Grantee Research Project Results
2015 Progress Report: Multi-Sensor Reporter Cell Technology to Assess Hazard Involving Endocrine Signaling Pathways
EPA Grant Number: R835165Title: Multi-Sensor Reporter Cell Technology to Assess Hazard Involving Endocrine Signaling Pathways
Investigators: LeBlanc, Gerald A.
Institution: North Carolina State University
EPA Project Officer: Aja, Hayley
Project Period: March 1, 2012 through February 28, 2016 (Extended to February 28, 2017)
Project Period Covered by this Report: March 1, 2015 through February 29,2016
Project Amount: $950,507
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The high-throughput evaluation of toxicity pathways is an emerging paradigm for future chemical toxicity characterizations. The development of assays that evaluate several endpoints along a defined signaling pathway or multiple-pathways within a network could provide insight into: (1) the identification of the most susceptible molecular targets of toxicant action along a cellular pathway/network, (2) the combined actions of different chemicals along a common pathway/network, and (3) multiple actions of a single chemical at different sites within a pathway/network resulting in enhanced toxic outcome. We have developed battery assays incorporating reporter gene transcription assays (Figure 1A) and bioluminescence resonance energy transfer (BRET) technology (Figure 1B) to examine the impact of chemicals on defined cellular signaling pathways. The following pathways were evaluated.
- Human PPARα signaling pathway. This ligand-activated pathway regulates aspects of glucose and lipid metabolism. PPARα activation favors glucose retention and lipid metabolism.
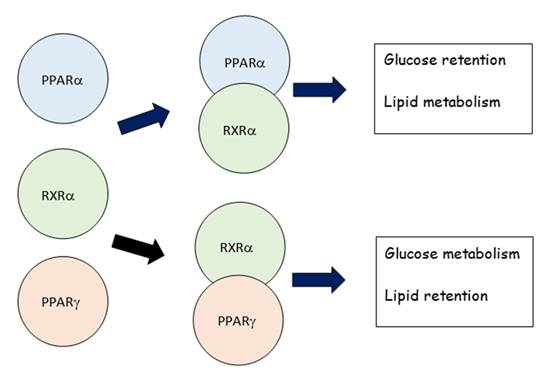
- Human PPARγ signaling pathway. This ligand-activated pathway also regulates aspects of glucose and lipid metabolism. PPARγ activation favors glucose metabolism and lipid retention. Together, the PPARα and PPARγ signaling pathways constitute a major portion of the PPAR signaling network (Figure 2).
- Daphnid MfR signaling pathway. This ligand-activated pathway regulates aspects of development and reproduction in arthropods.
- Daphnid HR3:E75 signaling pathway. This pathway regulates aspects of growth and reproduction in arthropods.
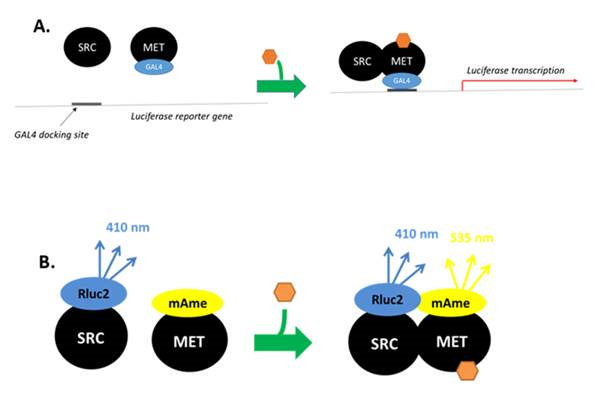
Figure 1. Assays used to assess dimerization and activation of the MfR. A.
Luciferase reporter gene transcription assay depicts transcription of the reporter gene
(luciferase) by agaoist-(⬢) activated SRC:MET. The transcription factor binds to the
reporter gene via a GAL4 docking unit. B. BRET assay depicting agonist-stimulated
demerization of SRC and MET. SRC is fused to the RenillaI luciferase 2 enzyme
metabolizes an added substrate that emits photons at 410 nm. MET is fused to the
flourosphore mAmetrine that exitation at 410 nm, emits photos at 535nm. The ration
of light emitted at 535 and 410 nm is the BRET ration used as a measure of
protine-protein binding.
Figure 2 PPAR signaling network. Agonist-activated PPARα:RXRα regulates aspects of slucose
retention and lipid metabolism.; whereas, agonist-activatd PPAR𝛶:RXRα regulates aspects of
glucose metabolism and lipid retention.
Progress Summary:
- The identification of the most susceptible molecular targets of toxicant action along a cellular pathway/network
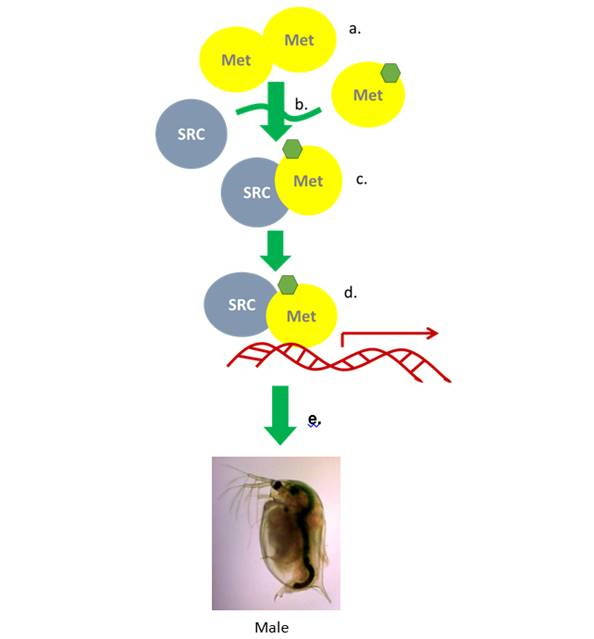
Assays were developed with which we determined that ligand activation of the MET protein, a constituent of the MfR, involves first the dissociation of MET from a MET multimer, followed by association of the MET protein with the SRC protein. These protein association/dissociation interactions were measured using BRET and employing a high-throughput-compatible assay format. Ligand-activated MET, when dimerized with SRC, comprised an active transcription factor which can stimulate transcription of a luciferase reporter gene. A readily discernible consequence of this activation in vivo is the production of male offspring by maternal daphnids exposed to the activating chemical. This pathway and its relevant endpoints are depicted in Figure 3. We evaluated the multiple endpoints in this signaling cascade (MET multimer dissociation, MET-SRC association, reporter gene activation, and in vivo male sex determination) for sensitivity, efficacy, suitability to high-throughput evaluation, along with time and cost considerations. MET-SRC association proved to be the most suitable endpoint for use in high-throughput screening of chemicals for activation or inhibition of the MfR (Figure 4).
Figure 3. MfR endocine signaling pathway MET spontaneously forms a multimer
(a) which dissociates in the presence of agonist (green hexagon) (b). Upon dissociation
the free agonist-bount MET may recruit SRC forming a transcription activator (d). In
daphnids, tis transcription activatin results in male sex determination of offspring (e).
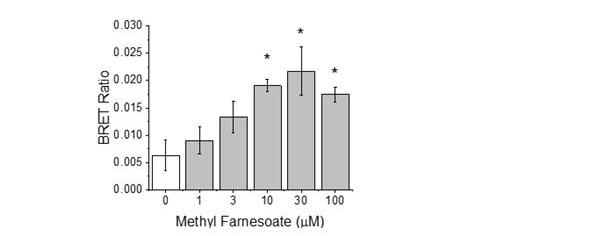
Figure 4. Agonist-stimulated dimerization of the MfR subunits, MET and SRC as
measured by BRET. Agonist used in the assay was the endogenous hormone methyl
farnesoate. Data are presented as the mean and standard deviation (n=3). An asterik
denotes a significant difference from the control (p<0.05).Identity of the DNA element to which the activated MfR binds to regulate gene transcription is unknown. Accordingly, the luciferase gene used in our reporter gene assays are driven by the GAL4 response element and the MET protein is fused to the GAL4 DNA-binding peptide sequence derived from yeast. Presently, we are performing a bioinformatics search of the daphnid genome for the identity of the endogenous MfR response element. We have thus far identified, synthesized, and evaluated 10 putative response elements using electrophoretic mobility shift assays (EMSAs). None were confirmed to be the MfR response element. Ultimately, success in this effort may expand the assay to include the endpoint of protein-DNA binding in response to ligand binding.
An external reviewer of this work suggested that the protein-protein interactions measured in this study by BRET should be confirmed using a different experimental approach. Presently, we are performing experiments using far-western blotting techniques to confirm our initial observations.
- The combined actions of different chemicals along a common pathway/network
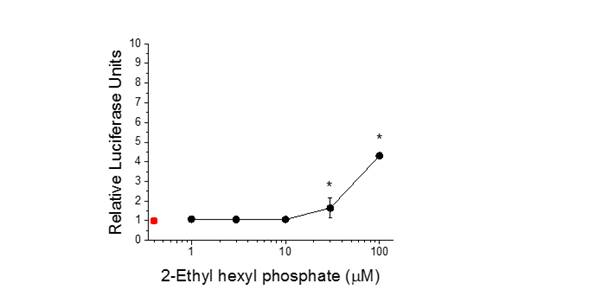
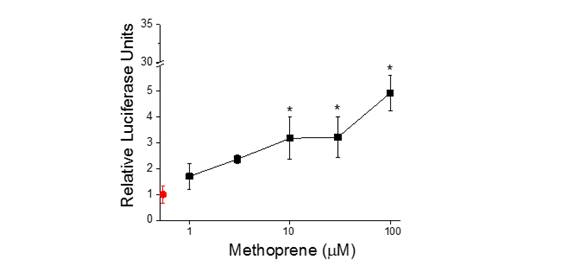
We have evaluated over 40 compounds for their ability to impact signaling of the cellular pathways under study. Chemicals have been identified that impact the same pathway through interaction at different targets. For example, the flame retardant 2-ethyl hexyl phosphate activates the PPARγ signaling pathway through interaction with the PPARγ protein (Figure 5). The insecticide methoprene also activates the PPARγ signaling pathway (Figure 6) but apparently through interaction with the PPARγ partner protein, RXRα. The consequence of co-incident exposure to these chemicals on the PPARγ signaling pathway is not known. Conceivably, co-incident exposure could result in additive or synergistic activation of the receptor. Such interactive effects are currently under study.
Figure 5. Activation of the PPAR𝛶:RXRα signaling pathway by 2-ethyl hexyl phosphate.
Red data point identifies the control (no 2-ethyl phophate). Data are presented as mean
and standard deviation (n=3). Asterik denotes a significant difference from the control (p<0.05)
Figure 6. Activation of the PPAR𝛶:RXRα signaling pathway by methoprene. Red data
point identifies the control (no methoprene). Data are presnted as mean and stardard
deviation (n=3). Asterik denotes a significant difference from the control (p<0.05). - Multiple actions of a single chemical at different sites within a pathway/network resulting in enhanced toxic outcome
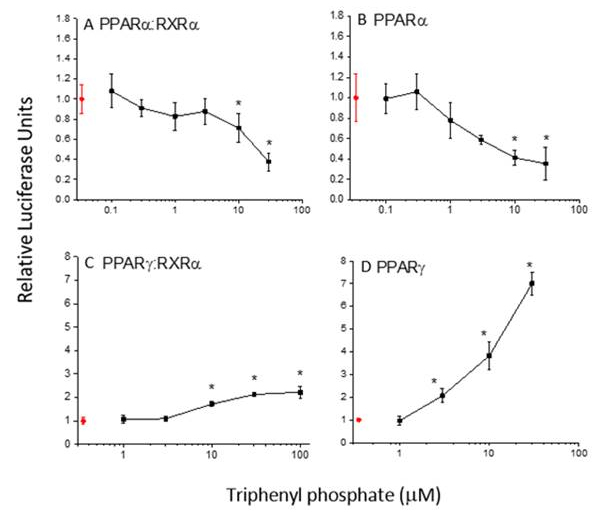
We have identified compounds that act on different pathways of a common network in a manner that would likely result in synergistic outcomes. For example, several flame retardants were identified that simultaneously activate the PPARγ signaling pathway, while inhibiting the PPARα signaling pathway (Figure 7). These opposing effects on the same signaling network would likely result in decreased lipid metabolism commensurate with enhanced lipid accumulation.
Figure 7. Modulation of the PPAR signaling netowrk by tripheyl phosphate. A. Inhibition of PPARα:RXRα. B. Inhibition of the
PPARα subunit. C. Activation of PPAR𝛶 subunit. C. Activation of PPAR𝛶:RXRα. D. Specific activation of the PPAR𝛶 subunit.
Data are presented as teh mean and standard deviation (n=3). Red data points depict the control (no triphenyl phosphate).
Asterisk indicates a significant difference from the control (p<0.05).
An external reviewer of our work suggested that we expand our assessments to determine whether pathway effects detected in our assays translate to alterations in cell function. To this end, we have evaluated several of the compounds that altered PPAR signaling in a manner that should lead to increased lipid accumulation for their ability to stimulate pre-adipocyte differentiation into adipocytes with commensurate lipid accumulation. All of the compounds found to be potentially obesogenic in our screening assays did stimulate differentiation of 3T3-L1 cells with commensurate lipid accumulation.
Several relevant conclusions were drawn from these experiments.
(1) BRET assays can be used to reliably evaluate some protein-protein interactions, but is non-informative for others. Examples follow.
- BRET is highly effective in evaluating ligand-mediated RXRα-RXRα associations.
- BRET is effective is detecting RXR ligand-mediated PPAR-RXRα associations.
- BRET is not effective in detecting PPAR ligand-mediated PPAR-RXRα association. Currently we are assessing whether this lack of detection is because PPAR ligands bind to existing PPAR-RXRα dimers and do not stimulate receptor assembly.
- Ligand-mediated recruitment of the co-activator protein SRC1 to PPAR-RXRα provides no additional information over the detection of PPAR-RXRα receptor assembly.
- Screening assay results obtained with PPARγ-RXRα are predictive of the ability of chemicals to stimulate preadipocyte differentiation and lipid accumulation.
- BRET is highly effective in evaluating ligand-mediated MET-SRC association. This endpoint shows greater potential for use in high-throughput testing than the more conventional reporter gene transcription assays.
- BRET can be used to measure the spontaneous association of the receptor proteins E75 and HR3.
- BRET can be used to measure ligand-mediated dissociation of receptor dimers.
- Combined use of BRET and reporter gene assays can provide a wealth of mechanistic information regarding the multiple effects of a single compound on a signaling network or the combined impact of multiple chemicals on a single signaling pathway/network.
Future Activities:
Experiments will be performed over the next year to further elucidate the specific interactions of the compounds evaluated and their putative molecular targets and to provide independent confirmation of some of the interactions observed using the BRET/report gene assays. These confirmatory experiments include the following.
- Experiments will be performed with compounds shown to activate the PPAR:RXRα signaling network along with the compound UVI801. This compound specifically inhibits the RXRα subunit and will establish unequivocally whether a compound activates the network via interaction with the PPAR subunits or the RXRα subunit.
- Experiments will be performed with compounds shown to activate the PPAR:RXRα signaling network along with the compound LG100754. This compound inhibits activity of the RXRα:RXRα receptor but does not inhibit the PPAR:RXRα receptors. Results will establish whether residual RXRα:RXRα receptors are present in the assays and are contributing to the activity that we are attributing to PPAR:RXRα.
- Confirmatory reporter gene transcription assays will be used when evaluating PPAR:RXRα activation using a reporter gene driven by the peroxisome proliferator response element (PPRE). Presently, we are using RXRα-GAL4 constructs in these assays with the reporter gene driven by the GAL4 response element. Confirmatory experiments will be performed using the endogenous PPRE response element to ensure that results observed are not artifacts of the experimental system used.
Once these experiments are completed, manuscripts will be written and submitted for publication.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 26 publications | 7 publications in selected types | All 7 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
LeBlanc GA, Medlock EK. Males on demand: the environmental-neuro-endocrine control of male sex determination in daphnids. The FEBS Journal 2015;282(21):4080-4093. |
R835165 (2015) R835165 (Final) |
Exit Exit Exit |
Supplemental Keywords:
Multi-sensor reporter cell signaling, signaling pathways, endocrine disruptors, assays, ligandsProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.