-Induced FKBP51 Expression in CD4+ T-Lymphocytes Is Uniquely Associated With Worse Asthma Control in Obese Children With Asthma. FRONTIERS IN IMMUNOLOGY 2021;12(74482) |
R836152 (Final) |
Exit |
|
|
Wu TD, Brigham EP, Peng R, Koehler K, Rand C, Matsui EC, Diette GB, Hansel NN, McCormack MC. Overweight/obesity enhances associations between secondhand smoke exposure and asthma morbidity in children. The Journal of Allergy and Clinical Immunology: In Practice 2018;6(6):2157-2159.e5. |
R836152 (2018) R836152 (2020) |
Exit Exit |
|
|
Wu T, Fawzy A, Brigham E, McCormack M, Rosas I, Villareal D, Hanania N. Association of Triglyceride-Glucose Index and Lung Health A Population-Based Study. CHEST 2021;160(3):1026-1034 |
R836152 (Final) R836150 (2021) |
Exit |
|
|
Wu T, Zaeh S, Eakin M, Koehler K, Davis M, Wohn C, Diibor I, Psoter K, Cronister C, Connolly F, Stein M, McCormack M. Association of School Infrastructure on Health and Achievement Among Children With Asthma. ACADEMIC PEDIATRICS 2023;23(4):814-820. |
R836152 (Final) R835639 (Final) |
Exit |
|
|
Zaeh S, McCormack M, Eakin M. Key policies to support asthma medication management for children. ANNALS OF ALLERGY ASTHMA & IMMUNOLOGY 2019;123(5):428-429 |
R836152 (Final) R835639 (Final) |
|
|
|
Zaeh S, Koehler K, Eakin M, WOhn C, Diibor I, Eckmann T, Wu T, Clemons-Erby D, Gummerson C, Green T, Wood M, Majid E, Stein M, Rule A, Davis M, McCormack M. Indoor Air Quality Prior to and Following School Building Renovation in a Mid-Atlantic School District. INTERNATIONAL JOURNAL OF ENVIRONMENTAL RESEARCH AND PUBLIC HEALTH 2021;18(22). |
R836152 (Final) R835639 (Final) |
Exit Exit |
|
|
Fawzy A, Putcha N, Paulin LM, Aaron CP, Labaki WW, Han MK, Wise RA, Kanner RE, Bowler RP, Barr RG, Hansel NN. Association of thrombocytosis with COPD morbidity:the SPIROMICS and COPDGene cohorts. Respiratory Research 2018;19(1):20. |
R836152 (2019) R836152 (2020) |
|
|
|
Galiatsatos P, Brigham EP, Pietri J, Littleton K, Hwang S, Grant MC, Hansel NN, Chen ES. The effect of community socioeconomic status on sepsis-attributable mortality. Journal of Critical Care 2018;46:129-133. |
R836152 (2019) R836152 (2020) |
|
|
|
Younas H, Vieira M, Gu C, Lee R, Shin MK, Berger S, Loube J, Nelson A, Bevans-Fonti S, Zhong Q, D’Alessio FR. Caloric restriction prevents the development of airway hyperresponsiveness in mice on a high fat diet. Scientific reports 2019;9(1):1-9. |
R836152 (Final) R834510 (Final) R836150 (2020) |
Exit |
|
|
Raju S, Keet CA, Paulin LM, Matsui EC, Peng RD, Hansel NN, McCormack MC. Rural residence and poverty are independent risk factors for chronic obstructive pulmonary disease in the United States. American journal of respiratory and critical care medicine. 2019;199(8):961-969. |
R836152 (2019) R836152 (2020) R836150 (2019) R836150 (2020) |
Exit |
|
|
Tsou PY, McCormack MC, Matsui EC, Peng RD, Diette GB, Hansel NN, Davis MF. The effect of dog allergen exposure on asthma morbidity among inner‐city children with asthma. Pediatric Allergy and Immunology 2020;31(2):210-3. |
R836152 (Final) R832139 (Final) R834510 (Final) R836150 (2020) |
Exit |
|
|
Wu TD, Fawzy A, Kinney GL, Bon J, Neupane M, Tejwani V, Hansel NN, Wise RA, Putcha N, McCormack MC. Metformin use and respiratory outcomes in asthma-COPD overlap. Respiratory research 2021;22(1):1-8. |
R836152 (Final) R836150 (2021) |
Exit |
|
|
Afshar-Mohajer N, Wu TD, Shade R, Brigham E, Woo H, Wood M, Koehl R, Koehler K, Kirkness J, Hansel NN, Ramchandran G. Obesity, tidal volume, and pulmonary deposition of fine particulate matter in children with asthma. European Respiratory Journal 2022;59(3). |
R836152 (Final) R836150 (2021) |
Exit |
Supplemental Keywords:
air pollution, obesity, asthma exacerbation, inner city.Progress and Final Reports:
Original Abstract Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R836152C001 Investigating obesity as a susceptibility factor for air pollution in childhood
R836152C002 Novel exposure metrics for assessing the effects of ultrafine and fine particulate matter on asthma in children
R836152C003 The Role of Obesity in Biological Responses to Particulate Matter in Mice
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- 2020 Progress Report
- 2019 Progress Report
- 2018 Progress Report
- 2017 Progress Report
- 2016 Progress Report
- Original Abstract
49 journal articles for this center