Grantee Research Project Results
2011 Progress Report: Toxicity of Drinking Water Associated with Alternative Distribution System Rehabilitation Strategies
EPA Grant Number: R834867Title: Toxicity of Drinking Water Associated with Alternative Distribution System Rehabilitation Strategies
Investigators: Mariñas, Benito J. , Plewa, Michael J.
Institution: University of Illinois Urbana-Champaign
EPA Project Officer: Page, Angela
Project Period: June 1, 2011 through May 31, 2015
Project Period Covered by this Report: June 1, 2011 through May 31,2012
Project Amount: $599,113
RFA: Advancing Public Health Protection through Water Infrastructure Sustainability (2009) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
This project will investigate the potential impact on public health brought about by emerging alternatives for rehabilitating drinking water distribution systems. The central hypothesis is that there will be differences in the toxicity of tap water associated with different rehabilitation approaches. Specific objectives are to perform: (a) comparative studies of mammalian cell cyto/genotoxicity for simulated distribution system waters representative of conventional rehabilitation with centralized and decentralized treatment, and emerging dual and multiple networks (hypothesis: changes in hydraulic residence time, disinfectant addition and sequential treatment associated with different rehabilitation strategies will affect water cyto/genotoxicity); (b) comparative analysis of the roles of residual disinfectants, free and combined chlorine, and relevant water quality for distribution system rehabilitation alternatives (hypothesis: switching residual disinfection practice from free to combined chlorine will increase tap water cyto/genotoxicity, more so in source waters affected by water reuse).
Progress Summary:
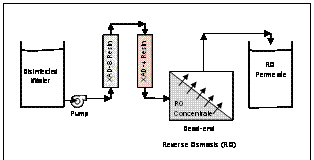
Figure 1. Experimental apparatus used to concentrate
DBPs formed in disinfected water.
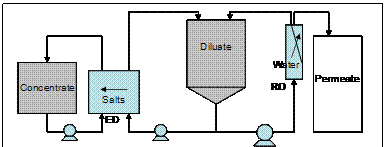
Figure 2. Hybrid RO/ED system
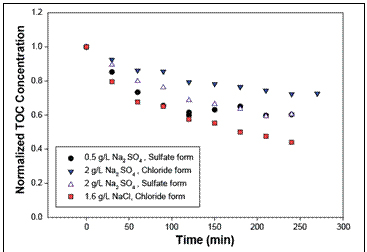
Figure 3. Changes in TOC concentrations after running
electrodialysis with added amounts of solutes.
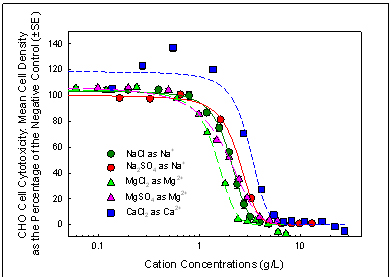
Figure 4. CO cell chronic cytotoxicity concentration-response curve
for inorganic salts expressed as cation concentrations.
Table 1. CHO cell chronic cytotoxicity of the HALs.
| Compound | (LC50)* (M) |
| Tribromoacetaldehyde (TBAL) | 3.58 X 10-6 |
| Chloroacetaldehyde (CAL) | 3.60 X 10-6 |
| Dibromoacetaldehyde (DBAL) | 4.70 X 10-6 |
| Bromochloroacetaldehyde (BCAL) | 5.34 X 10-6 |
| Iodoacetaldehyde (IAL) | 6.17 X 10-6 |
| Bromoacetaldehyde (BAL) | 1.62 X 10-5 |
| Dichloroacetaldehyde (DCAL) | 2.93 X 10-5 |
| Bromodichloroacetaldehyde (BDCAL) | 3.07 X 10-5 |
| Formaldehyde (FAL) | 7.29 X 10-5 |
| Trichloroacetaldehyde (TCAL) | 1.16 X 10-3 |
Table 2. CHO cell acute genotoxic potency values of the HALs.
| Compound | SCGE Genotoxic Potency (M)* |
| Dibromochloroacetaldehyde (DBCAL) | 1.51 X 10-4 |
| Chloroacetaldehyde (CAL) | 1.59 X 10-4 |
| Dibromoacetaldehyde (DBAL) | 1.64 X 10-4 |
| Tribromoacetaldehyde (TBAL) | 3.55 X 10-4 |
| Bromodichloroacetaldehyde (BDCAL) | 4.38 X 10-4 |
| Bromochloroacetaldehyde (BCAL) | 5.71 X 10-4 |
| Dichloroacetaldehyde (DCAL) | 8.83 X 10-4 |
| Bromoacetaldehyde (BAL) | 9.61 X 10-4 |
| Trichloroacetaldehyde (TCAL) | NS |
Future Activities:
Table 3. Test matrix for toxicological evaluation of RO/ED and XAD concentrates.
| | Halide addition | Disinfection | Work flow |
| RO/ED control | − | − | RO/ED → toxicity test RO/ED → XAD → toxicity test |
| RO/ED no halide, chlorination | − | Chlorination | RO/ED → disinfection → toxicity test RO/ED → disinfection → XAD → toxicity test |
| RO/ED no halide, chloramination | − | Chloramination | RO/ED → disinfection → toxicity test RO/ED → disinfection → XAD → toxicity test |
| RO/ED + halide, chlorination | + Br−, I− | Chlorination | RO/ED → disinfection → toxicity test RO/ED → disinfection → XAD → toxicity test |
| RO/ED + halide, chloramination | + Br−, I− | Chloramination | RO/ED → disinfection → toxicity test RO/ED → disinfection → XAD → toxicity test |
| XAD control | − | − | XAD → toxicity test |
| XAD no halide, chlorination | − | Chlorination | Disinfection → XAD → toxicity test |
| XAD no halide, chloramination | − | Chloramination | Disinfection → XAD → toxicity test |
| XAD + halide, chlorination | + Br−, I− | Chlorination | Disinfection → XAD → toxicity test |
| XAD + halide, chloramination | + Br−, I− | Chloramination | Disinfection → XAD → toxicity test |
References:
Journal Articles:
No journal articles submitted with this report: View all 9 publications for this projectSupplemental Keywords:
Sustainability, rehabilitation strategies, public health protection, safe drinking waterProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.