Grantee Research Project Results
2007 Progress Report: Effects of Nanomaterials on Human Blood Coagulation
EPA Grant Number: R832843Title: Effects of Nanomaterials on Human Blood Coagulation
Investigators: Perrotta, Peter L. , Gouma, Pelagia-Irene
Institution: West Virginia University , The State University of New York at Stony Brook
EPA Project Officer: Hahn, Intaek
Project Period: February 1, 2006 through January 31, 2009
Project Period Covered by this Report: February 1, 2007 through January 31,2008
Project Amount: $375,000
RFA: Exploratory Research: Nanotechnology Research Grants Investigating Environmental and Human Health Effects of Manufactured Nanomaterials: A Joint Research Solicitation - EPA, NSF, NIOSH (2005) RFA Text | Recipients Lists
Research Category: Safer Chemicals , Nanotechnology
Objective:
Determine the effects of nanomaterials on the blood coagulation system.Progress Summary:
- Dispersed nanoparticles in biological systems
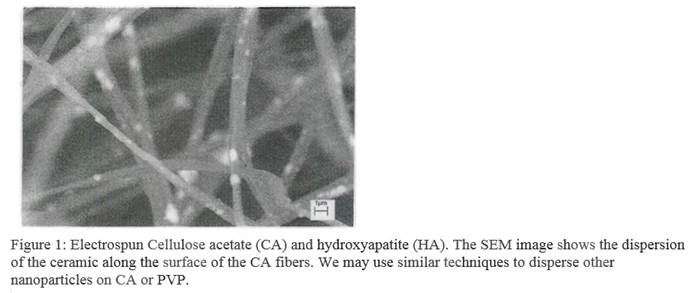
The implications of many nanotoxicology studies are difficult to interpret because investigators often do not adequately document the dispersion of the nanoparticles within their experimental system. In collaboration with Dr. Darran Cairns of the Mechanical Engineering Department at WVU, we have prepared dispersions of metal oxides down to the 10-20 nm range. Following preparation, the particles remain dispersed when the pH of the media is reduced toward physiologic ranges, and the average particle size does not change over several days as determined by dynamic light scattering (DLS). We found that DLS was the most rapid and reproducible means we had available to document particle size in solution. Other techniques such as atomic force microscopy were less reliable because the samples must by dried before analysis. We are also exploring the use of electrospinning for preparing nanoparticle dispersions (Figure 1).
- Cell interactions with nanostructured hydroxyapatite (BA) particles dispersed on cellulose acetate (CA) fibers
These experiments were carried out in collaboration with Berkeley Lab's Molecular Foundry. Human osteoblasts (SaOS-2) cells were used to assess their interactions with Cellulose Acetate/Hydroxyapatite (CA/HA) electrospun mats. 15 mm circular mats in 24-well plates in triplicates were seeded with cells. SaOS2 cells were cultured in Dulbeccos modified Eagles medium under standard culture conditions. For the cell attachment and proliferation study on plain CA and CA/HA mats, SaOS-2 cells were seeded at a density of 50,000 cells/well for MTS assay on the mats and empty wells as controls in triplicates. The cultures were placed at 37oC in a humidified atmosphere containing 5% C02 for l day and 3 days.
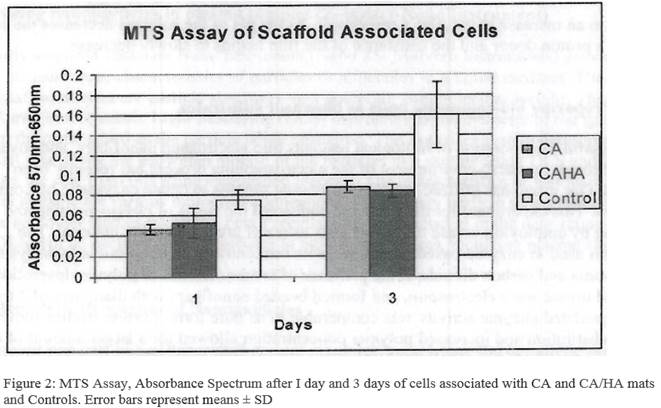
The Promega CellTiter 96® Non-Radioactive Assay is used for the metabolic activity and proliferation of SaOS-2 cells on CA and CA/HA. This assay uses the tetrazolium compound 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium; MTS. MTS is chemically reduced by cells into formazan product. Since the production of formazan is proportional to the number of living cells, the intensity of the produced color is a good indication of the viability of the cells. The measurement of the absorbance of the formazan was carried out using 96 well plates following the large volume overnight assay protocol following generation of a standard curve for the MTS assay. Six dilutions (78,500, 15,700, 19,625, 1,570, 157 and 15.7 cells/mL were prepared from the original 157,000 cells/mL. 1.0 mL of each cell suspension was seeded by transferring the cell dilutions into each well of a 24-well tissue culture plate. Each dilution was performed in three replicates. Then, 150 µL of the Dye Solution was added to each suspension. The plate was placed at 37oC in a humidified atmosphere containing 5% CO2 in dark. After 4 hours, 1.0 mL of Solubilization/Stop Mix was added to each well. Plate was sealed and kept at 37oC in a humidified atmosphere containing 5% CO overnight. 100 µL of the solutions from each well was transferred to a 96-well plate and absorbance was read at 570 nm wavelength and also at 650 nm as a reference wavelength using a Molecular Devices SpectraMax Spectrophotometer.
The cells were observed in control samples and on the mats after l day and 3 days (Figure 2). Significantly large differences in the number or growth rate of cells on mats vs. plastic well controls were not observed in these experiments. This indicates that the mats do not have a detrimental effect on cellular growth and proliferation. One might expect to see larger differences in favor of the mats in longer term experiments once the cells had time to exceed the growth area of the plastic control wells. Because of the low density of fibers, CA mats (consisting of belt shaped fibers) allowed the cells to pass through them and settle on the plastic bottom of the well during initial seeding. The CA/HA mats had fibrous consistency and retained more cells. Microscopy studies are currently under way to observe the nature of specific cell-HA interactions.
-
Conductimetric measurement of cation concentration in solutions using electrospun PANI composites as sensing matrices
One of our major focuses is in determining if the activity of thrombin, the most physiological important clotting protein, is altered by nanomaterials. Unfortunately, the current technology used to measure thrombin in blood is not ideal because of the relatively low concentrations of thrombin in blood. In addition, the half-life of thrombin is very short. Thus, we are using nanotechnology to develop sensors that are more capable of measuring thrombin in blood. The principle of operation of one such thrombin biosensor involves the conductimetric measurement of NH2 cations in analyte solutions by electroactive polymers, such as polyaniline (PANI) composites. The first step towards achieving this goal is to demonstrate that H+ concentration (i.e., pH changes) may be monitored accurately and reproducibly by using electrospun cellulose acetate and emeraldine salt (ES) polyaniline composites. The PANI concentration used was 20% and 50% in the electrospun matrix. The films were bonded using silver paste to gold wires. The distance between the gold electrodes was 2-3 mm. The films were immersed in a solution of 100 mL milieu water with 5 µL of pH ISA solution to increase and stabilize the pH of the solution to ~10. A Ross pH reference electrode was also immersed in the solution to monitor changes in pH. The resistance of the electrospun composite was measured using a Keithley high resistance multimeter. The pH of the solution was varied by adding 0.1-0.2 mL of acetic acid solutions 0.025%, 0.05%, 0.1%, and 0.2%. It was shown in our work that the 80-20 CA/PANI hybrid is more sensitive to the change in [H+] in the solution than the 50-50 composition. When acetic acid reacts with water the following reaction occurs:
Since acetic acid is monoprotic, for every unit of acetic acid, only one H+ is liberated. Water arranges itself around this proton forming an acidic molecule. As the pH of the solution decreases, the concentration of H+ increases in the solution. Above pH 9, polyaniline is deprotonated by the basic solution (considered a proton acceptor) due to the excess CH3COO-.
Future Activities:
- Incorporate nanoparticles in scaffolds (to minimize particle agglomeration) and assess platelet/smooth muscle cell growth in vitro
To date, we have confirmed the efficient dispersion of inorganic nanoparticles (i.e., HA) in cellulose acetate fibrous mats by means of electrospinning processing. We have also carried out cell culture studies using osteoblasts to assess the effect of (HA) nanoparticles on this type of cells. The next steps include: (1) dispersing commercial nanoparticles on CA fibrous mats; (2) performing in vitro studies using platelets and/or smooth muscle cells to explore nanoparticle-blood cell interactions (using the WVU facilities); (3) performing characterization studies (electron microscopy of nanoparticle-platelet and platelet-platelet interactions); and (4) assessing the role of particle size, shape, and surface characteristics (electrochemical charge, redox properties).
- Coagulation studies of rats exposed to nanoparticles through inhalation
We have developed a collaboration with Dr. Timothy Nurkewicz at WVU, who is using an inhalation model to study the vascular system of rats exposed to particles in the nanosized range. Blood is collected from the jugular vein to minimize clotting protein and platelet activation from control and exposed rats into sodium citrate (0.105 M, 3.2%) as the anticoagulant. This is important because blood is usually collected into EDTA, which is not useful for clotting studies. Platelet-poor plasma was removed using standard centrifugation protocol at 4°C and aliquots were stored at -80°C until use. Thrombin generation was measured using fluorogenic thrombin substrate (Benzoyl-FVR-AMC) and the Multimode Detector. Thrombin standards curves were first generated in order to quantify the amount of thrombin generated.
- Influence of Nanomaterials on Fibrinogen (FBG) Activity
Fibrinogen (FBG) plays a major role in coagulation, and both elevated and decreased levels have clinical significance. Elevated plasma FBG has been identified as an independent risk factor for coronary atherosclerosis and ischemic heart disease.Individuals with congenital absence of FBG, termed afibrinogenemia, have prolonged bleeding times. A Micro-plate Colorimetric ELISA Method was used to measure Fibrinogen levels in rat plasma samples. Fibrinogen Standard Curve was generated according to the protocol which facilitated the quantification of fibrinogen level in the samples. These studies are ongoing.
- Correlating changes in coagulation with cardiac marker status and inflammatory markers (Luminex Assays)
Inflammatory mechanisms play a vital role in the initiation, maintenance and progression of vascular diseases leading to coronary artery disease. Rat Multiplex panels (Millipore Corp) are available for the study of the pathogenesis of CVD. These panels are designed for the simultaneous analysis of multiple cardiac biomarkers reflecting oxidative stress, inflammation and vascular or cardiac integrity. Analytes tested include: fibrinogen, IL-6, MCP-1, MPO, Tnl-1, TnT-1, TNF-alpha, VEGF, and vWF. The latter is of particular importance to dotting and platelet responsiveness. Currently, we have plans to expand Luiminex assays and do parallel studies with citrate rat plasma samples in the future. This will show the effect of nanoparticles, if any, on the anticoagulant itself.
- Develop cell-based nanotoxicology assays to assess platelet cytotoxicity and platelet activation
Our initial focus has been on blood coagulation assays. Our next step is to modify our platelet assays so that we can assess the effects of nanomaterials on blood platelets. Specific areas that will be examined include the production of reactive oxygen species (ROS) and activation of apoptotic mechanisms. Because of the difficulties many groups have had with certain assays of ROS activation because of interference by nanoparticles, we are exploring the use of ESR spin trapping as an alternative in collaboration with Dr. Val Vallyathan at NIOSH. This facility has state-of-the-art equipment and extensive experience in this technique. Preliminary studies on platelet activation in a mouse model of nanoparticle exposure have been performed in collaboration with Dr. Petia Simeonova at NIOSH. Her group has performed extensive evaluations of nanoparticle toxicity using this model, but they have not focused on the blood coagulation system. However, we will continue to primarily focus on human platelets, using an in vitro model of nanoparticle exposure.
- Electrospin composite materials for nanostudies
Efforts will continue to develop nanosensors for thrombin (and other protein) measurement based on some of the work described thus far. We have designed a nanosensor that will detect low levels of thrombin that should overcome some of the limitations in the techniques currently used to measure this key clotting enzyme. This sensor detects the thrombin-catalyzed hydrolysis of a specific thrombin substrate (Benzoyl-Phe-Val-Arg-AMC, HCl) by using a conducting polymer that measures changes in electrical resistance in the presence of ions. Such a sensor would be useful in measuring small changes in thrombin that occur in a variety of human diseases, both in the blood, and at the surface of cells and blood vessels. Alternative designs for thrombin blood coagulation sensors are being explored in collaboration with Dr. Nick Wu at WVU that utilize aptamer-labeled quantum dots and impedance-based detection.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 22 publications | 5 publications in selected types | All 4 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Gadre SY, Gouma PI. Biodoped ceramics: synthesis, properties, and applications. Journal of the American Ceramic Society 2006;89(10):2987-3002. |
R832843 (2007) R832843 (Final) |
Exit |
|
|
Gouma PI, Ramachandran K. Electrospinning for bone tissue engineering. Recent Patents on Nanotechnology 2008;2(1):1-7. |
R832843 (2007) R832843 (Final) |
Exit |
Supplemental Keywords:
Nanotoxicology, coagulation, proteins, thrombosis, nanomaterials, biosensors, biocomposites, thrombin, Health, Scientific Discipline, ENVIRONMENTAL MANAGEMENT, Health Risk Assessment, Risk Assessments, Environmental Microbiology, Biochemistry, Risk Assessment, blood clotting, bioavailability, nanotechnology, blood coagulation enzymes, nanomaterials, nanoparticle toxicity, analysis of chemical exposureRelevant Websites:
Materials Science & Engineering at Stony Brook ExitProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.