Grantee Research Project Results
2008 Progress Report: Manganese, Iron, Cadmium, and Lead Transport from the Environment to Critical Organs During Gestation and Early Development in a Rat Model
EPA Grant Number: R831725C003Subproject: this is subproject number 003 , established and managed by the Center Director under grant R831725
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Health Effects Institute (2015 - 2020)
Center Director: Greenbaum, Daniel S.
Title: Manganese, Iron, Cadmium, and Lead Transport from the Environment to Critical Organs During Gestation and Early Development in a Rat Model
Investigators: Brain, Joseph D. , Wessling-Resnick, Marianne , Molina, Ramon
Institution: Harvard University
EPA Project Officer: Callan, Richard
Project Period: June 1, 2004 through May 31, 2009 (Extended to May 31, 2011)
Project Period Covered by this Report: June 1, 2008 through May 31,2009
RFA: Centers for Children's Environmental Health and Disease Prevention Research (2003) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
In Project 3 (R831725C003), we seek to better understand metal exposures of children and their mothers in settings like Tar Creek by (1) utilizing exposures during and after pregnancy, (2) using metal ions as well as complex environmental samples from Tar Creek, and (3) comparing different routes of entry from the environment into the body. The specific aims for Project 3 (R831725C003) have been expanded. We detail a collaborative experiment with Project 2 (R831725C002), where we look at the solubility and bioavailability of different mineral forms of zinc-containing materials.
This project also now explores the transport of zinc and arsenic, in addition to iron, manganese, and cadmium, which we have previously characterized. We also are studying the effects of exposure to high levels of lead or manganese as they interact with iron transport, fate, and metabolism. These studies are being carried out in collaboration with Project 4 (R831725C004), headed by Dr. Tim Maher. Studies on mechanisms of metal transport through the gut, lungs, and nose remain as major components of our project.
Progress Summary:
i. Studies on Zinc Mineral Particles – Characterization, Neutron Activation, and Pharmacokinetics of Particle-Associated Zinc
We hypothesize that dissolution of metal-containing particles deposited in the nose, gastrointestinal tract or lungs is a critical step influencing their bioavailability. We began with collected “chat” from Tar Creek provided by Project 2 (R831725C002). The combined samples from various sites were size-fractionated into a < 37 μm and < 3.5 μm samples. The samples then were sent to MIT for neutron activation and analyses. We finished pharmacokinetic studies comparing transport and retention of chat-associated metals (59Fe or 65Zn) from < 37 μm and < 3.5 μm particles. We previously found that 59Fe or 65Zn from neutron-activated “chat” particles was absorbed into the blood after intratracheal, intranasal, or gavage administration, and then were retained in various tissues. Our data show that the levels of 59Fe or 65Zn from “chat” in the blood, brain, kidneys, and liver are higher after intratracheal administration than after intranasal or gavage administration. This supports the hypothesis that bioavailability of metals like zinc or iron depends in part on the dissolution of particles. The results also showed that the smaller respirable chat fraction had a generally higher bioavailability for zinc and iron. There also is evidence that a much smaller component of the < 3.5 μm fraction may be absorbed through the lungs into the circulation as intact nanoparticles. The liver uptake for both 59Fe and 65Zn was much higher after intratracheal instillation than after intranasal or gavage administration for the < 3.5 μm chat particles.
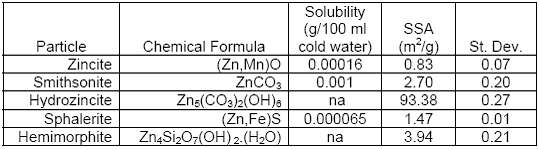
We now extend these studies to determine whether the chemical form of zinc in “chat” previously described in Project 2 (R831725C002) (Schaider LA, et al., Environ Sci Technol 2007;41:4164-4171) influences the bioavailability of zinc from those particles. Schaider, et al. found that in <37 μm weathered “chat” particles collected from several areas at the Tar Creek Superfund Site (Oklahoma), 50-65% of Zn, as well as Pb and Cd were present in the carbonate (exchangeable) fractions. This is in contrast to the originally mined ore at Tar Creek, which primarily consisted of metal sulfides with generally low bioavailability. Therefore, we sought to determine the relative bioavailability of zinc from different pure-phase zinc minerals. To date, no in vivo systematic quantitation of bioavailability of zinc has been reported. We obtained 5 pure-phase mineralogical samples of zinc from the Harvard Mineralogical Museum, Cambridge, MA: smithsonite (ZnCO3), hydrozincite (Zn5(CO3)2(OH)6), hemimorphite (Zn4Si2O7(OH)2(H2O)), sphalerite ((Zn,Fe)S) and zincite ((Zn,Mn)O). The samples were manually ground in a mortar with pestle, then sieved in a 38 μm pore size sieve. The zinc mineral particles were divided into three aliquots. One set was analyzed for specific surface area (SSA) by nitrogen adsorption at the National Institute for Occupational Safety and Health (NIOSH), Morgantown, WV. The results are summarized in Table 3.1. Although all the particles were passed through a 38 μm sieve, the measured SSAs were significantly different. These differences could be due to variations in size distribution and/or surface characteristics among zinc mineral particles. Surface area of particles is thought to influence their dissolution rates, and eventually the bioavailability of metals contained within them.
Table 3.1.
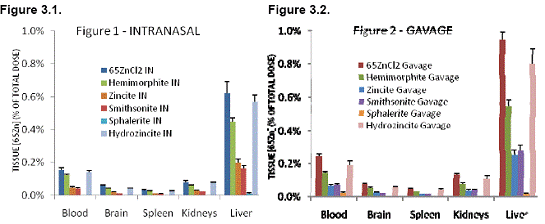
A second set of the zinc mineral particles were also neutron-activated at the MIT Nuclear Reactor Lab. The radioactive particles produced by neutron activation were then used in pharmacokinetic studies to compare the transport and retention of 65Zn from these < 37 μm zinc mineral particles. Soluble 65ZnCl2 was used as a control. Tissue 65Zn levels were analyzed 4 days after 6 dosings over a 3-week period. Two routes of exposure, intranasal and intragastric (gavage) administration, were compared. The results showed that for both intranasal and intragastric route of exposures, the bioavailability of zinc varies among the different mineral forms. Data for selected organs after intranasal or gavage administration are shown in Figures 3.1 and 3.2, respectively. Retained 65Zn in organs such as the brain, spleen, kidneys and the liver were highest from soluble 65ZnCl2. Retained 65Zn from neutron-activated zinc mineral particles were highest from hydrozincite followed, in decreasing order, by hemimorphite, zincite/smithsonite, and then sphalerite.
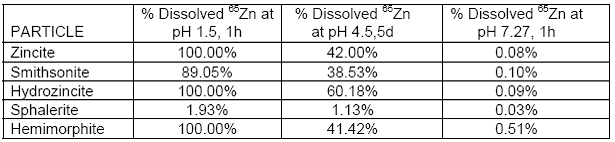
In addition to the in vivo experiments, we also characterize the lability and bioaccesibility of 65Zn in the third set of the same mineralogical samples in physiologically based extraction tests. This was performed to simulate zinc dissolution following ingestion of particles, which was actually tested in our in vivo experiments, or following inhalation, which we did not test since the large particle size (< 37 μm) was very unlikely to be encountered in natural exposure scenarios. Simulated gastric fluid extractions were done in a simple bioaccesibility extraction test (SBET) protocol. A pH 1.5 extraction (1 hour, 37oC) was designed to mimic gastric conditions. It is thought that a rate-limiting step in particulate metal absorption is dissolution in gastric fluids. Similarly, a phagolysosomal simulant fluid (pH 4.5, 5 days, 37oC) was used to simulate metal dissolution within the phagolysosomes of alveolar macrophages. It is thought that metal absorption from inhaled respirable particles occurs following ingestion by alveolar macrophages, residence within phagolysosomes, dissolution of particles and release of soluble metals into alveoli, then absorption into the blood. Finally, we ran similar extraction in a physiologic pH of 7.2 (1 hour, 37oC) to simulate nasal epithelial lining conditions. Results are summarized in Table 3.2.
Table 3.2.
Our data showed that at pH 7.2, an insignificant amount of 65Zn dissolved from any of the zinc mineral particles. It also showed that except for sphalerite, dissolution of 65Zn from the other minerals aselative in vivo bioavailability obtained after gavage in rats, suggesting that several other factors determine bioavailability of zinc from particles, and that SBET protocols alone are not good predictors of metal bioavailability from particles. For example, smithsonite and zincite yielded different SBET dissolution rates, but gave similar tissue retention results in rats. Finally, the simulated nasal epithelial lining conditions yielded very, very low dissolution of particles, and yet nearly equivalent amounts of 65Zn were absorbed and retained in most organs after intranasal dosing as those observed after gavage. In vitro simulated extraction protocols such as SBET by itself have limited application in evaluating potential bioavailabilities of metals associated with particles.
ii. Bioavailability of Zinc – Correlation with Dissolution Rates and Surface Area of Particles
There appears to be a good correlation between the surface area, as determined by nitrogen adsorption, and in vivo bioavailability results. The higher surface areas of particles result in higher zinc bioavailability. The highest surface area measured was for hydrozincite (93.38 m2/g), which correlated with highest concentrations of 65Zn in rat tissues in both intranasal and gavage groups. This is followed by hemimorphite (3.94 m2/g) and smithsonite (2.7 m2/g), which had similar surface areas and tissue 65Zn concentrations. Lower 65Zn concentrations with zincite and sphalerite were consistent with their lower surface areas. However, despite the relatively higher surface area of sphalerite than zincite (1.47 v. 0.83), the 65Zn tissue concentrations were significantly lower in both intranasal and gavage groups. These significant differences are likely due to inherent differences in dissolution rates in the stomach, consistent with our SBET results (Table 3.2).
iii. Studies on “Chat” Particles – Elemental Analysis by Neutron Activation and Pharmacokinetics of Chat-Associated Metals in Rats
We hypothesize that dissolution of metal-containing particles deposited in the nose or lungs is a critical step influencing their bioavailability. We began with collected “chat” from Tar Creek provided by Project 2. The combined samples from various sites were size-fractionated into < 37 μm and < 3.5 μm samples. The samples were then sent to MIT for neutron activation and analyses. For many metals of interest, the concentrations are generally greater in the respirable size range (<3.5 μm) than in the larger fraction. We finished pharmacokinetic studies comparing transport and retention of chat-associated metals (59Fe or 65Zn) from < 37 μm and < 3.5 μm particles. For this experiment, chat samples were neutron-activated using neutron flux of 50x1012 n/cm2s for 120 hours. A suspension of these activated chat particles was then administered intranasally, intratracheally, and by gavage to weanling rats (21-23 days old) twice weekly for 3 weeks. All rats were analyzed at day 42.
59Fe or 65Zn from neutron-activated “chat” particles was transported to the blood after intratracheal, intranasal, or gavage dosing and was retained in various tissues. Our data show that the levels of radioactive zinc or iron from chat in the blood, brain, kidneys, and liver are higher after intratracheal administration than after intranasal or gavage administration (Figure 3.3). This supports the hypothesis that bioavailability of metals such as zinc or iron depends in part on the dissolution of particles. The results also showed that the smaller respirable chat fraction had generally higher bioavailability for zinc and iron.
There also is evidence that much smaller components of the < 3.5 μm fraction may be absorbed through the lungs into the circulation as intact nanoparticles. The liver uptake for both 59Fe and 65Zn was much higher after intratracheal instillation than after intranasal or gavage administration for the < 3.5 μm chat particles. This was also the case with the larger < 37 μm chat particles but not to the same degree.
iv. Mechanisms of Manganese and Iron Absorption from the Lungs and Nose and the Influence of Iron
Status
Manganese transport into the blood can occur following inhalation of metal-containing particles. Intestinal manganese uptake is mediated by divalent metal transporter 1 (DMT1) and is up-regulated by iron deficiency. Since iron status varies significantly within human populations, and DMT1 may mediate metal absorption from the respiratory tract, we tested the hypothesis that iron status may alter absorption of inhaled manganese and iron and explored the potential role of known iron transport proteins in lung metal absorption. Our studies have established that manganese and iron are absorbed by the lungs through different mechanistic pathways. We have also provided the first detailed analysis of the expression pattern and regulation of proteins involved in iron transport in the respiratory and olfactory epithelium. In situ analysis detected DMT1 mRNA in airway epithelium, airway macrophages and bronchus-associated lymphatic tissue (BALT). However, mRNA levels did not change in iron deficient rats. In iron oxide-exposed rats, local increases in DMT1 mRNA transcript were seen in iron oxide particle-containing macrophages and adjacent epithelial cells. These data are consistent with the model that DMT1 levels are differentially regulated in the lungs in response to iron- containing particles to enhance clearance of the metal from the lungs. This pattern of regulation was unexpected since up-regulation of intestinal DMT1 is promoted by iron deficiency. We also identified, for the first time, the expression of DMT1 in olfactory epithelium, and cytolocalized its expression to basal endfeet of sustentacular cells using immunofluorescence microscopy. We studied olfactory absorption of 54Mn in Belgrade rats, an animal model of DMT1 deficiency, and established that transport of intranasally instilled metal from the nasal cavity to the blood was impaired. Moreover, this pathway of manganese absorption was enhanced in anemic rats relative to iron-sufficient controls. These findings are significant because they suggest that neurotoxicity of inhaled manganese may be modified by iron status.
We have also studied the role of DMT1 in neonatal absorption of iron using the Belgrade rat. We found that at time of weaning, impaired DMT1 function in the homozygous pups resulted in reduced absorption of 59Fe in the gut. However, total 59Fe absorption and retention in suckling pups were not different between the homozygous pups and their heterozygous siblings (with intact DMT1 function) when the 59Fe was injected intravenously to the lactating foster dams. Combined, these data suggest that at least during the early developmental period (during lactation), DMT1 function is not essential for iron assimilation from the milk.
v. Maternal Exposures to Mn and Pb Alter Behavior and Iron Metabolism in Weanling Rats
Exposure to manganese (Mn) and lead (Pb) during early development is closely related to impaired learning and cognitive functions, and higher distractibility and impulsiveness including attention deficit hyperactivity disorder (ADHD). Since Mn, Pb and Fe are all transport substrates of divalent metal transporter 1 (DMT1), metal exposure may influence iron status, which is also associated with brain development. Pharmacokinetics and molecular mechanisms of iron transport from the gut in early life from pregnant moms exposed to high levels of Mn or Pb has not been explored. Thus, we wanted to examine the effects of gestational and lactational exposure to Mn or Pb on in vivo kinetics of iron, DMT1 expression in the gut, and neurobehavioral outcomes such as impulsivity and hyperactivity.
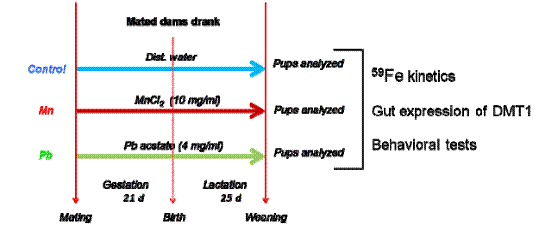
Pregnant female Sprague Dawley rats (3 dams/group) were exposed to MnCl2 (10 mg/ml) or Pb acetate (4 mg/ml) in drinking water from gestational day 1 (GD 1, day of positive sperm detection) and continued through the time of weaning on postnatal day (PND) 24. Distilled water was used as a control. The offspring were culled to 12 per group at age 2 days. The 36 pups/exposure group was randomly divided into groups of 8 (for behavioral studies) or 28 (for iron kinetics studies and other analyses).
We used 59FeCl3 (150 µCi/kg) to study iron kinetics. It was either injected intravenously through the tail vein or administered by gavage. Blood samples were then sequentially taken over a 24-hour period. Plasma and red blood cells were separated. At 4 or 24 hours post-dosing with 59FeCl3, animals were humanely killed and tissues were collected. Radioactivity of tissues, plasma, and red blood cells was measured using a WIZARD 1410 gamma counter. Data were analyzed and expressed as tissue concentration (µCi/g) and % of dose. Duodenal scrapes were collected from each pup for DMT1 expression analyses and blood samples from selected pups for zinc protoporphyrin measurement.
At the times indicated in Figure 3.4, we performed various behavioral tests. Elevated plus maze (EPM) consisted of 2 opposing open arms (31.5 cm x 10 cm) and 2 opposing enclosed arms (31.5 cm x 10 cm x 15 cm). These arms extended from a central platform of 10 cm2. EPM was elevated 37 cm above the floor. The apparatus was painted black. Pups were placed individually in the center of the apparatus facing an open arm and were allowed to freely navigate the maze for a total duration of 5 minutes (300 seconds). Arm entries were counted when more than half the body entered the arm. A video-tracking system (Ethovision Data Acquisition software, Noldus Information Technology, Sterling, VA) was used to record the movement of each pup within the maze. Parameters monitored were latency of the first occurrence to the open arm, frequency of open arm entry, duration in open arm (as seconds and percentage) and average velocity.
Figure 3.4 Experimental Design
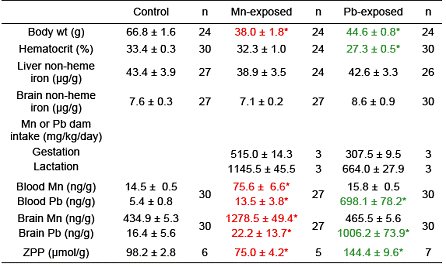
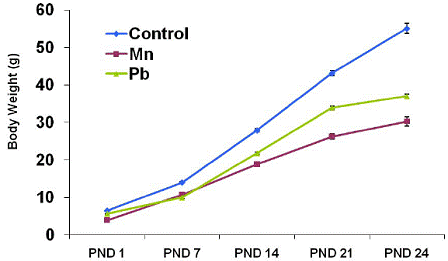
As can be seen in Table 3.3. and Figure 3.5, the body weight of pups exposed to Mn and Pb was lower compared with control pups at every PND determined (P < 0.05). At PND 24, the body weight of pups exposed to Mn and Pb was 45% and 33% lower than that of the control pups, respectively (P < 0.05). Data are mean ± sem (standard error measurement); n = 36 (control), 35 (Mn), 35 (Pb).
Table 3.3 and Figure 3.5 provide information about the consequences of these toxic metal exposures.
Table 3.3. Characteristics of Mn or Pb Gestationally-and Lactationally-exposed 25-d-Old Rat Pups
Figure 3.5. Weight of Pups over Time.
Plasma levels of 59Fe over a 24 h period post-intravenous and post-gavage dosing of pups were measured. Pharmacokinetic analyses of data of both gavage and intravenous data showed that the bioavailability (extent of absorption) and ka (absorption rate constant; the rate of absorption) of 59Fe was lower in both Mn-exposed and Pb-exposed groups, suggesting that exposure to Mn or Pb decreases iron absorption in the gut. The elimination rate constant (fraction eliminated per unit time, ke) of 59Fe in the Mn-exposed group was greater than in the control.
Partition coefficient (a measure of tissue uptake) of 59Fe is a ratio of blood-free tissue [59Fe] to plasma [59Fe]. When injected intravenously, the tissue uptake of 59Fe was significantly reduced in most tissues of Pb-exposed pups but not of the Mn-exposed group. When gavaged, 59Fe tissue uptake was also significantly reduced in some tissues of Pb-exposed pups whereas a higher 59Fe uptake was observed in RBC* of Mn-exposed pups. Mean ± sem; *P < 0.05 compared with control pups.
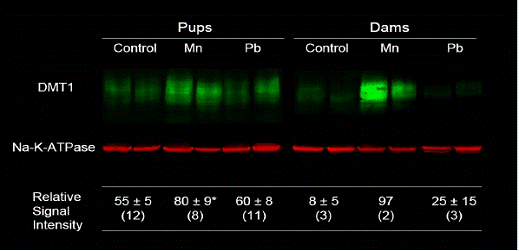
We measured duodenal DMT-1 expression in pups and dams (Figure 3.6). It can be seen that pups exposed to Mn but not to Pb upregulated duodenal expression of DMT-1. Data are mean ± sem (number of rats); *P <0.05 compared with control pups.
Figure 3.6. Duodenal DMT1 Expression in Pups and Dams.
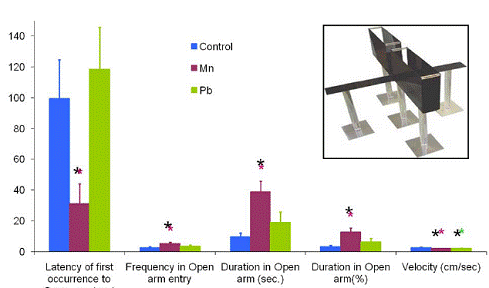
Figure 3.7 gives data obtained from measurements of behavior. Behavioral tests were done on PND 24 (*P <0.05 compared with control). Impulsivity is reflected by shortened latency of first open-arm entry, number of open-arm entries and total duration of time spent in open arms. Higher average velocity reflects greater hyperactivity. Mn-exposed pups (but not Pb-exposed) were more impulsive than control pups. Both Mn- and Pb-exposed pups displayed lower overall velocities when compared with control pups.
Figure 3.7. Behavioral Changes in Rat Pups Gestationally- and Lactationally- exposed to Mn or Pb.
We conclude that pups from dams given Mn or Pb had significantly lower body weight and higher blood and brain concentrations of the respective metal. Zinc protoporphyrin levels were significantly higher in the Pb- exposed group and lower in the Mn-exposed group compared with controls. Intestinal absorption of 59Fe in Pb- or Mn-exposed pups was lower than in controls. Fractional elimination of 59Fe and DMT1 expression in Mn- exposed pups were greater than in controls. Pups exposed to Mn or Pb had lower locomotor activity. Mn- exposed pups, but not Pb-exposed pups, had higher impulsivity. These data indicate that Mn and Pb exposures in early life alter behavior and influence iron metabolism.
Significance
The results of radioactive metal administration studies suggest that the absorbed dose of metals depends on the metal, the route of entry and duration, and indicate that the importance of uptake from the nose and lungs may be underappreciated. These data will be useful in assessing the relative risks for metal toxicity of various exposures to metals. The pharmacokinetic results from chat studies show differences in absorption, vascular kinetics and tissue retention of 59Fe or 65Zn from irradiated chat administered via different routes in rats. Significant differences were also observed as a function of particle size. Smaller, respirable chat had higher metal bioavailability when inhaled or ingested. Data from these studies will be used to assist in estimating the relative risks of metals from different exposures, e.g., eating contaminated food and water, inhaling airborne chat particles, or children playing in contaminated playgrounds. Our studies on molecular mechanisms of metal transport in both normal and mutant Belgrade rats are elucidating mechanistic differences between manganese and iron absorption in the nose, gut and lungs.
Future Activities:
We will complete these studies and write them up during the coming year. We will seek funding to continue studies on pharmacokinetics of and molecular mechanisms of transport of iron from the gut in rat pups from pregnant dams exposed to high levels of manganese or lead in drinking water. We clearly are benefiting from the interactions within the program project. Our data will be correlated with outcomes in both animal (Project 4, R831725C004) and human studies (Project 1, R831725C001). When data from Project 3 (R831725C0003) are combined with exposure assessment in Project 2 (R831725C002), we will be able to better identify which routes of exposure result in the most significant body burdens of toxic metals. From this knowledge, we should be able to craft optimal strategies in contaminated sites like Tar Creek to reduce the doses of toxic metals to mothers and children and thus better respond to the environmental concerns of the citizens in affected communities.
Journal Articles on this Report : 6 Displayed | Download in RIS Format
| Other subproject views: | All 12 publications | 12 publications in selected types | All 12 journal articles |
|---|---|---|---|
| Other center views: | All 63 publications | 46 publications in selected types | All 45 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Brain JD, Heilig E, Donaghey TC, Knutson MD, Wessling-Resnick M, Molina RM. Effects of iron status on transpulmonary transport and tissue distribution of Mn and Fe. American Journal of Respiratory Cell and Molecular Biology 2006;34(3):330-337. |
R831725 (2005) R831725 (2007) R831725 (2009) R831725 (Final) R831725C001 (2007) R831725C003 (2005) R831725C003 (2007) R831725C003 (2008) R831725C004 (2007) |
Exit Exit |
|
|
Heilig EA, Thompson KJ, Molina RM, Ivanov AR, Brain JD, Wessling-Resnick M. Manganese and iron transport across pulmonary epithelium. American Journal of Physiology–Lung Cellular and Molecular Physiology 2006;290(6):L1247-L1259. |
R831725 (2005) R831725 (2007) R831725 (2009) R831725 (Final) R831725C001 (2007) R831725C003 (2005) R831725C003 (2007) R831725C003 (2008) R831725C004 (2007) |
Exit Exit |
|
|
Thompson K, Molina RM, Brain JD, Wessling-Resnick M. Belgrade rats display liver iron loading. Journal of Nutrition 2006;136(12):3010-3014. |
R831725 (2007) R831725 (2009) R831725 (Final) R831725C001 (2007) R831725C003 (2007) R831725C003 (2008) R831725C004 (2007) |
Exit Exit |
|
|
Thompson K, Molina R, Donaghey T, Brain JD, Wessling-Resnick M. The influence of high iron diet on rat lung manganese absorption. Toxicology and Applied Pharmacology 2006;210(1-2):17-23. |
R831725 (2005) R831725 (2007) R831725 (2009) R831725 (Final) R831725C001 (2007) R831725C003 (2005) R831725C003 (2007) R831725C003 (2008) R831725C004 (2007) |
Exit Exit |
|
|
Thompson K, Molina RM, Donaghey T, Schwob JE, Brain JD, Wessling-Resnick M. Olfactory uptake of manganese requires DMT1 and is enhanced by anemia. FASEB Journal 2007;21(1):223-230. |
R831725 (2007) R831725 (2009) R831725 (Final) R831725C001 (2007) R831725C003 (2007) R831725C003 (2008) R831725C004 (2007) |
Exit |
|
|
Thompson K, Molina RM, Donaghey T, Brain JD, Wessling-Resnick M. Iron absorption by Belgrade rat pups during lactation. American Journal of Physiology-Gastrointestinal and Liver Physiology 2007;293(3):G640-G644. |
R831725 (2007) R831725 (2009) R831725 (Final) R831725C001 (2007) R831725C003 (2007) R831725C003 (2008) R831725C004 (2007) |
Exit Exit |
Supplemental Keywords:
children, Native American, tribal, mixtures, lead, PBPK, community, Superfund, intervention, environmental management, environmental management, international cooperation, Scientific Discipline, Waste, Health, RFA, Risk Assessment, Health Risk Assessment, Children's Health, Hazardous Waste, Biochemistry, Environmental Chemistry, Hazardous, iron, neurodevelopmental toxicity, developmental toxicity, fate and transport , children's environmental health, lead, mining wastes, cadmium, human health risk, mining waste, community-based intervention, metal contamination, metal wastes, manganese, metals, Human Health Risk Assessment,, Health, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Waste, ENVIRONMENTAL MANAGEMENT, Hazardous, Health Risk Assessment, Environmental Chemistry, Hazardous Waste, Children's Health, Biochemistry, Risk Assessment, Human Health Risk Assessment, lead, developmental toxicity, metal contamination, neurodevelopmental toxicity, cadmium, mining wastes, fate and transport , animal model, mining waste, community-based intervention, human health risk, manganese, metals, metal wastes, ironProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R831725 Health Effects Institute (2015 - 2020) Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R831725C001 Metals, Nutrition, and Stress in Child Development
R831725C002 Exposure Assessment of Children and Metals in Mining Waste: Composition, Environmental Transport, and Exposure Patterns
R831725C003 Manganese, Iron, Cadmium, and Lead Transport from the Environment to Critical Organs During Gestation and Early Development in a Rat Model
R831725C004 Metals Neurotoxicity Research Project
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
12 journal articles for this subproject
Main Center: R831725
63 publications for this center
45 journal articles for this center