Grantee Research Project Results
Final Report: Bioavailability of Metallic Nanoparticles and Heavy Metals in Landfills
EPA Grant Number: R833893Title: Bioavailability of Metallic Nanoparticles and Heavy Metals in Landfills
Investigators: Hu, Zhiqiang , Wall, Judy D. , Elias, Dwayne A.
Institution: University of Missouri - Columbia
EPA Project Officer: Aja, Hayley
Project Period: April 1, 2009 through March 31, 2012
Project Amount: $399,262
RFA: Exploratory Research: Nanotechnology Research Grants Investigating Fate, Transport, Transformation, and Exposure of Engineered Nanomaterials: A Joint Research Solicitation - EPA, NSF, & DOE (2007) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
Municipal landfills pose potential risks to the environment and human health because of landfill gas emission and groundwater contamination due to leachate leakage. These landfill sites emit millions of tons of methane, an important greenhouse gas, to the atmosphere. Compared to the traditional "dry tomb" municipal landfills, bioreactor landfills represent a new type of landfill designed to quickly transform and degrade organic waste through leachate recirculation for more effectively solids stabilization, methane recovery and energy production.
As nanotechnology-enhanced materials are emerging in landfills, there is a great concern regarding their fate, bioavailability, and toxicity in the complex solid stabilization systems. In particular, little is known about the leachate toxicity in the presence of nanoparticles in bioreactors as a result of leachate recirculation, which could significantly impact methane production. The objectives of this research are to determine bioavailability of nanoparticles and heavy metal species in bioreactor landfills and to elucidate the mechanisms governing bioavailability as well as the mode of antimicrobial action by nanoparticles.
Summary/Accomplishments (Outputs/Outcomes):
1. Impact of Silver Nanoparticles (AgNPs, nanosilver) on Anaerobic Digestion
Solids waste in landfills is decomposed mainly under anaerobic conditions. In a typical anaerobic landfill, a series of anaerobic reactions occur: (1) hydrolysis, where large organic molecules are broken down into sugars, amino acids, and fatty acids; (2) acidogenesis, where large molecules are further broken down into simple organic acids, ammonia, carbon dioxide and other byproducts; (3) acetogenesis, where acetate is produced by anaerobic bacteria from a variety of energy (e.g., hydrogen) and carbon (e.g., carbon dioxide) sources; and (4) methanogenesis, where methane is produced through hydrogen oxidation (4 H2 + CO2→CH4 + 2 H2O) and acetoclastic cleavage of acetate (CH3COO- + H+→ CO2 + CH4).
We first determined the impact of AgNPs on anaerobic degradation of glucose, sludge digestion and methanogenic assemblages. A modified biochemical methane potential (BMP) test was used to infer anaerobic microbial activity and nanosilver toxicity. The biogas production tests have proved to be very sensitive for the detection of metal toxicity through the evaluation of biogas generation rate and cumulative gas volume. Digested sludge was taken from a two-stage mesophilic digester (run at 20-30 days of hydraulic retention time) at the Columbia Waste Water Treatment Plant in Columbia, MO. A 16-bottle AER-200 Respirometer (CHALLENGE Technology, AK) was used for all batch anaerobic digestion experiments. The cumulative biogas production with and without AgNP treatment was automatically monitored and recorded. At ambient (22 °C) and mesophilic temperatures (37 °C), there was no significant difference in biogas and methane production between the sludge treated with AgNPs at the concentrations up to 40 mg Ag/L (13.2 g silver/Kg biomass COD) and the control. In these anaerobic digestion samples, acetate and propionic acid were the only detectable volatile fatty acids (VFAs) and they were depleted in 3 days. On the other hand, more than 90% of AgNPs was removed from the liquid phase and associated with the sludge, but almost no silver ions were released from AgNPs under anaerobic conditions. Quantitative PCR results indicated that Methanosaeta and Methanomicrobiales were the dominant methanogens, and the methanogenic diversity and population remained largely unchanged after nanosilver exposure and anaerobic digestion. The results suggest that under strictly anaerobic conditions AgNPs at moderate concentrations (e.g., £ 40 mg/L) have negligible impact on anaerobic digestion and methanogenic assemblages because of little to no silver ion release.
2. Fate and Toxicity of Silver Nanoparticles in Bioreactor Landfills
Nanosilver is one of the most commonly used nanomaterials because of its strong antimicrobial activity. In addition to direct landfill disposal, silver nanoparticles released from consumer products (e.g, socks, paints and food containers) in water may be discharged to the sewer system, enriched in sludge, and finally dumped in landfills. Bioreactor landfills are a relatively new technology. Unlike the traditional landfills, bioreactor landfills offer benefits that include rapid waste degradation, airspace gain, and generally improved landfill gas recovery. To determine the impact of AgNPs on anaerobic digestion of landfill waste, municipal solid waste (MSW) was loaded in identical landfill bioreactors (9 L volume each) and exposed to AgNPs (average particle size = 21 nm) at the final concentrations of 0, 1, and 10 mg Ag/kg solids. The landfill anaerobic digestion was carried out for more than 250 days, during which time the cumulative biogas production was recorded automatically and the chemical property changes of leachates were analyzed.
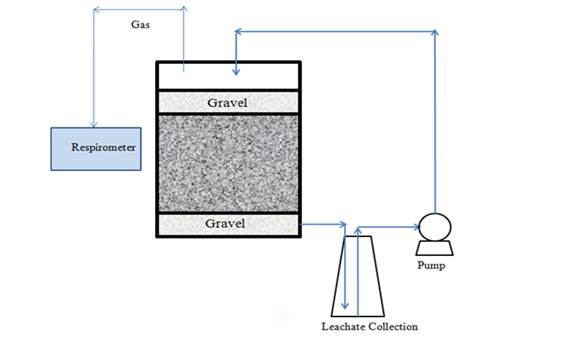
Fresh municipal solid waste (MSW) was obtained from the Columbia Sanitary Landfill in Columbia, MO. The solid waste samples were separated into different types of materials and shredded into 2-5 cm pieces, then well mixed before they were transferred to 9 L glass bottles. The composition of the solid was as follows in wet weight percent: metal 0.9%, paper 14.1%, brick 17.7%, wood and shredding 4.8%, soil 36.7%, organic (mainly food debris) 15.6%, and plastic bags 10.2%. A total of 6 bench-scale landfill bioreactors of equal volume were originally set up for duplicate testing (Figure 1). Each glass bottle had a diameter of 20 cm and a height of 30 cm. In each bioreactor, a 20 mm thick layer of gravel (average gravel particle size = 10 mm) was placed at the bottom to simulate the leachate collection layer and avoid clogging problems in leachate pipes. Then 2.925 kg (air-dried weight) of well-mixed MSW was loaded onto the gravel layer and manually compacted. To expedite biodegradation, 1000 mL portion of anaerobic sludge with a dry solids content of around 7% was taken from the Columbia Wastewater Treatment Plant in Columbia, MO, and added to each bioreactor for microbial seeding. The silver concentration of this sludge was measured at around 0.23 mg/L, so the final silver concentration contributed from the sludge would be 0.078 mg silver/ kg solids. A total of 387 mL liquid (deionized water or nanosilver suspension) was added gradually and evenly on top of solids over a 5-min period to reach the final silver concentrations of 0, 1 and 10 mg/kg, respectively. Thus, the final water content in the solids of all bioreactors was maintained at the same level, which was measured to be about 40% gravimetrically (oven-dried solid samples at 105oC for 24 hours). A 20 mm-thick gravel layer was finally placed on top of solids. The bioreactors were sealed and placed in a constant temperature room at 37 oC. Leachates collected from a sealed 1 L Erlenmeyer flask were recirculated once (lasting for a total of 35 minutes) everyday using a peristaltic pump at a target leachate recirculation ratio of 5 waste volume.
Figure 1. A chematic of lab-scale landfill reactor with leachate recirculation.
The cumulative gas volume from each bioreactor was recorded once every 10 minutes by the AER-200 Respirometer (Challenge Technology, AK) during the period of study. Detailed contents of hydrogen, carbon dioxide and methane in the biogas were analyzed later by gas chromatography (GC, Shimadzu 2014). An aliquot (20 mL) of the leachates was taken from each bioreactor biweekly for chemical analysis. DI water (20 mL) was then added back to the leachates so that the chemical concentrations in leachate could be comparable without the change of total liquid volume.
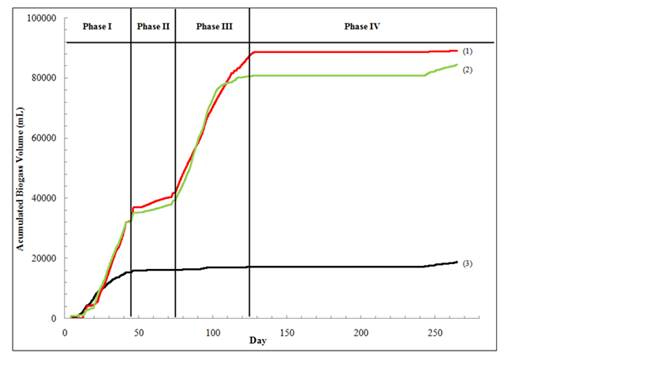
The major components of biogas in landfill are CO2, CH4, and a small amount of H2 and N2. In all bioreactors during Phase I (before day 42), carbon dioxide was determined to be the dominant gas component (close to 100%). In the phase II and III, the methane concentration in the control and the bioreactor treated with AgNPs at 1 mg Ag/kg solids were almost the same, which were in the range of 40-50%. In the last phase (Phase IV, after day 130), there was negligible biogas production in both the control and the treatment with AgNPs at 1 mg/kg solids and only carbon dioxide was detected in the gas samples, indicating the completion of methanogenesis. In comparison, mainly CO2 was detected in the first three stages in the bioreactor treated with AgNPs at 10 mg/kg solids. Coupled with the cumulative gas production profiles (Figure 2), the results indicated that methanogenesis was strongly inhibited in the bioreactor treated with AgNPs at 10 mg/kg.
Figure 2. Cumulative biogas volume over time during the solids stabilization process in
9-L bench-scale landfill bioreactors. Lines 1, 2, 3 represent cumulative gas volume in
bioreactors treated with no AgNPs (control), 1mg AgNPs/kg, and 10mg AgNPs/kg respctively.
Phase I, Transition and Acid Formation; Phase II, Hudrogenotrophic Methane Fermentation;
Phase III, Acetoclastic Methand Fermantation; Phase IV, Final Maturation.
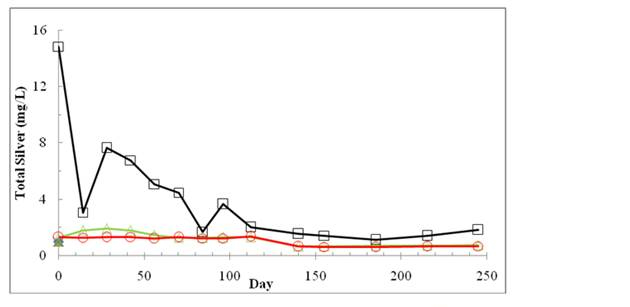
As shown in Figure 3, the total silver concentration in the leachate sample was 14.8 mg/L right after the nanosilver dose at the level of 10 mg AgNPs/kg. Based on mass balance calculation, about 20-30% of total silver was in the leachate at the beginning. The total silver in the leachate generally decreased to below 2 mg/L after 112 days. The total silver concentrations in leachates from the groups of control and 1 mg AgNPs/kg were both maintained at about 1.5 mg/L throughout the study indicating solid wastes, including digested sludge containing silver in the amount much higher than 1 mg Ag/kg added to the bioreactor. From day 140 to the end of our experiments, the total silver concentrations in leachates from the treatments of control, 1 mg AgNPs/kg and 10 mg AgNPs/kg were 0.70 ± 0.05 mg/L, 0.65 ± 0.03 mg/L and 1.46 ± 0.25 mg/L, respectively. The results indicate that silver may be precipitated or absorbed onto solids.
Figure 3. The change of the total silver concentrations in leachates of landfill bioreactors
treated with 0 AgNPs (control)(Ο), 1mg AgNPs/kg (Δ), and 10mg AgNPs/kg (□).
Overall, there were no significant differences in the cumulative biogas volume or gas production rate between the groups of control and 1 mg Ag/kg. However, landfill solids exposed to AgNPs at 10 mg/kg resulted in the reduced biogas production, the accumulation of volatile fatty acids (including acetic acid), and the prolonged period of low leachate pH (between 5 and 6). The results suggest that AgNPs at the concentration of 1 mg/kg solids have minimal impact on landfill anaerobic digestion, but a concentration at 10 mg/kg or higher inhibit methanogenesis and biogas production from MSW.
3. Nanosilver and Silver Ion Effects on Bioreactor Landfill Operations
To determine the bioavailability of metallic nanoparticles and heavy metals in landfills, we chose silver nanoparticles and silver ions and compared the effects of these two forms of silver on landfill operations. In this experiment lasting for more than 200 days, MSW in six identical lab-scale bioreactors (working volume = 9 L each) was exposed to AgNPs (stabilized with 0.06% polyvinyl alcohol) or Ag+ at the silver concentrations of 10 mg/kg solids. The landfill anaerobic digestion was operated at 37 °C and the cumulative methane production and the changes in chemical and microbial compositions of leachates were determined. There was no significant difference in the cumulative methane volume or methane production rate between the groups of control and 10 mg Ag+/kg solids. However, MSW exposed to 10 mg AgNPs/kg resulted in reduced methane production (by up to 80%) and accumulation of volatile fatty acids (including acetic acid). This was accompanied by higher total Ag concentrations (an average of 3.7 ± 0.3 mg/L) in the leachates after day 132 as compared to those in the groups of control and 10 mg Ag+/kg at 0.7 ± 0.4 and 1.1 ± 0.3 mg/L, respectively. The results suggest that at the concentration of 10 mg/kg, silver ions have minimal impact on landfill methane production. Nanosilver is more toxic than its counterpart resulting in reduced methane production and methanogenic population, likely because of higher silver concentration available in the leachate due to the slow nanosilver dissolution under landfill operations.
4. Changes in Microbial Population Dynamics in Landfill Bioreactors After Nanosilver Exposure
Two classes of acetoclastic methanogens (Methanosaeta and Methanosarcina) that convert acetate into methane and three orders of hydrogenotrophic methanogens (Methanobacteriales, Methanococci and Methanomicrobiales) that convert hydrogen and carbon dioxide into methane play an important role in anaerobic digestion. The Methanococci were not studied because they are often not detected in the digestion systems and mainly exist at high salt concentrations. DNA was extracted from 1.5 mL of leachates from each bioreactor. The qPCR internal standards were prepared from the 16S rRNA gene clones of known methanogenic species. For instance, to quantify the populations of Methanosaeta and Methanosarcina, a 25-μL PCR contained 0.75 μL (each) of the corresponding forward primer and reverse primers (stock concentration of 20 μM), 12.5 μL of SYBR Green PCR master Mix (Applied Biosystems, CA), 6 μL of PCR water, and 5 μL sample DNA. The qPCR reactions were performed starting at 50°C for 2 min, followed by an initial denaturation at 95°C for 10 min, and then 40 cycles of 95°C for 30 s, 60°C for 30 s, and 72°C for 45s. The dissociation step at 95°C for 15 s and 60°C for 1 min was added at the end to check the specificity of the PCR results.
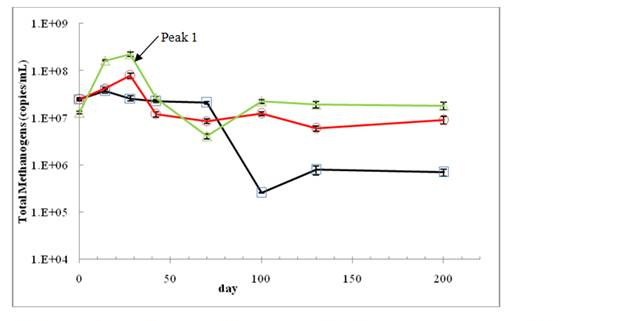
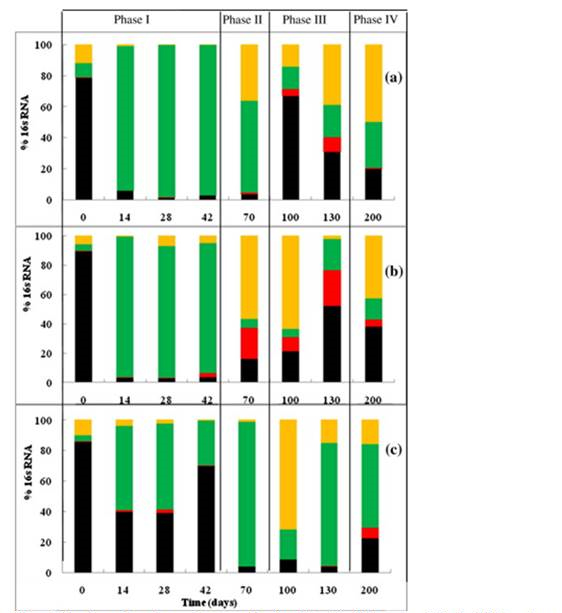
Corresponding to the cumulative gas production profiles (Figure 2), the methanogenic population (Figure 4) and structure of methanogens (Figure 5) also changed during the solids stabilization process. At the beginning of landfill operations, acetoclastic methanogens (Methanosaeta and Methanosarcina) were dominant (80%) in all bioreactors. This ratio represented the methanogenic population in the seed anaerobic sludge from the wastewater treatment plant (data not shown). Just after 14 days of operations, the hydrogenotrophic methanogens (Methanobacteriales and Methanomicrobiales) took the advantage over the acetoclastic methanogens in the control and the 1 mg AgNPs/kg groups, where the fraction of Methanobacteriales was higher than 90%. In contrast, in the bioreactor treated with AgNPs at 10 mg/kg solids, the acetoclastic methanogens Methanosaeta still represented more than 40% of total methanogens, perhaps suffering the lower growth rate of hydrogenotrophic methanogens.
In Phase II and Phase III, hydrogenotrophic methanogens represented 80% to 96% of total methanogens in the bioreactor treated with 10 mg AgNPs/kg. Slowly growing acetoclastic methanogens such as Methanosaeta began to increase. The percent of Methanosaeta population reached the highest on day 100, 130, and 200 for the groups of control, 1 mg AgNPs/kg, and 10 mg AgNPs/kg, respectively (Figure 5). In Phase IV (after day 130), as the acetate was almost exhausted in the groups of control and 1 mg AgNPs/kg treatment, the fraction of acetoclastic methanogens decreased.
Figure 4. The change of the total methanogenic gene copies in the leachates of bioreactors
treated with 0 AgNPs (control)(Ο), 1mg AgNPs/kg (Δ), and 10 mg AgNPs/kg(□). Error bars
represent one standard deviataion of triplicate samples.
Quantitative PCR results after day 100 indicated that the total copy numbers of 16S rRNA gene of methanogens in the groups of control and 1 mg AgNPs/kg were 1.97 ± 0.21 ×107 and 0.90 ± 0.03 ×107, respectively. These numbers were significantly reduced to 5.79 ± 2.83 ×105 (copies/mL) in the bioreactor treated with 10 mg AgNPs/kg.
The project provided some of the first data on the bioavailability and risk assessment of metallic nanomaterials in solid waste disposal systems. Because of the growing concerns of the release of nanomaterials in landfills, the research findings of this project help to better understand the fate and toxicity of nanoparticles to syntrophic anaerobic communities in landfills that could significantly impact the environment and public health.
Figure 5. The changes in methanogenic composition (in relative 16s rRNA
gene copies) in landfill bioreactors treated with zero AgNPs (a), 1mg
AgNPs (b), and 10 mg AgNPs (c). Acetoclastic methanogens: Black,
Methanosaeta; Red, Methanosarcina;, Hudrogenoptophic methanogens:
Green, Methanobacteriales;, Yellow, Methanomicrobiales.
Journal Articles on this Report : 4 Displayed | Download in RIS Format
| Other project views: | All 4 publications | 4 publications in selected types | All 4 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Yang Y, Chen Q, Wall JD, Hu ZQ. Potential nanosilver impact on anaerobic digestion at moderate silver concentrations. Water Research 2012;46(4):1176-1184. |
R833893 (2010) R833893 (Final) |
Exit Exit |
|
|
Yang Y, Xu M, Wall JD, Hu Z. Nanosilver impact on methanogenesis and biogas production from municipal solid waste. Waste Management 2012;32(5):816-825. |
R833893 (2009) R833893 (Final) |
Exit Exit Exit |
|
|
Yang Y, Gajaraj S, Wall JD, Hu ZQ. A comparison of nanosilver and silver ion effects on bioreactor landfill operations and methanogenic population dynamics. Water Research 2013;47(10):3422-3430. |
R833893 (Final) |
Exit Exit |
|
|
Yang Y, Zhang C, Hu ZQ. Impact of metallic and metal oxide nanoparticles on wastewater treatment and anaerobic digestion. Environmental Science: Processes & Impacts 2013;15:39-48. |
R833893 (Final) |
Exit |
Supplemental Keywords:
Bioavailability, cellular, heavy metals, analytical, absorption, adsorption, bioaccumulation, gene expression, growth, mechanisms, metabolism, toxins, nanotechnology, microbiology, qPCR;Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.