Grantee Research Project Results
2020 Progress Report: Center for Native American Environmental Health Equity Research
EPA Grant Number: R836157Center: Center for Native American Environmental Health Equity Research
Center Director: Lewis, Johnnye Lynn
Title: Center for Native American Environmental Health Equity Research
Investigators: Lewis, Johnnye Lynn , Gonzales, Melissa , Hudson, Laurie , Cerrato Corrales, Jose Manuel , MacKenzie, Debra Ann
Institution: University of New Mexico
EPA Project Officer: Callan, Richard
Project Period: July 1, 2015 through June 30, 2020 (Extended to June 30, 2021)
Project Period Covered by this Report: July 1, 2019 through June 30,2020
Project Amount: $1,500,000
RFA: NIH/EPA Centers of Excellence on Environmental Health Disparities Research (2015) RFA Text | Recipients Lists
Research Category: Human Health
Objective:
Project 1: Specific Aim 1. Investigate the impact of specific metals and metal mixtures in human populations using biomarkers of toxicity including measures of oxidative stress, DNA damage and PARP activity.
We will test a) DNA damage and repair capacity in peripheral blood mononuclear cells (PBMCs), b) target zinc finger protein PARP-1 activity in PBMCs, and c) measure biomarkers of oxidative stress and oxidative damage in PBMCs and urine from samples collected in the partner communities. The findings will be analyzed with respect to biomonitoring results conducted through the Environmental Monitoring and Interpretation Core. These studies will give us insights into the impact of metals and metal mixtures in the Native communities.
Specific Aim 2. Establish mechanism of specific metal disruption of zinc finger target proteins and potential metal interactions in generation of oxidative stress, DNA damage and PARP activity.
We will use controlled in vitro and cell based assays to test a) metal binding and zinc displacement from zinc finger peptides using analytical techniques, b) metal binding, zinc displacement and PARP-1 function in protein isolated from cells exposed to concentrations of metals spanning the maximum contaminant level (MCL) values and measured exceedance levels found in the communities, c) impact of metals and metal combinations of oxidative stress and oxidative damage, d) effects of metals and metal combinations on cytotoxicity, DNA damage and DNA repair and e) determine whether zinc is protective against the effects of metals. These studies will provide mechanistic insights on the actions of metals and metal mixtures at concentrations relevant to the communities.
Project 2: Aim 1: Determine whether exposure to metals or metal mixtures (measured through biomonitoring) increases the prevalence of anti-nuclear antibodies (ANA) and/or lymphocyte apoptosis in individuals from our three affected Tribal communities.
AIM 2: Determine whether exposures to metals or metals mixtures are associated with changes in cytokine profiles or T and B cell phenotypes in individuals from our three Tribal populations.
AIM 3: Determine the ability of metal mixtures relevant to our three participating Tribal communities to exacerbate or to induce autoimmune disease and immune dysregulation via a drinking water exposure in animal models.
Pilot Projects:
Aim 1. Promote the Center’s goal and objectives by filling research gaps of scientific and community concern.
Aim 2. Promote collaboration between Center members and the partner communities.
Aim 3. Assist promising new investigators to establish independent research in environmental health disparities research.
Progress Summary:
Project 1:
DNA damage, PARP activity measurements, 8-OHdG and urine metal biomonitoring is complete for the Navajo, Cheyenne River Sioux Tribe (CRST) and Crow participants. CRST participant samples were significantly below the values obtained for Navajo participant samples for all three outcome measures. DNA damage and repair measures show similar values for CRST and Crow participant samples despite a significant difference in oxidative stress as measured by urinary 8-OHdG. Significant differences in DNA damage between male and female participants were detected in the Navajo, but not the CRST cohorts. This may be due to pregnant status of the Navajo Birth Cohort Study female population and will warrant further investigation. Final statistical analysis to establish potential relationships between these outcomes measures and urinary metals and serum zinc is pending and will be conducted in collaboration with the statistical team. Studies to investigate and compare arsenic and uranium responses in human monocytes (THP-1 cells) and human T-cells (Jurkat cells) have been completed and the paper has been accepted for publication as of the report date, but after the report period end date of 6/30/2020. Distinct differences in cell sensitivity to metal cytotoxicity were observed. Concentration-dependent cytotoxicity occurred following exposure to arsenite, whereas cells remained viable after 48-hour treatment with up to 100 µM uranyl acetate despite uptake of uranium into cells. Proposed mechanisms of metal toxicity include generation of oxidative stress. Arsenite stimulated an oxidative stress response as detected by Nrf-2 nuclear accumulation and induction of the oxidative stress response genes HMOX-1 and NQO1. No evidence of oxidative stress was observed upon exposure with up to 30 µM uranyl acetate. . Cellular oxidative stress can promote DNA damage and arsenite, but not uranium, stimulated DNA damage as measured by pH2AX. Arsenic enhanced the cytotoxic response to etoposide suggesting an inhibition of DNA repair, yet uranium did not modify the etoposide effect. Similarly, uranium did not inhibit Poly(ADP-Ribose) polymerase-1 activity. Because uranium reportedly stimulates oxidative stress, DNA damage and cytotoxicity in adherent epithelial cells, the current study suggests distinct cell type differences in response to uranium that may relate to generation of oxidative stress and associated downstream consequences. Delineating the actions of uranium across different cell targets will be important for understanding the potential health effects of uranium exposures. The findings for differential oxidative stress response between arsenic and uranium were confirmed in normal human T cells upon activation (manuscript submitted). The findings with uranium suggest that oxidative stress may be a key pathway in metal response. Because many environmental metals have been reported to induce reactive oxygen species (ROS) in biological systems through free radical production, glutathione depletion, and or direct oxidative damage, Jurkat T-cells were used to determine contributions of individual metal and metal combinations in cell toxicity and generating a ROS response as measured by the expression of key genes downstream of ROS signaling. Environmental metals tested include arsenic, uranium, cadmium, cobalt, vanadium, and manganese that were found to be elevated in the blood and urine of Navajo Birth Cohort Study participants (Ong J, poster presentation, 2019). Jurkat T-cell viability was assessed in the presence of metals for up to 48 hours. We detect varying cytotoxic effects of each metal; IC50 values were calculated after 48 hours and relative toxicity is as follows: As>V>Cd>Co>Mn>>U. However, the metals may be impacting normal cellular function without inducing death so oxidative stress response and DNA damage were measured. Of the metals interrogated, only arsenic and cadmium elicited increased expression of HMOX1, an oxidative-stress response gene. When cells were exposed to a mixture of arsenic and cadmium, HMOX1 expression was greatly enhanced compared to single metal exposures (Fig. 1) while the other metals combined with arsenic did not modify the arsenic response. Additionally, exposure to arsenic and cadmium led to an increase in DNA damage as measured by pH2AX and a decreased trend in activity of the DNA repair enzyme, PARP-1.
Vanadium also led to an increase in DNA damage, albeit it may be due to a different mechanism since vanadium did not elicit an oxidative stress response unless the cells were treated with very high levels of the metal (100 μM). Additionally, vanadium did not enhance the ability of arsenic or cadmium to inhibit PARP-1 activity.
Outcomes based on specific mixed metals exposure could help identify and prioritize remediation strategies for certain metal mixtures to advance Native environmental health equity. The work is based on understanding a possible underlying mechanism (ROS generation) that could help predict which mixed metals exposures may be more immunotoxic or immuno-disruptive.
Figure 1. As and Cd work synergistically to produce an oxidative stress response in Jurkat T-cells. Jurkat cells were treated for (A) 6 hrs or (B) 24 hrs with 10 μM As, 30 μM Cd, or a mixture of the two metals. HMOX1 expression was measured via RT-PCR.
Project 2: Significant positive associations between urine uranium levels and the presence of anti-nuclear antibodies (ANA) were found in male participants of the Navajo Birth Cohort Study (NBCS) (Aim 1). We also detected approximately 30% of participants in a Cheyenne River Sioux cohort with ANA positivity, compared to a reported population positivity rate of 13.2% nationally. We previously reported higher than expected (vs NHANES national averages) levels of antibodies against denatured DNA and other nuclear components (histone, native DNA, chromatin). Overall, the specificity profiles are consistent with those associated with environmentally induced autoimmune responses, which are not typically evaluated in clinical panels.
We continued to examine the increased prevalence of serum autoantibodies during the current reporting period with more tissue-specific autoantigen assessments. From CRST samples, specific autoantibodies were detectable in a significant portion of the 225 samples analyzed. Denatured DNA positivity was 36%, about an 8-times higher rate than expected in healthy populations based on literature reports (4-5%).
In our Crow cohort, 41 sera samples were collected in Y4-Y5 and tested for the same 6 antigens (denatured DNA, native DNA, histone, chromatin, thyroglobulin and thyroid peroxidase) in relation to metals exposures. Overall, the age distribution of participants was similar to CRST participants, (mean age 47 years), but lower autoimmune biomarker positivity rates were found, and no associations with age.
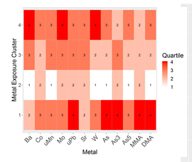
Aim 2. We used a Bayesian clustering technique to assign study participants into groups based on their metals exposure profiles. Through this method, participants “clustered” into 6 groups based on whether their metals levels were in the first through 4th quartile for that metal. About ¼ of study participants were in the lowest quartile (group 1) and about the same proportion were in the highest exposure cluster (group 6).
Aim 3. We are using animal models to address immunologic alterations resulting from low-dose, chronic exposure to mixed metals in drinking water which represent the primary contaminants of concern on the Sioux, Navajo and Crow Reservations. To model “lifetime” exposure, beginning in utero, adult female C57BL/6N mice were exposed to drinking water (control or As+U+Mn or U+Mn water) for 7 days and then paired with male C3H/HeJ mice. Offspring continued to be exposed and were analyzed at 12 weeks. A manuscript resulting from these findings is in preparation. The most remarkable findings include significant changes observed in maturation and activation of T and/or B lymphocytic populations related to mixed metal exposure. The change in these populations could reflect a metals-induced immune dysregulation phenotype similar to that observed in our human population studies and as such warrant further investigation to better understand the impact of these changes on immune function. It will also be important to determine the role of mixed metal exposures and the mechanism by which this exposure alters immune responses.
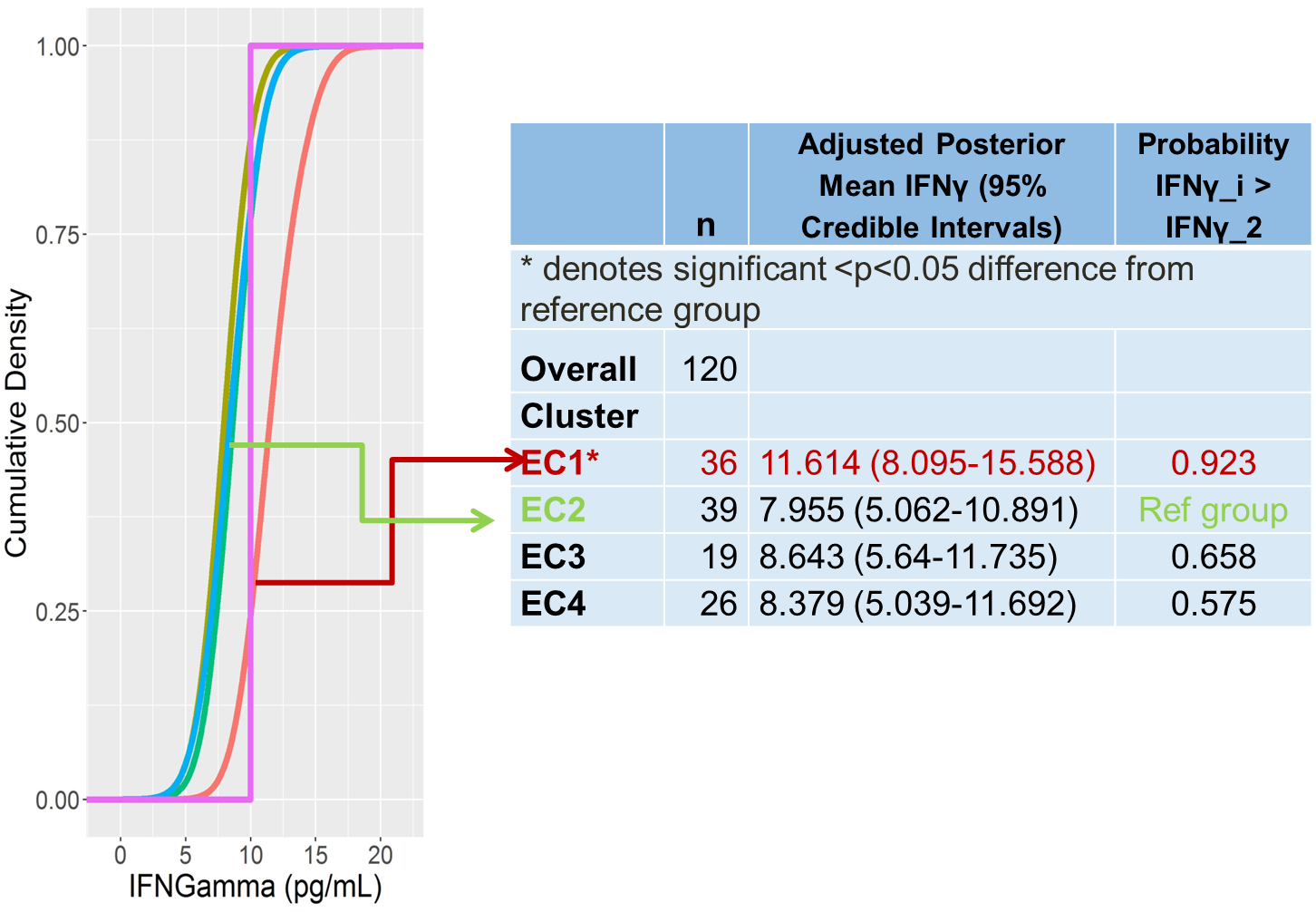
Figure 1. Bayesian Profile Regression Clustering approach to understand expression of cytokines
Pilot Projects:
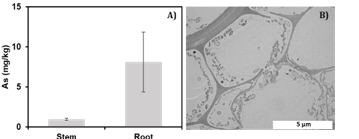
Project 1: Task 1: An 80% decrease in As concentrations from was detected in the hydroponic solution within 12 hours, indicating rapid As removal in all experimental systems. The reduced As levels may have been due to plant update, with or without fungi, or to adsorption and precipitation of As in the hydroponic system. The pH in hydroponic media ranged between 7.04 and 8.10. The biggest changes in pH occurred in Control plants (not innoculated) over 168 hours, suggesting the influence of a chemical reaction on the decrease of As. In control plants from Task 1, As was detected in mainly the roots with an average of 8.17 mg/kg (Fig1A). TEM analyses of control plants detected the presence of microorganisms, indicating that complete microbial sterilization was not achieved (Fig 1B).
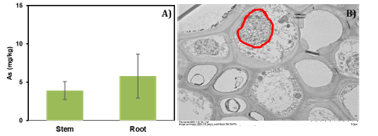
Task 2: Arsenic was detected in mainly the roots (mean 5.78 mg/kg) (Figure 2A) of plants inoculated with Cadaphora species endophytic fungi. In contrast to non-inoculated plants, more As was translocated to the stems of plants inoculated with fungi. Dark septate fungal communities associated with roots were detected as hyphae in intracellular spaces of plants by TEM (Figure 2B). The largest clusters of endophytes were detected in grasses colonized with Cadaphora specie. TEM EDS analyses suggest that As above background in cross-sectional rootlet samples of inoculated plants. EDS and X-Ray STEM map identified low concentrations of As in the cellular structure (De Vore, et al., in preparation). These results indicate that As is heterogeneously distributed in the plant with limited As in intracellular spaces. Calcium phosphate precipitate in solution indicates that substitutions of PO43− ions and other anions on the root surface may limit intracellular AS.
Task 3: An 80% decrease in As concentrations was also detected in the hydroponic solution within the first 12 hours of plants inoculated with native rhizosphere microbes Glomerellales and Eurotiales. Difficulties encountered and Modified Goals. Our original goal to compare As uptake in a native grass (little bluestem) to a hyperaccumulator or non-tolerant plant was modified due to university research restrictions in response to COVID-19. The modified goal determined the effect of dark septate endophytes (DSE) on As uptake. The focus on DSE and little bluestem grass alone was justified as their association is novel and our analyses fill a current gap in the literature with DSE, little bluestem grass and As uptake. Also due to COVID-19 delays, the following analyses are still in progress: scanning electron microscopy of root surfaces of inoculated/non-inoculated plants exposed to 1 PPM As; (Tasks 1, 2); and Illumina DNA sequencing results for fungal microbial community characteristics after As exposure. (Tasks 2, 3).
Project 2:
In Aim 1 of this study, we identified both As and Cd at environmentally relevant concentrations as oxidative stress inducing metals in Jurkat T-cells. A mixture of the two metals enhanced the expression of the oxidative response gene HMOX1 compared to each metal alone (Fig 1). Additionally, there was a trend of increased oxidative stress signaling in response to treatment with vanadium, but only at longer time points. These findings will be verified in normal human CD4 T-cells. An optimal level of oxidative stress is required for proper T cell receptor (TCR) activation, leading to tightly regulated immune response. Any changes to the redox state of the naïve T-cells could alter TCR activation suggesting that metal exposure may contribute to immune dysregulation.
In Aim 2 each metal was tested for its ability to alter normal TCR activation by CD3/CD28 antibodies. The expression of key cytokines and receptors known to increase in response to TCR activation were determined by qRT-PCR. Markers of TCR activation used in this study include: IL2 , IL2RA, IFNg, CD69, ICOS, and CTLA4. Preliminary results from Jurkat T-cells demonstrate that cobalt treatment alone alters downstream TCR signaling genes. In all cases, cobalt significantly increased the expression of TCR activation markers in the absence of TCR activation with CD3/CD28 antibodies. Further investigation will work to elucidate how the increases in gene expression alter cellular function.
Figure 1. A) Arsenic uptake (ug/g) in stems and roots of little bluestem grass grown without fungal inoculation; B) Transmission Electron Microscopy (TEM) image of non-inoculated rootlet cross section using tannic acid counterstained with uranyl acetate and lead citrate (DeVore et al. in preparation)
Figure 2. A) Arsenic uptake (ug/g) in stems and roots of little bluestem grass grown with Cadaphora species fungi (DS 1656); B) Transmission Electron Microscopy (TEM) image of hyphae (dark spots, red outline) in root cross sections of Little bluestem using tannic ccid counterstained with uranyl acetate and lead citrate.
Future Activities:
Project 1: We will complete the statistical analysis of participant samples for Aim 1 and submit a manuscript for peer reviewed publication. We will work with the CEC and RTC to properly communicate findings back to the community after analysis is complete. For Aim 2 we will focus on completing the metal mixtures analysis using the approaches described in the aim. We will also conduct careful analysis of the ability of different metals or metal mixtures to generate oxidative stress as this appears to be a critical component DNA damage and cell toxicity based on our data. We are preparing a manuscript on mixed metal findings to be submitted before Jan 2021. All research is conducted within a QA/QC program to ensure rigorous, robust and unbiased results
Project 2: We will continue to address associations between mixed metal exposures on immune parameters including ANA specificity patterns, cytokine profiles, lymphocyte phenotypes, within the three tribal communities. Final statistical modeling will be completed and reports prepared.
Pilot Projects:
Project 1: the following analyses are still in progress: scanning electron microscopy of root surfaces of inoculated/non-inoculated plants exposed to 1 PPM As; (Tasks 1, 2); and Illumina DNA sequencing results for fungal microbial community characteristics after As exposure. (Tasks 2, 3)
Project 2: Remaining Aim 2 experiments required normal T-cells from healthy, human donors. During the Covid-19 pandemic, donor availability was limited, overnight shipping services were disrupted and donor screening for COVID-19 infection was not available. T-cell isolation, which required close contact, was restricted by institutional policy. A no cost extension was requested and obtained to complete Aim 2 of this project.
Journal Articles: 14 Displayed | Download in RIS Format
| Other center views: | All 90 publications | 14 publications in selected types | All 14 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Dasher-Titus EJ, Hoover J, Luo L, Lee J-H, Du R, Liu KJ, Traber MG, Ho E, Lewis J, Hudson LG. Metal exposure and oxidative stress markers in pregnant Navajo Birth Cohort Study participants. Free Radical Biology and Medicine 2018;124:484-492. |
R836157 (2018) R836157 (2019) |
Exit Exit Exit |
|
|
Doyle JT, Kindness L, Realbird J, Eggers MJ, Camper AK. Challenges and opportunities for tribal waters:addressing disparities in safe public drinking water on the Crow Reservation in Montana, USA. International Journal of Environmental Research and Public Health 2018;15(4):567. |
R836157 (2018) R836157 (2019) R835594 (2018) R835594 (Final) |
Exit |
|
|
Eggers MJ, Doyle JT, Lefthand MJ, Young SL, Moore-Nall AL, Kindness L, Other Medicine R, Ford TE, Dietrich E, Parker AE, Hoover JH, Camper AK. Community engaged cumulative risk assessment of exposure to inorganic well water contaminants, Crow Reservation, Montana. International Journal of Environmental Research and Public Health 2018;15(1):76 (34 pp.). |
R836157 (2018) |
Exit Exit Exit |
|
|
Eggers MJ, Doyle JT, Lefthand MJ, Young SL, Moore-Nall AL, Kindness L, Other Medicine R, Ford TE, Dietrich E, Parker AE, Hoover JH, Camper AK. Community engaged cumulative risk assessment of exposure to inorganic well water contaminants, Crow Reservation, Montana. International Journal of Environmental Research and Public Health 2018;15(1):76. |
R836157 (2019) R835594 (2017) R835594 (2018) R835594 (Final) |
Exit |
|
|
Erdei E, O'Donald E, Luo L, Enright K, OlLeary M, Mackenzie D, Doyle J, Eggers M, Keil D, Lewis J, Henderson J, Rubin R. Comparison of circulating and excreted metals and of autoimmunity between two Great Plains Tribal communities. JOURNAL OF AUTOIMMUNITY 2024;149 |
R836157 (Final) |
Exit |
|
|
Girlamo C, Lin Y, Hoover J, Beene D, Woldeyohannes T, Liu Z, Campen M, MacKenzie D, Lewis J. Meteorological data source comparison-a case study in geospatial modeling of potential environmental exposure to abandoned uranium mine sites in the Navajo Nation. ENVIRONMENTAL MONITORING AND ASSESSMENT 2023;195(7):834 |
R836157 (2020) |
Exit |
|
|
Gonzales M, King E, Bobelu J, Ghahate DM, Madrid T, Lesansee S, Shah V. Perspectives on biological monitoring in environmental health research: a focus group study in a Native American community. International Journal of Environmental Research and Public Health 2018;15(6):1129 (8 pp.). |
R836157 (2018) R836157 (2019) |
Exit Exit Exit |
|
|
Harmon ME, Lewis J, Miller C, Hoover J, Ali AS, Shuey C, Cajero M, Lucas S, Zychowski K, Pacheco B, Erdei E, Ramone S, Nez T, Gonzales M, Campen MJ. Residential proximity to abandoned uranium mines and serum inflammatory potential in chronically exposed Navajo communities. Journal of Exposure Science & Environmental Epidemiology 2017;27(4):365-371. |
R836157 (2018) R836157 (2019) |
Exit Exit |
|
|
Hoover JH, Coker E, Barney Y, Shuey C, Lewis J. Spatial clustering of metal and metalloid mixtures in unregulated water sources on the Navajo Nation – Arizona, New Mexico, and Utah, USA. Science of The Total Environment 2018;633:1667-1678. |
R836157 (2018) R836157 (2019) |
Exit Exit Exit |
|
|
Hoover J, Gonzales M, Shuey C, Barney Y, Lewis J. Elevated arsenic and uranium concentrations in unregulated water sources on the Navajo Nation, USA. Exposure and Health 2017;9(2):113-124. |
R836157 (2016) R836157 (2017) R836157 (2019) |
Exit Exit Exit |
|
|
Hoover J, Coker E, Erdei E, Luo L, MacKenzie D, Lewis J. Preterm Birth and Metal Mixture Exposure among Pregnant Women from the Navajo Birth Cohort Study. ENVIRONMENTAL HEALTH PERSPECTIVES 2023;131(12). |
R836157 (Final) |
|
|
|
Lewis J, Hoover J, MacKenzie D. Mining and environmental health disparities in Native American communities. Current Environmental Health Reports 2017;4(2):130-141. |
R836157 (2017) R836157 (2019) |
Exit Exit Exit |
|
|
Rodriguez-Freire L, Avasarala S, Ali AS, Agnew D, Hoover JH, Artyushkova K, Latta DE, Peterson EJ, Lewis J, Crossey LJ, Brearley AJ, Cerrato JM. Post Gold King Mine spill investigation of metal stability in water and sediments of the Animas River watershed. Environmental Science & Technology 2016;50(21):11539-11548. |
R836157 (2016) R836157 (2017) R836157 (2019) |
Exit Exit Exit |
|
|
Saup CM, Williams KH, Rodríguez-Freire L, Cerrato JM, Johnston MD, Wilkins MJ. Anoxia stimulates microbially catalyzed metal release from Animas River sediments. Environmental Science: Processes & Impacts 2017;19(4):578-585. |
R836157 (2017) R836157 (2019) |
Exit Exit |
Supplemental Keywords:
metals, DNA repair, arsenic, uranium, management, resource allocation, communication, career development, ANA, Immune activity, Native AmericanProgress and Final Reports:
Original Abstract Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R836157C001 Metals and metal mixtures in DNA damage and repair
R836157C002 Development of biomarkers of autoimmunity in 3 tribal communities exposed to mixed metal contaminants
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2019 Progress Report
- 2018 Progress Report
- 2017 Progress Report
- 2016 Progress Report
- Original Abstract
14 journal articles for this center