Grantee Research Project Results
2021 Progress Report: Reducing the reliance on early-life stage testing with relevance to euryhaline fishes: Development and implementation of in-vitro assays predictive of early life stage toxicity and population-level effects in Menidia beryllina
EPA Grant Number: R839503Title: Reducing the reliance on early-life stage testing with relevance to euryhaline fishes: Development and implementation of in-vitro assays predictive of early life stage toxicity and population-level effects in Menidia beryllina
Investigators: Brander, Susanne M , Chappell, Patrick , Armbrust, Kevin , White, Wilson
Institution: Oregon State University , Louisiana State University
EPA Project Officer: Chung, Serena
Project Period: August 1, 2019 through April 23, 2025
Project Period Covered by this Report: August 1, 2020 through July 31,2021
Project Amount: $849,988
RFA: Advancing Actionable Alternatives to Vertebrate Animal Testing for Chemical Safety Assessment (2018) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Over the past decade great strides have been made in minimizing the use of vertebrates for toxicity testing focused on human health outcomes. However, although numerous approaches and models that facilitate the reduction of vertebrates in human toxicity testing currently exist, far fewer alternatives are available for use in ecotoxicological applications, particularly for marine and estuarine ecosystems. For example, the current marine and estuarine species approved for EPA Whole Effluent Toxicity (WET) testing are Cyprinodon variegatus (Sheepshead minnow) and Menidia beryllina (Inland silverside). Neither species has appropriate in vitro alternatives available to limit the use of live animals for regulatory and research purposes. Assessing ecotoxicological effects in estuarine systems is critical, given the importance and value of those ecosystems and their vulnerability to pollution. Further complicating the assessment of toxicity in estuarine systems is the altered behavior of chemicals in seawater, which influences water solubility and concomitant bioavailability and thus raises the potential for differential responses to chemicals dependent on salinity, not to mention the likelihood of differences in osmoregulatory strategies influencing the uptake and metabolism of chemicals between fishes inhabiting fresh and saline waters. For example, fish in saline waters drink constantly to remain isotonic with their surroundings, while freshwater fish urinate frequently to achieve the same goal. Regardless of these challenges, a marine model that uses cell lines to reduce reliance on live fish for the purposes of identifying, prioritizing, and evaluating potential toxicants is greatly needed. Additionally, the development of dosimetry that accurately reflects bioavailability across a salinity gradient would refine this testing, allowing cell lines to be used at a minimum as a first-tier approach to limit the number of toxicants that necessitating in vivo testing.
Progress Summary:
Experimental Approach: We are developing multiple novel in vitro Menidia assays from tissue types (cardiovascular, hepatic, bone) with demonstrated sensitivity in early life across fish species that are complementary to commonly used fish early life stage and larval growth and survival tests. We also detail plans for the generation of dosimetry for mimicking chemical (pesticide) exposures across a salinity gradient in vitro, and the use of genomic tools (RNA seq, high-resolution bioluminescence gene expression monitoring) and demographic modeling in a “middle-out” approach linking in vivo phenotypic anchors (e.g. developmental deformities, altered growth) to biomarker candidates and population-level outcomes. Ultimately this approach would be informative of existing adverse outcome pathways (AOPs) and may also generate novel AOPs. For the purposes of validating our euryhaline in vivo – in vitro linked model, we are conducting exposures with a suite of pesticides from different chemical classes, previously established to be cardiotoxic, hepatotoxic, and osteotoxic in the zebrafish model, and all commonly used in the control of rodents, insects, weeds, or fungus.
Anticipated Results: The major accomplishment of this work will be the creation and validation of a euryhaline in vivo-in vitro linked model with a proposed focus on pesticides already known to affect development in embryonic and larval life stages in freshwater fish, yet with a dearth of information on responses in euryhaline fishes. Ultimately, the approaches developed herein would not only reduce in the number of live fish needed for such testing and improve the efficiency of testing for pesticide registration, but would also reduce the cost and refine the accuracy of risk assessment for other classes of chemicals. This is of mounting importance given the number of new compounds on the market and limited resources available for toxicity testing. We expect to find that most compounds are more toxic at higher salinities, based on results from in vitro and in vivo assays and due to a combination of salting-out of solution and hence becoming more bioavailable, or due differences in metabolism and osmo-regulation.
Progress Summary:
We have conducted and published data from acute exposures (Hutton et al. 2021), intended to be used for range-finding of values for use in sublethal in-vivo exposures, on the Inland Silverside (Menidia beryllina), and eventually in vitro exposures in the cell lines mentioned above that are being developed. Bioconcentration and preliminary sublethal exposures were run at 5 and 25 ppt, since we observed that salinities used for acute exposures may not have been far enough apart to detect a difference in toxicity for some of the biocides. Thus far sublethal in-vivo bioconcentration exposures have been completed for triademefon, but further experiments with fish are on hold while additional bioaccumulation experiments are performed and efforts to lower the limit of detection for analytical confirmation of biocide uptake are explored at LSU. LC25 values derived from this study were initially being used to inform future sub-lethal exposures, but high toxicity was observed with triademefon in fish, especially at higher salinities (25 ppt). Thus we are now working with LSU to lower this to the original goal of the LC10 for each chemical being our highest sublethal concentration.
Analytical Chemistry Summary (LSU)
Method Development : Extraction, Sample Preparation and Analysis
Water Sample Extractions
Due to their low concentrations, the bifenthrin and chlorpyrifos residues were extracted from water samples using C8 solid phase extraction (SPE) cartridges (Thermo Fisher, 500 mg sorbent, 3 mL reservoir volume) placed on a vacuum manifold to capture the analytes. Cartridges were preconditioned with 3 mL of methanol and 3 mL of deionized water prior to adding the sample. Residual pesticide was eluted from the cartridge with 1.5 mL of dichloromethane for both compounds and immediately analyzed by GC/MS/MS. All other compounds did not require solid phase extraction and were analyzed directly by HPLC as received. Extraction recoveries averaged 74% +/- 3.3% (n=5) and 85% +/- 2.43% (n=6) for chlorpyrifos and bifenthrin respectively.
Tissue Extractions
Pesticide residues in fish embryos were extracted using a modified QuEChERS method. Each tissue sample was first transferred to a polypropylene 50 mL centrifuge tube
(VWR). 4.5 milliliters (mL) of a solvent mixture containing 80% ethyl acetate and 20% hexane were then added into each centrifuge tube containing the tissue samples. The samples and extraction solvent were vigorously vortexed (3000 rpm) for 1 min. using a Thermolyne Maxi Mix II. This was immediately followed by sonication for 15 min. using a Branson 2210 sonication bath filled to 2.5 cm with deionized water. The extracts were then removed from the centrifuge tubes and filtered using a 20 µm polytetrafluoroethylene (PTFE) filter. The filtered extracts were transferred to glass vials and blown down with a gentle stream of ultra-high purity nitrogen (AirGas) using an Organomation Associates, Inc. N-EVAP111 evaporator to a volume of 250 µL. The evaporated extracts were immediately analyzed by GC/MS/MS.
High Performance Liquid Chromatographic (HPLC) Analysis – Water Samples
An Agilent 1260 Infinity high performance liquid chromatograph coupled with a diode array detector (DAD) was used to directly measure dicloran, myclobutanil, paraquat, penconazole, and triadimefon in water samples. Dicloran, myclobutanil, and triadimefon were separated on an Agilent Zorbax Eclipse C8 column using a gradient mobile phase consisting of acetonitrile and water with a flow rate of 0.7 mL/min. and injection volume of 40 µL. Penconazole was analyzed under the same conditions but was separated using a Zorbax Eclipse C18 column. Dicloran, myclobutanil, penconazole, and triadimefon were detected at 380 nm, 230 nm, 220 nm, and 225 nm respectively. Paraquat was separated using an Agilent InfinityLab Poroshell 120 HILIC-Z column using a gradient mobile phase consisting of 0.05 M ammonium formate in water (pH: 3) and 0.1% formic acid in acetonitrile with a 0.5 mL/min. flow rate and 2.5 µL injection volume with detection at 258 nm.
Gas Chromatographic Analysis – Water and Tissues Samples
A Bruker Scion 456-GC/MS/MS was used to analyze pesticide residues from all the tissue extractions and the SPE extracts from the bifenthrin and chlorpyrifos water samples. Residues were separated on a Restek RXI-PAH column, 60 m length, 0.25 internal diameter, 0.10-micron film thickness programmed at 90°C for 3 min. and then ramped 5°C/min to 300°C where the temperature was held for 10 min. The carrier gas was ultra-high purity helium (AirGas) and the collision gas was ultra-high purity argon (AirGas). External standards were used to quantify all the concentrations of all analytes whether measured by GC or HPLC.
Table 1.
| Capelin vs. Silverside Tissue Extractions - Myclobutanil and Bifenthrin | |||
| Capelin | |||
| Bifenthrin | Myclobutanil | ||
| SDEV | 4.354731143 | SDEV | 6.247633574 |
| average | 73.04254018 | average | 71.68518738 |
| RSDEV | 6.907607356 | RSDEV | 9.966682457 |
| N | 3 | N | 3 |
| VALUES | 73% +/- 6.9 | 72% +/- 8.8 | |
| Silverside | |||
| Bifenthrin | Myclobutanil | ||
| SDEV | 3.4431336 | SDEV | 5.563774823 |
| average | 70.2287361 | average | 66.62929726 |
| RSDEV | 5.813282246 | RSDEV | 9.82490529 |
| N | 3 | N | 3 |
| 70% +/- 5.8 | 67% +/- 8.9 | ||
Table 2.
| 570 ppb Triadimefon Exposures | |||
| Salinity (ppt_ | Sample | Measured Conc (ppm) | Avg Conc (ppm) |
| 5 | A | 2.3 | 2.71 |
| B | 3.11 | ||
| 25 | A | 3.9 | 4.32 |
| B | 4.74 | ||
Cell lines
Following Covid delays that extended through fall 2020, we restarted cell cuturing efforts. This included the purchase of new media and extensive decontamination procedures to prevent yeast growth (a problem in 2019), thus harvesting and culture has resumed. Antifungal-resistant yeast contamination is still present in small amounts, but changes in culture protocols (e.g. buffer used, immortalization step moved before dispersal) has resulted in minimization of this contamination. Cardiomyocytes and hepatocytes isolated from silversides were recently successfully immortalized using lentiviral infection of dispersed tissues with plasmids encoding large T antigen. We have not been successful in isolating osteoblasts but are continuing to try.
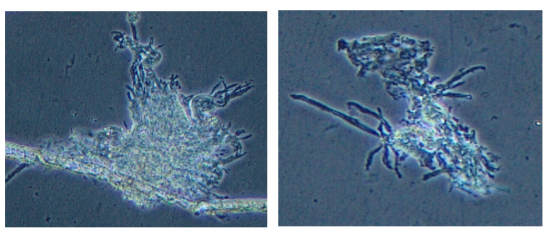
Cardiomyocytes consist of both non-adherent striated cells and slow-growing adherent focal clusters of spherical, transformed cells (Figure 1). Immortalized hepatocytes are also predominantly non-adherent, with transformed cells proliferating in clumps of several cells. While significant resistant fungal and bacterial contamination remain, this is being kept at bay via higher concentrations of penicillin-streptomycin and Fungizone, and minimizing pathogenic takeover in culture via media dilution. Persistent contamination may be deleterious to cell growth rate. Plates of cells are currently being harvested for mRNA isolation and validation with cell-specific primers (Table 3), with RT-converted cDNA screened for cell-specific expression of natriuretic peptides (cardiomyocytes) and liver-specific cytochrome p450 enzymes (hepatocytes). Osteoblasts will require further processing for successful culturing and immortalization, and will continue in parallel with validation of cardiomyocyte and hepatocyte cells. We may try using gelatin or other proven methods to increase the adherence of cardiomyocytes and hepatocytes.
Figure 1. Immortalized cardiomyocytes (CMs) from juvenile M. beryllina, taken with an Olympus CKX41 inverted microscope, and images were taken at 20X mag
Table 3. Reference genes for validation of immortalized CMs.
| Gene | Function |
| cyp1a1 | P450 enzyme |
| nppa | contractability, stress response |
| nppb | contractability, stress response |
| my16 | myosin light chain 1α |
| my17 | myosin light chain 2α |
| 18S ribosome | Housekeeping |
In-vivo exposures in larval M. beryllina
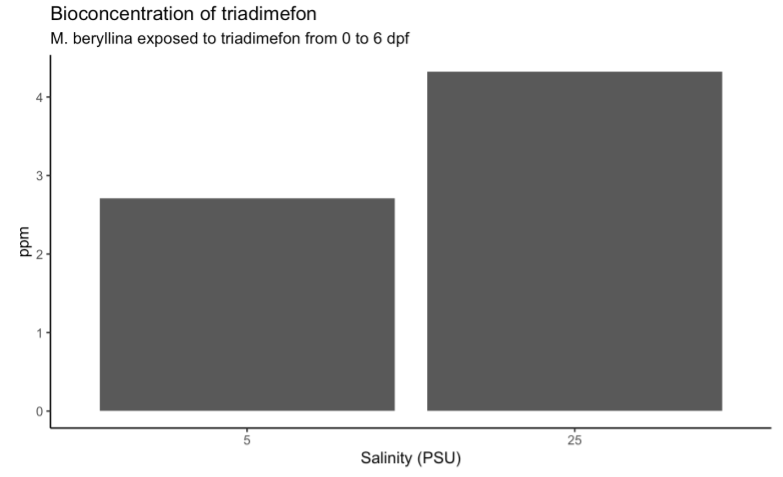
Preliminary data was collected from bioconcentration exposures of triadimefon exposed to Menidia beryllina embryos from 0-6 days post fertilization (dpf) at 5 and 25 practical salinity units (psu). Preliminary data suggests increased bioconcentration of triadimefon at higher salinities (Figure 2), and this was reflected in higher than expected mortality in larval fish exposed at 25 psu to triadimefon.
Due to the large number of embryos required to complete bioconcentration analysis of all compounds we have transitioned our efforts towards establishing a model for bioconcentration analysis at different salinities using commercially available capelin
(tobiko) eggs for the time being. Bioconcentration exposures and analyses with capelin are being conducted at the Louisiana State University. Exposures in live fish and cell lines will still be happening at OSU.
Figure 2: Preliminary plot of triadimefon bioconcentration data after M. beryllina embryos were exposed at OSU for 6 days at two different salinities. Preliminary data suggests increased bioconcentration at higher salinities.
Preliminary sublethal testing was begun on M. beryllina embryos and larvae exposed to triadimefon. However, water quality issues at the OSU Hatfield Marine Science Center (HMSC) because of labor shortages caused by the COVID-19 pandemic has delayed the start of other sub-lethal testing. Sublethal testing will now take place in the spring/summer of 2022 after the Brander Lab has relocated to a new lab space at HMSC. Sublethal testing will occur for triadimefon, bifenthrin, chlorpyrifos, paraquat, penconazole, dicloran, and myclobutanil at 5 and 25 psu. We will investigate the differential responses to the 7 pesticides at the two salinities for the following endpoints: behavior, gene expression, development, and growth.
Finally, our group has published our recent findings on altered LC50 values for the above-mentioned seven compounds in a special issue of Toxics: Impacts of Agrochemicals on Aquatic Ecosystems: Assessing Responses across Biological Scales. We found significant differences in the toxicity of triadimefon between 5 and 15 psu at the LC50 value (Hutton et al. 2021). All other biocides had higher toxicity at 15 psu, but were not significantly different from the 5 psu treatments.
Future Activities:
We are in the process of preparing a manuscript, led by Ph.D. student Sara Hutton, on behavioral toxicity of pesticides for a grant related to the EPA work, using methods developed for use with sublethal exposures for this grant that are planned for spring and summer 2022. This coming year’s work includes finalization of protocols for biocide extraction from water and embryos at LSU, validating immortalized cell lines (cardiomyocytes, hepatocytes) via qPCR, isolating and immortalizing osteocytes, as well as in vivo and in vitro exposures to all biocides at OSU. As a team we implemented successful work-arounds via the use of staggered schedules in 2020-2021 to reduce densities in the laboratory, and protective protocols (masks, additional sanitation procedures). LSU also dealt with a closure of several weeks due to a hurricane hitting Louisiana in summer 2021, no damage was incurred to the lab there. Overall the second- half of 2021 has been better, and we are very optimistic about 2022 now that vaccines will soon be available for all age groups (three of the PIs have children under 12 who were out of in-person school until late April 2021). We will request a no-cost extension due to Covid-related challenges and a lab move for Brander’s group that is planned for spring 2022. This is a move to a new building on the coast, at OSU’s Hatfield Marine Science Center, with renovated aquarium facilities as well as more space for genomic research.
Journal Articles on this Report : 4 Displayed | Download in RIS Format
| Other project views: | All 9 publications | 9 publications in selected types | All 8 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Mundy PC, Hartz KE, Fulton CA, Lydy MJ, Brander SM, Hung TC, Fangue N, Connon RE. Exposure to permethrin or chlorpyrifos causes differential dose-and time-dependent behavioral effects at early larval stages of an endangered teleost species. Endangered Species Research 2021;44:89-103. |
R839503 (2021) |
Exit |
|
|
Hutton SJ, St Romain SJ, Pedersen EI, Siddiqui S, Chappell PE, White JW, Armbrust KL, Brander SM. Salinity Alters Toxicity of Commonly Used Pesticides in a Model Euryhaline Fish Species (Menidia beryllina). Toxics 2021;9(5):114. |
R839503 (2021) |
Exit |
|
|
Derby AP, Fuller NW, Hartz KE, Segarra A, Connon RE, Brander SM, Lydy MJ. Trophic transfer, bioaccumulation and transcriptomic effects of permethrin in inland silversides, Menidia beryllina, under future climate scenarios. Environmental Pollution 2021;275:116545. |
R839503 (2021) |
Exit |
|
|
Segarra A, Mauduit F, Amer NR, Biefel F, Hladik ML, Connon RE, Brander SM. Salinity changes the dynamics of pyrethroid toxicity in terms of behavioral effects on newly hatched delta smelt larvae. Toxics. 2021;9(2):40. |
R839503 (2021) |
Exit Exit |
Supplemental Keywords:
Menidia beryllina, in vivo, in vitro, salinity, biocide, cell lineProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final
- 2024 Progress Report
- 2023 Progress Report
- 2022 Progress Report
- 2020 Progress Report
- Original Abstract
8 journal articles for this project