Grantee Research Project Results
2008 Progress Report: Cardiovascular Responses in the Normative Aging Study: Exploring the Pathways of Particle Toxicity
EPA Grant Number: R832416C001Subproject: this is subproject number 001 , established and managed by the Center Director under grant R832416
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Health Effects Institute (2015 - 2020)
Center Director: Greenbaum, Daniel S.
Title: Cardiovascular Responses in the Normative Aging Study: Exploring the Pathways of Particle Toxicity
Investigators: Schwartz, Joel , Suh, Helen H. , Sparrow, David , Vokonas, Pantel
Institution: Harvard University
EPA Project Officer: Chung, Serena
Project Period: October 1, 2005 through September 30, 2010 (Extended to September 30, 2011)
Project Period Covered by this Report: August 1, 2007 through July 31,2008
RFA: Particulate Matter Research Centers (2004) RFA Text | Recipients Lists
Research Category: Human Health , Air
Objective:
In our original EPA-funded Particle Center, we examined air pollution mediated responses of individuals participating in the Normative Aging Study (NAS), a large prospective cohort living in Eastern Massachusetts. As part of this effort, we collected ECGs and blood samples from each study participant and analyzed these samples for HRV and CRP, respectively. In analyses of these data, we found ambient PM2.5 and ambient black carbon (BC) concentrations to be associated with decrements in HRV, with these decrements greatest for hypertensive individuals. Ambient BC concentrations were further found to be associated with increased CRP and fibrinogen levels. These results suggest that the PM-mediated autonomic changes may be brought about through pathways involving the autonomic nervous system and systemic inflammation. Definitive identification of PM-mediated biological mechanisms was limited, however, by the lack of other intermediate cardiac and inflammation endpoints, the use of central site monitoring to characterize exposures for the entire cohort, and by the traditional epidemiologic approaches used to examine exposure-effect associations.
In Project 1 of our current Center, we are continuing our analysis of the NAS cohort, with continued ECG, CRP and fibrinogen measurements and importantly with additional exposure and health measurements for each NAS participant to enhance our ability to identify important biological pathways. These additional measurements include ECG, blood inflammatory markers, medication, genotypic, food frequency, and particle exposure measurements for each of the current NAS participants. ECG and blood samples are being analyzed for a variety of measures (HRV, ST segments, QT intervals, CRP, sICAM-1, sVCAM-1, and homocysteine); these measures will be used as intermediate markers of the inflammatory, endothelial, and autonomic pathways. In addition, they will be related to individual-specific indoor PM2.5, SO42-, and BC exposures that are being measured for one week prior to the clinic visit and to ambient air pollution (PM2.5, PM10, PM2.5-10, SO42, NO3-, BC, EC, OC, and PC) concentrations that are being measured at our stationary ambient monitoring (SAM) site. The study will use these data to test three primary hypotheses:
Hypothesis 1: Cardiovascular effects of particles (PM) will differ by source and by different source-related components. Specifically, short-term exposures to sulfate and traffic particles will be associated with increases in:
- acute inflammation and/or endothelial dysfunction, as measured by increases in CRP, soluble intercellular adhesion molecule 1 (sICAM-1), and soluble vascular cell adhesion molecule 1 (sVCAM-1);
- autonomic dysfunction, as measured by reduced heart rate variability (HRV) and;
- general cardiovascular responses, as measured by increases in blood pressure and ECG changes including ST-segment level and QT-interval.
Hypothesis 2: Effects of PM on these outcomes will be modified by subject characteristics (genetic, dietary, or pharmacological) that influence susceptibility to:
- oxidative stress, endothelial dysfunction, and/or acute inflammation, specifically Glutathione-s-trasferase (GSTM1) null or the long repeat Hemeoxygenase-1 (HO-1) genotypes; statin, beta blocker, or calcium channel blocker use; dietary intake of Vitamin C or omega-3 (Ω-3) fatty acids;
- autonomic dysfunction, specifically beta blocker use, calcium channel blocker use or dietary intake of Ω-3 fatty acids;
- general cardiovascular disease, specifically hypertension and
- reactive airways disease, specifically methacholine reactivity.
Hypothesis 3: Long-term exposure to PM from traffic is associated with increased risk of inflammation (e.g., CRP, sICAM-1, sVCAM-1, and homocysteine); autonomic dysfunction (e.g., reduced HRV), and impaired cardiovascular outcomes (e.g., elevated blood pressure). This association is modified by the same factors that modify acute responses.
Progress Summary:
We have made substantial progress in our NAS study, both in the analysis of data previously collected as part of our original Harvard-EPA Particle Center and in the collection of new data for our current Harvard-EPA Particle Center. This progress is discussed briefly below.
Analysis of Previously Collected Data
In Year Three, we continued our analysis and interpretation of data collected on the NAS participants as part of our original Harvard-EPA Particle Center. These analyses focused on the relation between ambient pollution and intermediate pulmonary (pulmonary function) and cardiac (HRV) outcomes and on how these relationships vary with individual-specific factors, such as genetic polymorphisms, weight, and airway hyper-reactivity (AHR). Results from these analyses have helped to identify important PM-mediated biological pathways and susceptible populations. Findings from these analyses have been presented at conferences and have been submitted or published in peer-reviewed journals, with representative papers discussed briefly below.
- Relation Between PM and Cardiac Function: Importance of Oxidative Stress Pathways
We published two papers this year that identified factors that modify the impacts of PM on cardiac function. Our paper by Chahine et al. (2007) extends our work on the oxidative stress pathway to examine polymorphisms in hemeoxygenase (HMOX-1). Polymorphisms in HMOX-1 have been shown to vary its ability to respond to oxidative stress, with a high number of (GT)n dinucleotide repeats in the 5'-flanking region of the gene related to reduced response and to increased risk of coronary artery disease in high-risk groups with hyperlipidemia, diabetes or current smoking. Consistent with these findings, our paper shows that individuals with a high number of (GT)n repeats are also more susceptible to the effects of airborne particles. We found, for example, that PM2.5 was not associated with decreased heart rate variability (HRV) in subjects with the short repeat variant of HMOX-1, but was significantly associated in subjects with any long repeat. We also found that HMOX-1 interacts with GSTM1 and PM2.5 in predicting HRV. In subjects with the GSTM1 deletion and the HMOX-1 long repeat, high frequency (HF) power of HRV decreased by 28% (95% CI -8, -43) per 10 µg/m3 increase in PM2.5.In a related paper by Park et al. (2008), we show that our elderly NAS individuals with long-term exposure to higher levels of lead may be more sensitive to cardiac autonomic dysfunction on high air pollution days. We found graded, significant reductions in both HF and low-frequency (LF) powers of HRV in relation to ozone and sulfate across the quartiles of tibia lead. Also interquartile range increases in ozone were associated with a 38% decrease (95% CI: -54.6% to -14.9%) in HF and a 38% decrease (-51.9% to -20.4%) in LF in the highest quartile of tibia lead after controlling for potential confounders.
- Relation Between Lung Function and Air Pollution: Oxidative Stress, Genes and Other Modifying Factors
In three papers, we investigated the impact of air pollution on lung function in our NAS participants. Together, these papers provide important evidence that oxidative stress is a key pathway by which ozone causes damage. The first of these papers investigated the effect of statins, an anti-inflammatory and antioxidant medication, on decline in lung function in the elderly and whether smoking modified this effect (Alexeeff et al., 2007a). We found that statin use attenuates decline in lung function in the elderly, with the size of the beneficial effect modified by smoking status. For those not using statins, the estimated decline in lung function (as assessed using FEV1) was 23.9 ml/year (95% CI: 227.8, 220.1 ml/yr), whereas those taking statins had an estimated 10.9-ml/year decline (95% CI: 216.9, 25.0 ml/yr). This paper suggests that statin use may lessen the impact of exposures to pollutants that generate oxidative stress and inflammation, such as PM and ozone.This suggestion was examined in part in a second paper (Alexeeff et al., 2007b), in which we found that acute ozone exposures are associated with decrements in lung function in our elderly NAS participants. This finding is contrary to the previous belief that the elderly are not responsive to ozone exposures. This paper further found that ozone has the greatest effect on lung function in elderly who are obese or have AHR. For example, a 15 ppb increase in O3 during the previous 48 hours was associated with a greater decline in FEV1 in the obese (-2.07%, 95% CI: -3.25%, -0.89%) than in the non-obese (-0.96%, 95% CI: -1.70%, -0.20%).
We explored the relation between ozone and pulmonary function further in a third paper by Alexeeff et al. (2008). In this paper, we showed that the effects of ozone on lung function in the elderly are modified by the presence of specific polymorphisms in antioxidant genes (HMOX1 and glutathione S-transferase pi [GSTP1]). Specifically, we found that a 15 ppb increase in O3 during the previous 48 hours was associated with a 1.25% decrease in FEV1 (95% CI: -1.96%, -0.54%). This estimated effect was worsened with either the presence of a long (GT)n repeat in HMOX1 (-1.38%, 95% CI: -2.11%, -0.65) or the presence of an allele coding for Val105 in GSTP1 (-1.69%, 95% CI: -2.63%, -0.75). A stronger estimated effect of O3 on FEV1 was found in subjects carrying both the GSTP1 105Val variant and the HMOX1 long (GT)n repeat (-1.94%, 95% CI: -2.89%, -0.98%).
- PM-Mediated Toxicity: Metabolic Pathways
In two papers, we investigated the role of the methionine cycle, which is involved in the control of biological processes such as methyl-group transfers, homocysteine synthesis, and redox states, and which may be involved in PM-mediated toxicity. The activity of the methionine cycle is dependent on availability of dietary methyl nutrients and is modified by genetic variations in metabolic genes. In particular, the CT and TT genotypes of the C677T methylenetetrahydrofolate reductase (MTHFR) polymorphism have been associated with reduced enzyme activity, and linked, though not consistently, with increased risk of cardiovascular diseases. Conversely, the CT and TT genotypes of the C1420T cytoplasmic serine hydroxymethyltransferase (cSHMT) polymorphism have been associated with lower homocysteine levels, a methionine metabolism product that may be a risk factor of atherosclerosis, myocardial infarction, stroke, and thrombosis.Baccarelli et al. (2008) specifically examined whether PM-mediated effects on autonomic function are modified by dietary methyl nutrients, such as folate, the B-vitamins pyridoxine (B6), cyanocobalamin (B12), and methionine, and genes related to processing these nutrients. We found the effects of particles on HRV were modified by MTHFR and dietary methionine. In carriers of [CT/TT] MTHFR genotypes, for example, HRV (as assessed by SDNN) was 16.3% (95% CI, 25.8-5.6; p=0.004) lower than in CC MTHFR subjects. In the same [CT/TT] MTHFR subjects, each 10 µg/m3 increase in PM2.5 in the 48 hours before the examination was associated with a further 9.9% (95%CI: 17.6, 1.3; p=0.02) decrease in SDNN. The negative effects of PM2.5 were abrogated in subjects with higher intakes (>median) of B6, B12, or methionine.
In Park et al. (2008), we examined whether PM exposures impact a specific part of the methyl group pathway, homocysteine. We found exposures to particles from traffic (black carbon BC, organic carbon OC) to be associated with elevated plasma total homocysteine. No association was observed with sulfate, an indicator of coal and oil combustion particles or PM2.5. The effects of BC and OC were more pronounced in persons with low concentrations of plasma folate and vitamin B12.
Data Collection
In Year Three, we continued to collect information for NAS participants as they came in for their every 3-year health exam. At each exam, we specifically collected ECG, blood inflammatory marker, and daily diet information. We supplemented these measurements with those of 8-OHdG and creatinine in urine, which were added in Year 2 to obtain direct measures of oxidative stress in our participants. In total, we obtained new data (between July 1, 2007 and June 30, 2008) for 147 NAS participants.
Figure shows locations through March 25, 2008; total of 160 samples
In addition to the health measurements, we also collected one-week long indoor PM2.5 104 NAS participants (July 2007 through June 2008). In total, indoor concentrations were measured for 202 participants. In Year Three, 107 individuals completed indoor sampling, with three of these individuals completing indoor monitoring but not having in a clinic visit (one was hospitalized, two did not come in for their visit). Forty-three participants with a clinic visit did not participate in indoor monitoring, The reasons for non-participation were varied, including lived out of state (7 participants), declined toparticipate (14), clinic visit scheduled too late to send monitor (13), lived in smoking household (4), received but forgot to operate indoor monitor (2), excessive monitor noise (1), technician error (1), and unable to contact (1). For both clinic visits and indoor monitoring, our participation rate was high and was greater than we had anticipated from our last year’s visit number. Of those that participated, subjects generally lived within the Route 495 beltway, although several participants also lived in southern Massachusetts, southern New Hampshire and Rhode Island (Figure 1).
Laboratory Analysis
We continued to analyze collected health data, with laboratory analysis of HRV, blood and urine samples conducted in the appropriate laboratories. ECG tapes were analyzed using software from Forest Medical for ST segments, arrhythmias, and QT length. Indoor PM2.5 samples have been analyzed for mass and reflectance.
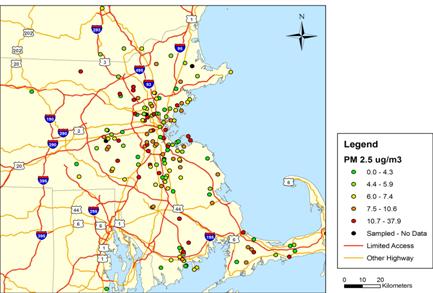
Figure 1. Indoor PM2.5 Concentrations
Figure shows locations through March 25, 2008; total of 160 samples.
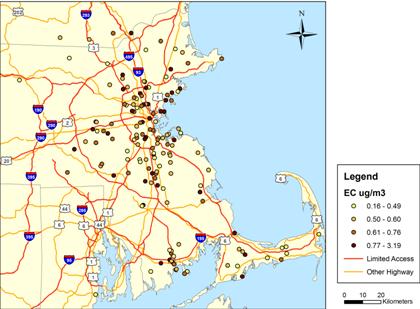
Figure 2. Week-Long Indoor BC Concentrations
Figure shows locations through March 25, 2008; total of 155 samples.
Indoor Concentrations
One-week integrated indoor PM2.5 concentrations varied substantially, with values ranging between zero and 37.9 ug/m3 (Figure 2). Week-long indoor BC concentrations (as estimated using reflectance) also ranged widely (Figure 3). The minimum and maximum concentrations equaled 0.16 and 3.19 ug/m3, respectively. Indoor PM2.5 concentrations were generally lower than outdoor levels measured at our stationary ambient monitoring (SAM) site located in downtown Boston , on the roof of Countway Library, with lower mean and median concentrations (Figure 4). Lower indoor levels suggest the presence of few PM2.5 sources inside our NAS participant homes. The association between indoor and outdoor PM2.5 concentrations was weak, with an R2 of only 0.09 (Figure 5). Observed variation in indoor PM2.5 and BC concentrations and its relation to corresponding outdoor concentrations likely reflects temporal variation, as samples were collected over different time periods. It also likely reflects variation in home-specific factors, such as distance to road and home ventilation. The impact of these factors is currently being examined in our analysis of indoor concentrations.
Figure 3. Week-Long Indoor, Outdoor PM2.5

Data collected July 2006-Sept. 2007
Figure 4. Week-Long Indoor vs. Outdoor PM2.5
Includes data collected between July 2006-Sept. 2007
Future Activities:
We are continuing to collect HRV, blood and urine samples at each participant’s health visit and to recruit participants for indoor sampling. For Study Year 4, we anticipate that 120 subjects will be seen at their NAS health appointment, with approximately 60% of these individuals participating in the indoor monitoring component of the study. This number is lower than previous years, due to attrition from death, illnesses, and general age-related issues. For participants that have moved since our last address geocoding, we will update residential addresses and geocodes.
In addition, we will continue to analyze health and exposure data using statistical methods. Laboratory analyses will include analysis of ECG, blood and urine markers discussed above. We plan to begin our statistical analyses of PM exposures and ST, arrhythmias, QT length, 8-OHgD, and ICAM and VCAM data. We will characterize our indoor PM2.5 and BC concentrations and will predict indoor PM2.5 levels from outdoor levels, location-specific data, housing characteristics, and GIS-based spatial models. Once complete, indoor predictions will be incorporated into our health analyses.
Journal Articles on this Report : 19 Displayed | Download in RIS Format
| Other subproject views: | All 68 publications | 67 publications in selected types | All 67 journal articles |
|---|---|---|---|
| Other center views: | All 206 publications | 199 publications in selected types | All 199 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Alexeeff SE, Litonjua AA, Suh H, Sparrow D, Vokonas PS, Schwartz J. Ozone exposure and lung function: effect modified by obesity and airways hyperresponsiveness in the VA Normative Aging Study. Chest 2007;132(6):1890-1897. |
R832416 (2006) R832416 (2009) R832416 (Final) R832416C001 (2008) R832416C001 (2009) R832416C001 (Final) R827353 (Final) |
Exit Exit Exit |
|
|
Alexeeff SE, Litonjua AA, Sparrow D, Vokonas PS, Schwartz J. Statin use reduces decline in lung function: VA Normative Aging Study. American Journal of Respiratory and Critical Care Medicine 2007;176(8):742-747. |
R832416 (2006) R832416 (Final) R832416C001 (2008) R832416C001 (Final) R827353 (Final) |
Exit Exit Exit |
|
|
Alexeeff SE, Litonjua AA, Wright RO, Baccarelli A, Suh H, Sparrow D, Vokonas PS, Schwartz J. Ozone exposure, antioxidant genes, and lung function in an elderly cohort:VA Normative Aging Study. Occupational and Environmental Medicine 2008;65(11):736-742. |
R832416 (Final) R832416C001 (2008) R832416C001 (Final) R827353 (Final) |
Exit Exit |
|
|
Baccarelli A, Cassano PA, Litonjua A, Park SK, Suh H, Sparrow D, Vokonas P, Schwartz J. Cardiac autonomic dysfunction: effects from particulate air pollution and protection by dietary methyl nutrients and metabolic polymorphisms. Circulation 2008;117(14):1802-1809. |
R832416 (2006) R832416 (Final) R832416C001 (2008) R832416C001 (Final) R827353 (Final) |
Exit Exit Exit |
|
|
Baccarelli A, Martinelli I, Zanobetti A, Grillo P, Hou L-F, Bertazzi PA, Mannucci PM, Schwartz J. Exposure to particulate air pollution and risk of deep vein thrombosis. Archives of Internal Medicine 2008;168(9):920-927. |
R832416 (2006) R832416 (Final) R832416C001 (2008) R832416C001 (Final) R827353 (Final) |
Exit Exit |
|
|
Chahine T, Baccarelli A, Litonjua A, Wright RO, Suh H, Gold DR, Sparrow D, Vokonas P, Schwartz J. Particulate air pollution, oxidative stress genes, and heart rate variability in an elderly cohort. Environmental Health Perspectives 2007;115(11):1617-1622. |
R832416 (2006) R832416 (2008) R832416 (2009) R832416 (Final) R832416C001 (2008) R832416C001 (2009) R832416C001 (Final) R827353 (Final) |
Exit Exit |
|
|
Franklin M, Schwartz J. The impact of secondary particles on the association between ambient ozone and mortality. Environmental Health Perspectives 2008;116(4):453-458. |
R832416 (2006) R832416 (2008) R832416 (Final) R832416C001 (2008) |
|
|
|
Franklin M, Koutrakis P, Schwartz J. The role of particle composition on the association between PM2.5 and mortality. Epidemiology 2008;19(5):680-689. |
R832416 (2006) R832416 (2008) R832416 (Final) R832416C001 (2008) |
Exit |
|
|
Gryparis A, Coull BA, Schwartz J. Controlling for confounding in the presence of measurement error in hierarchical models: a Bayesian approach. Journal of Exposure Science & Environmental Epidemiology 2007;17(Suppl 2):S20-S28. |
R832416 (2008) R832416 (Final) R832416C001 (2008) |
Exit Exit |
|
|
McCracken JP, Smith KR, Diaz A, Mittleman MA, Schwartz J. Chimney stove intervention to reduce long-term wood smoke exposure lowers blood pressure among Guatemalan women. Environmental Health Perspectives 2007;115(7):996-1001. |
R832416C001 (2008) R827353 (Final) |
|
|
|
Medina-Ramon M, Schwartz J. Temperature, temperature extremes, and mortality:a study of acclimatisation and effect modification in 50 US cities. Occupational and Environmental Medicine 2007;64(12):827-833. |
R832416C001 (2008) R827353 (Final) |
Exit Exit Exit |
|
|
Medina-Ramon M, Goldberg R, Melly S, Mittleman MA, Schwartz J. Residential exposure to traffic-related air pollution and survival after heart failure. Environmental Health Perspectives 2008;116(4):481-485. |
R832416 (2006) R832416 (2008) R832416 (Final) R832416C001 (2008) R832416C001 (Final) |
Exit |
|
|
Medina-Ramon M, Schwartz J. Who is more vulnerable to die from ozone air pollution? Epidemiology 2008;19(5):672-679. |
R832416C001 (2008) R827353 (Final) |
Exit Exit Exit |
|
|
Park SK, O’Neill MS, Vokonas PS, Sparrow D, Spiro III A, Tucker KL, Suh H, Hu H, Schwartz J. Traffic-related particles are associated with elevated homocysteine: the VA Normative Aging Study. American Journal of Respiratory and Critical Care Medicine 2008;178(3):283-289. |
R832416 (2006) R832416 (2009) R832416 (Final) R832416C001 (2008) R832416C001 (2009) R832416C001 (Final) R827353 (Final) |
Exit Exit Exit |
|
|
Park SK, O’Neill MS, Vokonas PS, Sparrow D, Wright RO, Coull B, Nie H, Hu H, Schwartz J. Air pollution and heart rate variability: effect modification by chronic lead exposure. Epidemiology 2008;19(1):111-120. |
R832416 (2006) R832416 (2009) R832416 (Final) R832416C001 (2008) R832416C001 (2009) R832416C001 (Final) R827353 (Final) |
Exit Exit Exit |
|
|
Schwartz J, Sarnat JA, Coull BA, Wilson WE. Effects of exposure measurement error on particle matter epidemiology:a simulation using data from a panel study in Baltimore, MD. Journal of Exposure Science and Environmental Epidemiology 2007;17(Suppl 2):S2-S10. |
R832416 (2008) R832416 (Final) R832416C001 (2008) R832416C001 (Final) |
Exit Exit |
|
|
Schwartz J, Coull B, Laden F, Ryan L. The effect of dose and timing of dose on the association between airborne particles and survival. Environmental Health Perspectives 2008;116(1):64-69. |
R832416 (2006) R832416 (2008) R832416 (Final) R832416C001 (2008) |
|
|
|
Stafoggia M, Schwartz J, Forastiere F, Perucci CA, the SISTI Group. Does temperature modify the association between air pollution and mortality? A multicity case-crossover analysis in Italy. American Journal of Epidemiology 2008;167(12):1476-1485. |
R832416C001 (2008) |
Exit Exit |
|
|
Zanobetti A, Schwartz J. Is there adaptation in the ozone mortality relationship:a multi-city case-crossover analysis. Environmental Health 2008;7:22 (6 pp.). |
R832416 (2006) R832416 (2008) R832416 (Final) R832416C001 (2008) |
Exit Exit Exit |
Supplemental Keywords:
Normative Aging Study, inflammation, autonomic function, oxidative stress,, RFA, Health, Air, Scientific Discipline, Health Risk Assessment, Risk Assessments, particulate matter, Environmental Chemistry, Epidemiology, biological mechanisms, chemical characteristics, autonomic dysfunction, biological mechanism , airborne particulate matter, human exposure, cardiovascular vulnerability, chemical composition, biological pathways, oxidative stress, ambient particle health effects, atmospheric particulate matter, automobile exhaust, toxicology, ambient air quality, human health effects, traffic related particulate matterRelevant Websites:
http://www.hsph.harvard.edu/epacenter/ ![]()
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R832416 Health Effects Institute (2015 - 2020) Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R832416C001 Cardiovascular Responses in the Normative Aging Study: Exploring the Pathways of Particle Toxicity
R832416C002 Cardiovascular Toxicity of Concentrated Ambient Fine, Ultrafine and Coarse Particles in Controlled Human Exposures
R832416C003 Assessing Toxicity of Local and Transported Particles Using Animal Models Exposed to CAPs
R832416C004 Cardiovascular Effects of Mobile Source Exposures: Effects of Particles and Gaseous Co-pollutants
R832416C005 Toxicological Evaluation of Realistic Emission Source Aerosol (TERESA): Investigation of Vehicular Emissions
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2010 Progress Report
- 2009 Progress Report
- 2007 Progress Report
- 2006 Progress Report
- Original Abstract
67 journal articles for this subproject
Main Center: R832416
206 publications for this center
199 journal articles for this center