Grantee Research Project Results
Final Report: Consortium for Manufactured Nanomaterial Bioavailability & Environmental Exposure
EPA Grant Number: R834575Title: Consortium for Manufactured Nanomaterial Bioavailability & Environmental Exposure
Investigators: Lead, Jamie , Alvarez, Pedro J. , Colvin, Vicki L. , Klaine, Stephen J. , Luoma, Sam , Stone, Vicki , Tyler, Charles , Valsami-Jones, Eugenia , Chipman, Kevin , Viant, Mark , Fernandes, Teresa
Institution: Rice University , Clemson University , University of California - Davis , University of Exeter , University of Birmingham , Edinburgh Napier University , Natural History Museum (London)
EPA Project Officer: Aja, Hayley
Project Period: August 1, 2010 through August 1, 2013 (Extended to December 31, 2015)
Project Amount: $2,000,000
RFA: Environmental Behavior, Bioavailability and Effects of Manufactured Nanomaterials - Joint US – UK Research Program (2009) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
In order to fully and sustainably apply nanotechnology innovations, it is crucial to minimize any unintended environmental impacts resulting from the application of manufactured nanomaterials (NM). To realize this vision, industry and policymakers must base risk management decisions on sound scientific information about the environmental fate of NM; their availability to receptor organisms (including related concepts such as uptake); and any resultant biological effects (toxicity). This basic knowledge can be effectively conveyed by validated models which describe NM behavior and relate this data to information about NM structure and relevant environmental chemistry. These models, whether conceptual or predictive in nature, will give decision-makers the tools to grapple with the nearly infinite forms of possible NM, as well as explore the effects of various risk mitigation strategies. Towards this end, the nanoBEE consortium will develop and refine, using empirical data, a critical subset of models focused on exposure to NM and their bioavailability in the environment.
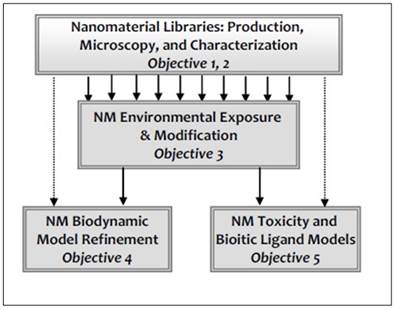
The methodological approach in the nanoBEE consortium is summarized in Figure 1. Tunable and uniform NMs are characterized appropriately with existing and novel methods developed in this consortium. The libraries will underpin steady-state and dynamic laboratory and mesocosm experiments to understand NM environmental chemistry, transport, bioavailability and toxicity. These data sets are being used to validate exposure, bioavailability and toxicity based models, and then merged and refined through experiments. The result will be an integrated and validated set of models (EEM-BDM-BLM) (EEM: Environmental Exposure Model; BDM: Biodynamic Model; BLM: Biotic Ligand Model), with, we anticipate, some direct integration between BDM-BLM. Research in the nanoBEE consortium is structured by 5 primary objectives, and twelve tasks listed below. The produced data and models will link to risk assessment modelling strategies.
Aim 1: Nanomaterial Library Production, Characterization
Aim 2: Aqueous electron tomography of nanomaterials
Aim 3. Environmental chemistry, fate and exposure models (EEMs)
Aim 4. NM association with organisms and the biodynamic model
Aim 5. Effects on aquatic organisms towards a nano-BLM model
Summary/Accomplishments (Outputs/Outcomes):
This report summarizes progress made by the US-UK jointly funded project nanoBEE, on which we (Dr Lead and Dr Colvin) are overall Lead/UK PI and US PI, respectively. The project has had a number of management issues, including the inherent difficulty of managing project work that is split between two continents, with the majority of work performed in two countries but with a number of other European countries involved. Other issues have included, for instance, three quarters of the UK Principal or Co-investigators moving institution within the project lifetime, including the Lead/UK-PI moving continents: Lead to the University of South Carolina; Valsami-Jones to the University of Birmingham and Stone/Fernandes to Heriot Watt University. Nevertheless, these issues have been successfully overcome and nanoBEE is making excellent progress.
The project has five main aims, which encompass 12 tasks all aimed at developing, refining and testing available models for nanoparticle exposure, fate, bioavailability and toxicity and these aims are considered below.
Aim 1: Production of nanomaterial libraries and full characterization.
The focus of this work has been around silver, with some work on gold, titania, ceria and other nanoparticles. In addition to core size, we have worked to make different shapes, sizes, and coatings, producing a large library of available nanomaterials, which has been used systematically across the consortium. Particularly, silver nanoparticles with tunable size within a broad range and a great variety of surface chemistry have been synthesized to systematically study the influence of these attributes on their environmental chemistry.
A multi-method approach to characterization as prepared and in complex media has been previously developed and applied. The approach was discussed, further developed and applied within nanoBEE to quantify the physico-chemical parameters of pristine nanomaterials and to investigate their behavior in both environmental samples and in ecotoxicological media. Tightly constrained, in-house produced NPs of silver, gold, ceria and other NM types have been produced and characterized in terms of dissolution, aggregation, surface properties and other effects in relevant exposure media and in environmental media including with added natural organic macromolecules (NOM). The Valsami-Jones and Colvin laboratories have developed protocols for measuring dissolution and solubility while the Lead laboratories have developed protocols for measurement of surface properties e.g. quantification of NOM coronas on NP surface, oxidation state etc., all with sub-nm resolution, along with development of validated protocols for number particle concentration to be applied. Aggregation in various media has been investigated quantitatively, in some cases showing that aggregation impacts on toxicity by reducing the inherent toxicity rather than alteration of exposure concentration/dose. Figure 1 shows STEM images of AgNPs at very high resolution on exposure to NOM, and their related EELS spectra. From such evidence we were able to image and quantify the behavior at the Ag interface with the NOM, showing the absence of any silver oxide in spite of the presence of oxygen from the NOM.
Jointly, but with Valsami-Jones providing leadership, stable isotope labeled NPs have been developed and used in order to enable detection and tracing of such modified nanoparticles. We have adapted synthesis protocols commonly used to produce Ag NPs (via reduction of a suitable Ag salt) to introduce the stable isotope label. Several batches of electrostatically stabilised (via citrate capping) Ag NPs (TEM size: 15±5nm, -47mV, SPR=400nm), highly enriched (99%) with 109Ag were produced and extensively characterized (size and size distribution, surface charge, purity by ICP-AES, crystal phase by XRD, Surface Plasmon Resonance, dispersability by DLS and NTA). The synthesis protocol delivers highly reproducible batches of isotopically modified Ag NPs and can be used to introduce 107Ag or 109Ag stable isotope label. Characterization of the unlabelled and labelled Ag NPs produced via the same protocol indicates, that introduction of the stable isotope label does not change particle physico-chemical properties in any significant way. The Ag NPs produced at NHM were characterized in pristine form (as synthesized) and in the relevant exposure media used in eco-toxicology experiments (i.e. moderately hard freshwater) and delivered to the project partners at USGS and University of Exeter for ecotoxicological tests. Similarly, labeled ceria NPs are developed and will be used for relevant exposure studies (aims 4-5).
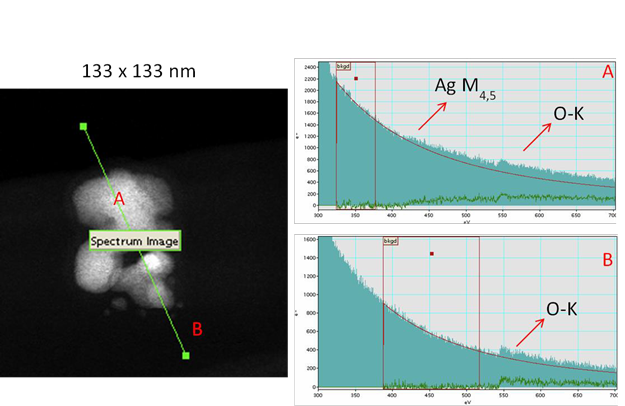
Figure 1. STEM image at sub-nanometer scale, showing associated EELS spectra, again at sub-nanometer length scales. Such data have been used to quantify surface phenomena at the near atom scale, including oxidation, sulfidation and NOM interactions. In particular these processes have been studied dynamically in relevant environmental, exposure and biological media
Aim 2: Development of a non-perturbing and accurate tomography
Nano-clusters, size selected to 5% by mass, are used for accurate alignment of cryo-tomography, particularly in the study of the eco-corona formation i.e. the surface coating developed by NPs in environmental media, known to have significant effects on fate and effects of NMs and linked strongly to aims 3-5. In TEM tomography, images are acquired at a number of positions by varying the position of the sample with respect to the incoming electron beam. The acquired tilt series must be realigned to negate sample movement during the mechanical tilt stage. The principle is that the more accurately the series of images at different tilts can be aligned, the better resolution and accuracy of the final 3D reconstruction. In room temperature tomography this is not a problem as the (gold) alignment markers are added after the sample and therefore the position can be determined from >30 markers with ease, so the accuracy is high even with a dispersed size range. However, under cryo-conditions i.e. at very low temperature, this is not the case. In particular, the patch of interest within the tomogram can be small and over-sampling is a requirement of the resolution enhancing algorithms. Therefore, magnification must be high leading to a small field of view. Furthermore the work is performed in suspended vitreous ice, it is unlikely that a sufficient number of markers (effectively internal standards) within a usable field of view will be obtained. Therefore, small and highly size-selected markers are required. The cryo-conditions are required to reduce the likelihood of alteration of NOM and other easily perturbed and hydrated environmental structures.
To date, ferritin, ceria, Ag, AgO, AgS and others have been imaged under relevant conditions using aberration corrected (near-atom resolution) STEM. This has been followed EELs analysis to determine chemical behavior at the same spatial resolution. The <0.1nm resolution of the microscope allows the structure throughout the nanoparticle to be determined. The results show that an outer surface (<2 atomic layers) can be determined on the particle. Ferritin and MoS2 have been co-deposited with size-selected clusters to act as a mass balance (internal standard). Separately automatic cryo-tilt series have been acquired so the expertise is in place for generalized cryo-tomography. Further, lipid disks filled with proteins and/or selected contrast metallic atoms have been imaged under appropriate conditions. A number of trial and error developmental experiments have been performed showing that it is not possible to collect a tilt series with the size-selected clusters on the carbon support and imaging the sample in a neighbouring ice suspension, as the carbon/beam interaction melt the ice under preferred imaging conditions. Therefore, size selected clusters, previously put onto a solid substrate, must be dispersed at a sufficiently high concentration. Currently we have used an aluminium foil support to dissolve the clusters and redeposit on a grid for imaging but not at the concentration required. We are in the process of using a citrate substrate, which can be directly dissolved and obtaining a KBr substrate (dissolvable and optically clear). The initial step is to concentrate the clusters using low vacuum evaporator and to functionalize them, enabling concentration and separation allowing also chemical labeling, along with cryo-tomography.
Aim 3; Environmental chemistry, fate and exposure
NanoBEE has studied solubility, aggregation, surface alterations and sulfidation processes in detail at the laboratory and mesocosm scale, using expertise gained from previous NERC projects. For instance, at the laboratory scale, the aggregation of isotopically-modified Ag NPs in different types of freshwaters, marine waters and ecotoxciological exposure waters has been quantified and modeled using a variety of methods. In general, charge stabilized NPs aggregate to ca 100 nm stable aggregates, while polymer coated particles are much more resistant to aggregation. Chloride seems to have a stabilizing effect to dissolution, while organic matter such as humic substances colloidally stabilizes charge-stabilized NPs while having little effect on many polymer-coated NPs.
Dissolution of Ag NPs (variety of sizes and coatings) was tested using different methods in order to establish a reliable dissolution measurement protocol. The following methods were tested: dialysis membranes, ultrafiltration, ultracentrifugation and centrifugal filtration. Particles from one synthesis batch were used in all experiments, ionic Ag was used to control recovery and Ag was analyzed in the acidified samples by ICP-MS. Estimated particle dissolution varied from 1.2 to 17.5% depending on the method used. The recovery for ionic Ag varied from 26% to 102%, with best recovery and reproducibility (7.4% RSD on repeated samples) achieved with centrifugal filtration. This method was then used to test dissolution of Ag NPs in different media (1mM NaNO3 and moderately hard freshwater), in short term (hours) and long-term (up to 30 days) experiments. Estimated dissolution of citrate coated Ag NPs varied from 2% (labelled particles in moderately hard freshwater) to 7.5% (unlabeled particles in 1mM NaNO3).
A series of silver nanoparticles samples with the same surface coating but different sizes and another series of samples with the same size but different surface coatings were used to study the influence of size and surface coatings on the dissolution chemistry of silver nanoparticles. It was found that both nanoparticle size and surface chemistry have significant influence on the dissolution profile of silver nanoparticles.
Exposure models developed externally to nanoBEE have been investigated in a non-experimental study investigating gold used in nanomedicines and potential exposures. A probabilistic material flow model developed at EMPA, Switzerland was validated against production data to quantify mass concentrations in a range of environmental compartments. Predicted environmental concentrations were in all cases insufficient to indicate significant risk, although this may not be the case when taking into consideration all gold sources. However, the use of nanomedicine products is a relatively information-rich area and there would be considerable uncertainty in extending this analysis to other sectors.
Larger, mesocosm-scale behavior has been investigated in simulated water-sediment systems and in simulated waste water treatment plants. In both cases, simple systems have been used initially to aid in data interpretation of later, more realistic systems. Initial cases have used low ionic strength, organic-free solutions to investigate aggregation and dissolution in freshwater- sediments systems, and simulated sewage to study the same processes. Later studies have used real waters/sediments and actual waste waters and sludge, respectively. In all cases, the data collection and validation is coming to an end and the data are being included into relevant fate and exposure models. Collaboration with PNNL in the United States has been developed to model the fate and behavior of the Ag NPs introduced, including diffusion, agglomeration and dissolution.
Aim 4 and 5; NM association with organisms and the biodynamic and biotic ligand models
Extensive work has been performed on the particles mentioned in aims 1-3, using silver, gold and ceria, with different sizes, surface coatings and shapes. In all cases, we have followed dynamic changes in exposure concentration and NP properties to quantify the system, never relying on nominal doses. Exposures have been performed on Daphnia magna, Lumbriculus, Lymnaea stagnalis and several types of fish. Work on cell cultures has been performed and algal exposures are planned. In addition, water quality (hardness, humic substances, waste waters) and exposure routes (water and dietary) have been studied experimentally and via modelling approaches.
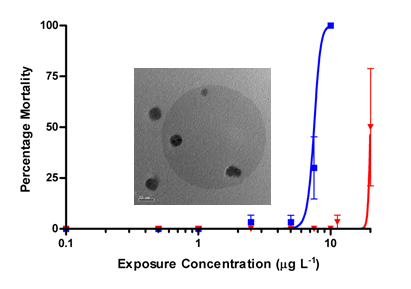

Figure 2. Effect of colloidal stability of silver nanoparticles on toxicity of Daphnia magna. The dose-response data shows the effect of 10 nm citrate-coated Ag NPs on D. magna in the dispersed phase (blue line; EM image showing dispersion on left) and agglomerated form of the same NPs (red line, EM image showing aggregation on right). Aggregation reduces toxicity by approximately an order of magnitude due to inherent biouptake differences of agglomerated and dispersed NPs. Dynamic alteration in mass dose is not required to explain data. The toxicity data is backed by other measures on effects, biouptake data and omic data. Characterization was performed utilizing a multi-method approach including TEM, AFM, FFF, CE, DLS and dissolution.
As an example, work with zebra fish and rainbow trout has included:
Rainbow trout Gill Cell Cultures
Initial attempts were made to develop and apply a rainbow trout gill epithelial cell culture systems for high throughput analysis of factors that affect NP bioavailability. A culture system was successfully developed, but the high variability in cell viability and lack of consistency between the cultures in cell attachment suggested the system was inappropriate for studies on MNP uptake.
Mechanism of action of bulk versus nano silver- zebrafish embryos and early life stages
The toxicological properties of Ag and TiO2 in zebrafish embryos and at early life stages were investigated, including the application of advanced imaging methods including CARS and EM. Evidence that the toxicological properties of silver MNPs largely, although not entirely, relates to the release of silver ions was provided.
Building on this work, a novel whole mount in situ hybrisidation approach in the zebrafish has been developed to assess for exposure effects to silver NMs compared with bulk counterparts. In this work we have developed significant information on the ontogeny of expression of a suite of genes that are involved in detoxification and oxidative stress and tissue specific responses to the silver materials.
The zebrafish model (embryos) has been further used for transcriptomic analysis of the genome wide response to silver materials (nanos and bulk and ionic), again providing evidence that the main effects of the nanomaterial are as a function of silver ions.
Bioavailability, target organs, and molecular effect pathways of nano versus bulk-sized zinc oxide and silver particles in zebrafish
This work has included a 7 day dietary exposure of fish in individual flow-through tanks. Measurement of BCF was performed by ICP-MS, including isotopic analysis, along with qPCR for 5 known zinc or silver-responsive genes across 5 tissues (gill, brain, gut, liver, kidney) to select the most-responsive tissue. Finally, the transcriptome has been analysed to identify mechanisms of action
Transgenerational effects of silver MNPs
In addition in zebrafish a long term exposure to silver MNPs and a bulk counterpart via the diet to assess effects on reproduction has been performed, assessing effects of parental exposure to these material on the offspring including effects on responses of genes involved with metal handing and oxidative stress. The fish were dosed at concentrations of 20ug/g body weight and no effects on egg production were found, but earl results suggest the offspring are more highly responsive to silver.
Assessment of the uptake and biological effects of metal and metal oxide nanoparticles in a treated WWTW effluent in fish.
We have undertaken a large scale exposure of rainbow trout for 21 days to a waste water effluent spiked with TiO2, Ag and Zn MNPs. Various tissue were dissected for ICP-MS and gene expression analyses. Genomic and characterization data are being collected and the study is on- going.
Xenotoca- a new model for studies on maternal transfer of MNPs
A pilot study exposure and a longer term exposure of Xenotoca sp. to silver materials via the diet to assess for effects on transfer to the offspring and health effects in those offspring is being performed. The pilot data indicate a higher level of silver in offspring in the adult fish fed silver NPs compared with the bulk counterpart. Analysis of many hundreds of samples for Ag uptake is on-going, although there were no effects of the treatment on numbers or mass of the offspring produced.
Finally, the various exposures performed on a range of organisms have partly been modelled, especially using the biodynamic model and further work is on-going with the biotic ligand model. In a number of cases we were able to perform modelling on exposures to the same organisms, improving data utility. Figure 3 shows shows that at low exposure concentrations (<0.1 ug/L), 70% of the bioaccumulated Ag can be ascribed to uptake from newly solubilized Ag, whereas 80% of the bioaccumulated Ag originated from the AgNPs above this concentration i.e. there are concentration dependent changes in the species or form of the silver that is bioaccumulated.
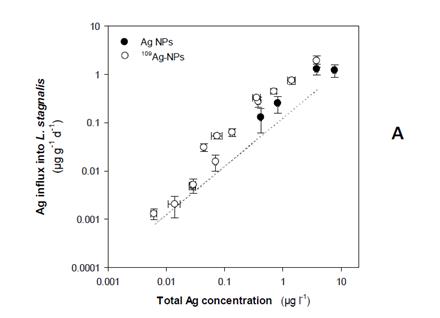
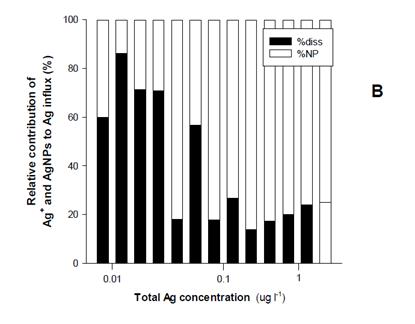
Figure 3. A) Silver uptake rates (µg g-1 d-1 ± 95% C.I.) in L. stagnalis soft tissues after waterborne exposure to citrate-capped 109Ag NPs. The solid symbols are from the study of Croteau et al[32]. Each symbol represent Ag concentrations for 10 individuals and 5 water samples (± 95% C.I.). The dotted line represents Ag influxes in L. stagnalis predicted using equation 6 assuming 109Ag NPs dissolution of 7%. B) Relative contribution of Ag+ and Ag NPs to the overall Ag influxes in L. stagnalis as predicted using equation 6 and assuming 109Ag NPs dissolution of 7%.
Journal Articles on this Report : 35 Displayed | Download in RIS Format
| Other project views: | All 37 publications | 37 publications in selected types | All 35 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Arkill KP, Mantell JM, Plant SR, Verkade P, Palmer RE. Using size-selected gold clusters on graphene oxide films to aid cryo-transmission electron tomography alignment. Scientific Reports 2015;5:9234 (4 pp.). |
R834575 (Final) |
Exit Exit |
|
|
Baalousha M, Arkill KP, Romer I, Palmer RE, Lead JR. Transformations of citrate and Tween coated silver nanoparticles reacted with Na2S. Science of the Total Environment 2015;502:344-353. |
R834575 (Final) |
Exit Exit Exit |
|
|
Benoit DN, Zhu H, Lilierose MH, Verm RA, Ali N, Morrison AN, Fortner JD, Ayendano C, Colvin VL. Measuring the grafting density of nanoparticles in solution by analytical ultracentrifugation and total organic carbon analysis. Analytical Chemistry 2012;84(21):9238-9245. |
R834575 (Final) |
Exit Exit Exit |
|
|
Collin B, Auffan M, Johnson AC, Kaur I, Keller AA, Lazareva A, Lead JR, Ma X, Merrifield RC, Svendsen C, White JC, Unrine JM. Environmental release, fate and ecotoxicological effects of manufactured ceria nanomaterials. Environmental Science-Nano 2014;1(6):533-548. |
R834575 (Final) |
Exit Exit |
|
|
Contreras EQ, Cho M, Zhu H, Puppala HL, Escalera G, Zhong W, Colvin VL. Toxicity of quantum dots and cadmium salt to Caenorhabditis elegans after multigenerational exposure. Environmental Science & Technology 2013;47(2):1148-1154. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Cross RK, Tyler C, Galloway TS. Transformations that affect fate, form and bioavailability of inorganic nanoparticles in aquatic sediments. Environmental Chemistry 2015;12(6):627-642. |
R834575 (Final) |
Exit Exit |
|
|
Croteau MN, Dybowska AD, Luoma SN, Misra SK, Valsami-Jones E. Isotopically modified silver nanoparticles to assess nanosilver bioavailability and toxicity at environmentally relevant exposures. Environmental Chemistry 2014;11(3):247-256. |
R834575 (Final) R834557 (Final) |
Exit |
|
|
Croteau MN, Misra SK, Luoma SN, Valsami-Jones E. Bioaccumulation and toxicity of CuO nanoparticles by a freshwater invertebrate after waterborne and dietborne exposures. Environmental Science & Technology 2014;48(18):10929-10937. |
R834575 (Final) R834557 (Final) |
Exit Exit Exit |
|
|
Croteau M-N, Misra SK, Luoma SN, Valsami-Jones E. Silver bioaccumulation dynamics in a freshwater invertebrate after aqueous and dietary exposures to nanosized and ionic Ag. Environmental Science & Technology 2011;45(15):6600-6607. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Croteau M-N, Cain DJ, Fuller CC. Novel and nontraditional use of stable isotope tracers to study metal bioavailability from natural particles. Environmental Science & Technology 2013;47(7):3424-3431. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Edgington AJ, Petersen EJ, Herzing AA, Podila R, Rao A, Klaine SJ. Microscopic investigation of single-wall carbon nanotube uptake by Daphnia magna. NanoToxicology 2014;8(Suppl 1):2-10. |
R834575 (Final) R834092 (Final) |
Exit Exit |
|
|
Fabrega J, Luoma SN, Tyler CR, Galloway TS, Lead JR. Silver nanoparticles: behaviour and effects in the aquatic environment. Environment International 2011;37(2):517-531. |
R834575 (2011) R834575 (Final) |
Exit Exit Exit |
|
|
Glenn JB, White SA, Klaine SJ. Interactions of gold nanoparticles with freshwater aquatic macrophytes are size and species dependent. Environmental Toxicology and Chemistry 2012;31(1):194-201. |
R834575 (2013) R834575 (Final) R834092 (Final) |
Exit Exit |
|
|
Glenn JB, Klaine SJ. Abiotic and biotic factors that influence the bioavailability of gold nanoparticles to aquatic macrophytes. Environmental Science & Technology 2013;47(18):10223-10230. |
R834575 (2013) R834575 (Final) R834092 (Final) |
Exit Exit Exit |
|
|
Goodhead RM, Moger J, Galloway TS, Tyler CR. Tracing engineered nanomaterials in biological tissues using coherent anti-Stokes Raman scattering (CARS) microscopy--a critical review. NanoToxicology 2015;9(7):928-939. |
R834575 (Final) |
Exit Exit |
|
|
Kalman J, Paul KB, Khan FR, Stone V, Fernandes TF. Characterisation of bioaccumulation dynamics of three differently coated silver nanoparticles and aqueous silver in a simple freshwater food chain. Environmental Chemistry 2015;12(6):662-672. |
R834575 (Final) |
Exit Exit |
|
|
Khan FR, Paul KB, Dybowska AD, Valsami-Jones E, Lead JR, Stone V, Fernandes TF. Accumulation dynamics and acute toxicity of silver nanoparticles to Daphnia magna and Lumbriculus variegatus: implications for metal modeling approaches.Environmental Science & Technology 2015;49(7):4389-4397. |
R834575 (Final) |
Exit Exit Exit |
|
|
Lewicka ZA, Yu WW, Oliva BL, Contreras EQ, Colvin VL. Photochemical behavior of nanoscale TiO2 and ZnO sunscreen ingredients. Journal of Photochemistry and Photobiology A: Chemistry 2013;263:24-33. |
R834575 (Final) |
Exit |
|
|
Lowry GV, Gregory KB, Apte SC, Lead JR. Transformations of nanomaterials in the environment. Environmental Science & Technology 2012;46(13):6893-6899. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Merrifield RC, Wang ZW, Palmer RE, Lead JR. Synthesis and characterization of polyvinylpyrrolidone coated cerium oxide nanoparticles. Environmental Science & Technology 2013;47(21):12426-12433. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Misra SK, Dybowska A, Berhanu D, Luoma SN, Valsami-Jones E. The complexity of nanoparticle dissolution and its importance in nanotoxicological studies. The Science of the Total Environment 2012;438:225-232. |
R834575 (2013) R834575 (Final) R834557 (Final) |
Exit Exit Exit |
|
|
Newton KM, Puppala HL, Kitchens CL, Colvin VL, Klaine SJ. Silver nanoparticle toxicity to Daphnia magna is a function of dissolved silver concentration. Environmental Toxicology and Chemistry 2013;32(10):2356-2364. |
R834575 (2013) R834575 (Final) |
Exit Exit |
|
|
Osborne OJ, Johnston BD, Moger J, Balousha M, Lead JR, Kudoh T, Tyler CR. Effects of particle size and coating on nanoscale Ag and TiO2 exposure in zebrafish (Danio rerio) embryos. Nanotoxicology 2013;7(8):1315-1324. |
R834575 (2013) R834575 (Final) |
Exit Exit |
|
|
Romer I, Gavin AJ, White TA, Merrifield RC, Chipman JK, Viant MR, Lead JR. The critical importance of defined media conditions in Daphnia magna nanotoxicity studies. Toxicology Letters 2013;223(1):103-108. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Seda BC, Ke P-C, Mount AS, Klaine SJ. Toxicity of aqueous C70-gallic acid suspension in Daphnia magna. Environmental Toxicology and Chemistry 2012;31(1):215-220. |
R834575 (2013) R834575 (Final) R834092 (Final) |
Exit Exit |
|
|
Stoiber T, Croteau MN, Romer I, Tejamaya M, Lead JR, Luoma SN. Influence of hardness on the bioavailability of silver to a freshwater snail after waterborne exposure to silver nitrate and silver nanoparticles. NanoToxicology 2015;9(7):918-927. |
R834575 (Final) |
Exit |
|
|
Tejamaya M, Romer I, Merrifield RC, Lead JR. Stability of citrate, PVP, and PEG coated silver nanoparticles in ecotoxicology media. Environmental Science & Technology 2012;46(13):7011-7017. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
van Aerle R, Lange A, Moorhouse A, Paszkiewicz K, Ball K, Johnston BD, de-Bastos E, Booth T, Tyler CR, Santos EM. Molecular mechanisms of toxicity of silver nanoparticles in zebrafish embryos. Environmental Science & Technology 2013;47(14):8005-8014. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Wray AT, Klaine SJ. Modeling the influence of physicochemical properties on gold nanoparticle uptake and elimination by Daphnia magna. Environmental Toxicology and Chemistry 2015;34(4):860-872. |
R834575 (Final) R834092 (Final) |
Exit |
|
|
Xiu Z-M, Ma J, Alvarez PJJ. Differential effect of common ligands and molecular oxygen on antimicrobial activity of silver nanoparticles versus silver ions. Environmental Science & Technology 2011;45(20):9003-9008. |
R834575 (2011) R834575 (2013) R834575 (Final) |
Exit Exit Exit |
|
|
Xiu Z-M, Zhang Q-B, Puppala HL, Colvin VL, Alvarez PJJ. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Letters 2012;12(8):4271-4275. |
R834575 (2013) R834575 (Final) R834557 (Final) |
Exit Exit Exit |
|
|
Yang Y, Mathieu JM, Chattopadhyay S, Miller JT, Wu T, Shibata T, Guo W, Alvarez PJJ. Defense mechanisms of Pseudomonas aeruginosa PAO1 against quantum dots and their released heavy metals. ACS Nano 2012;6(7):6091-6098. |
R834575 (2013) R834575 (Final) R834557 (Final) |
Exit Exit Exit |
|
|
Yang Y, Wang J, Zhu H, Colvin VL, Alvarez PJJ. Relative susceptibility and transcriptional response of nitrogen cycling bacteria to quantum dots. Environmental Science & Technology 2012;46(6):3433-3441. |
R834575 (2013) R834575 (Final) R834557 (Final) |
Exit Exit Exit |
|
|
Yang Y, Wang J, Xiu Z, Alvarez PJJ. Impacts of silver nanoparticles on cellular and transcriptional activity of nitrogen-cycling bacteria. Environmental Toxicology and Chemistry 2013;32(7):1488-1494. |
R834575 (2013) R834575 (Final) |
Exit |
|
|
Yang Y, Quensen J, Mathieu J, Wang Q, Wang J, Li M, Tiedje JM, Alvarez PJJ. Pyrosequencing reveals higher impact of silver nanoparticles than Ag+ on the microbial community structure of activated sludge. Water Research 2014;48:317-325. |
R834575 (2013) R834575 (Final) |
Exit Exit Exit |
Supplemental Keywords:
Nanotechnology, nanoscience, bioavailability, emerging contaminants, environmental modeling, manufactured nanoparticles, biotic ligand model, biodynamic model, environmental exposure modelProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
