Grantee Research Project Results
2019 Progress Report: Cardiotoxicity Adverse Outcome Pathway: Organotypic Culture Model and in vitro-to-in vivo Extrapolation for High-throughput Hazard, Dose-response and Variability Assessments
EPA Grant Number: R835802Center: Organotypic Culture Models For Predictive Toxicology Center
Center Director: Rusyn, Ivan
Title: Cardiotoxicity Adverse Outcome Pathway: Organotypic Culture Model and in vitro-to-in vivo Extrapolation for High-throughput Hazard, Dose-response and Variability Assessments
Investigators: Rusyn, Ivan , Wright, Fred A. , Threadgill, David W.
Current Investigators: Rusyn, Ivan
Institution: Texas A & M University , North Carolina State University , The Hamner Institutes
Current Institution: Texas A & M University , North Carolina State University
EPA Project Officer: Callan, Richard
Project Period: June 1, 2015 through May 31, 2019 (Extended to May 31, 2022)
Project Period Covered by this Report: June 1, 2019 through May 31,2020
Project Amount: $6,000,000
RFA: Organotypic Culture Models for Predictive Toxicology Center (2013) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Project 1:
Important target areas for addressing data gaps through in vitro screening include evaluations of cardiotoxicity. Despite the fact that current conservative estimates relate at least 23% of all cardiovascular disease cases to environmental exposures, the identities of the causative environmental agents remain largely uncharacterized. Moreover, cardiotoxicity remains among the most pronounced reasons, comparable to those associated with hepatotoxicity, for drug attrition during clinical trials and post-marketing. Current pre-clinical approaches for cardiophysiologic evaluations of chemicals rely almost exclusively on large animal models and this approach has significant limitations in terms of cost and complexity of the studies. Hence, there is a pressing demand for the development of comprehensive, multiparametric screening strategies that provide improved predictability of cardiotoxic effects.
The central hypotheses of this project are that: (i) human induced pluripotent stem cell (iPSC)-derived cardiomyocyte cultures constitute an effective organotypic culture model for predictive toxicity screening of environmental chemicals; (ii) a population-based experimental design can assess variation in toxicity to better characterize uncertainties; and (iii) integration of pharmacokinetic and high-throughput screening data further improves confidence in NexGen health assessments. We are testing these hypotheses by pursuing the following specific objectives.
Specific Objective 1: To conduct population-based concentration-response high-content/throughput in vitro screening of up to 200 ToxCast chemicals in human iPSC-derived cardiomyocytes from 100 individuals.
Specific Objective 2: To demonstrate how in vitro testing-derived phenotypes are informative of the molecular and cellular events in the adverse outcome pathway (AOP) for cardiotoxicity.
Specific Objective 3: To collect pharmacokinetic data using human hepatocytes and plasma to perform in vitro-to-in vivo extrapolations of the dose-response information from cardiotoxicity screening to human exposures.
Project 2:
Important target areas for addressing data gaps through in vitro screening include evaluations of cardiotoxicity. Despite the fact that current conservative estimates relate at least 23% of all cardiovascular disease cases to environmental exposures, the identities of the causative environmental agents remain largely uncharacterized. Moreover, cardiotoxicity remains among the most pronounced reasons, comparable to those associated with hepatotoxicity, for drug attrition during clinical trials and post-marketing. Current pre-clinical approaches for cardiophysiologic evaluations of chemicals rely almost exclusively on large animal models and do not consider underlying genetic variation in evaluation of risk. Use of large animals has significant limitations in terms of cost and complexity of the studies, especially for the inclusion of genetic diversity. Hence, there is a pressing demand for the development of comprehensive, multi-parametric screening strategies that provide improved predictability of cardiotoxic effects.
The central hypotheses of this project are that: (i) genetic variation influences how mouse iPSC-derived embryoid body (EB)-OCM respond to insults; (ii) analyses using the CC population can be used to uncover genetic and transcriptional networks that control cardiotoxic responses within AOP; and (iii) discoveries using in vitro OCMs are predictive of in vivo cardiotoxic responses. We are testing these hypotheses by pursuing the following specific objectives.
Specific Objective 1: To perform in vitro screening of ToxCast chemicals using iPSC-derived EB-OCM CC genetic reference population.
Specific Objective 2: To collect RNA-seq data from EB-OCM to inform molecular and cellular events in the AOP for cardiotoxicity.
Specific Objective 3: To validate the IVIVE paradigm and risk prediction using genetically matched and exposure-naïve CC mice, respectively.
Project 3:
The activities in Projects 1 and 2 necessitate a targeted, yet comprehensive analytic pipeline for analyzing organotypic culture model data screened for dosimetry, physiological parameters, and genetic and transcriptomic profiles, culminating in an informed basis for ranking and prioritization. Project 3 is responsible to collate, analyze, and synthesize the results from Projects 1 and 2. The data gathered in Projects 1 and 2 has included numerous screening measurements in human iPSC-derived cardiomyocytes and rodent embryonic stem cells, building upon recent advances to maximize the informativeness of beating cardiomyocyte models. The activities in the Project followed the original proposal, with screening methods focusing on concentration-response modeling and multivariate analysis. By harnessing these efforts in a coherent pipeline, the Project synergized with Projects 1 and 2 and added value to the entire Center activity.
In this past year the Project was in no-cost extension, having largely reached its goal of developing a coordinated analysis and decision-support pipeline based on complex data from organotypic culture model systems in humans and mice. These standard approaches and tools that can be used in future cardiotoxicity screening and inform human health assessments.
Efforts in the upcoming no-cost extension year will be devoted to finalizing publications associated with the methods, and polishing the associated software. The research goals are being achieved by pursuing the following specific objectives.
Specific Objective 1: To apply and refine methods to use the pharmacokinetic data from Project 1 to perform in vitro-to-in vivo extrapolation and subsequent generation of oral equivalent doses. [This Objective was moved to Project 1 at Texas A&M University, as described in detail in previous reports]
Specific Objective 2: To apply concentration-response modeling to establish robust and appropriate points of departure.
Specific Objective 3: To perform annotation-informed analyses of population variation and association.
Specific Objective 4: To perform ranking and prioritization analyses of the ToxCast chemicals screened.
Progress Summary:
Project 1:
Most of the work in the past year has focused on the activities under all proposed Specific Objectives. To date, we have accomplished the following:
• We are continuing the analysis of the data from testing 140 compounds selected in consultation with FDA, NTP and EPA-NCCT in a population-based in vitro iPSC-derived cell model with myCell cardiomyocytes from 43 normal donors. Several manuscripts detailing the outcomes have been published or are in submission. For instance, we are demonstrating how population Bayesian concentration-response modeling can be used to characterize toxicodynamic (TD) variability and replace the default uncertainty factor for TD variability. Additionally, we demonstrate how this modeling, when combined with high throughput toxicokinetics, can be used to characterize risk through calculation of margins of exposure.
• We showed that human iPSC-derived cardiomyocytes are among most informative cell types for rapid hazard characterization of environmental chemicals. Our study showed that human induced pluripotent stem cell (iPSC)-derived cells (hepatocytes, neurons, cardiomyocytes and endothelial cells) and a primary endothelial cell line are a practical high-throughput in vitro model for rapidly evaluating potential hazards of chemicals using a small number of human cells. We demonstrated the potential of this in vitro screening model to inform rapid risk-based decision making through ranking, clustering, and assessment of both hazard and risks of diverse environmental chemicals.
• Our studies also show that human iPSC-derived cardiomyocytes can be effectively used to study cardiovascular effects of polychlorinated biphenyls and their major metabolites. In a study of 25 PCBs and their major metabolites detected in humans, or in vitro data-based risk characterization analysis showed that while in vitro derived effective concentrations exceed the levels measured in the general population, risks could not be ruled out due to the potential for population variability in susceptibility and the need to fill data gaps using read-across approaches. This study demonstrated a strategy for how in vitro data can be used to characterize human health risks from PCBs and their metabolites.
• We have completed testing 1,000+ compounds selected in consultation with FDA, NTP and EPA in a population-based in vitro iPSC-derived cell model with myCell cardiomyocytes from 5 donors. Data analyses are underway.
• We have completed collecting samples for whole genome analysis of the effects of drugs and environmental chemicals (650+) on human iPSC-derived cardiomyocytes. These samples have been sent to collaborators at BioSpyder for TempO-seq analysis.
These experiments and observations are significant because they demonstrate novel possibilities of utilizing a human microphysiological system that is population-based for readacross and grouping of the individual chemicals, mixtures, and complex substances.
Project 2:
Most of the work in the past year has focused on the activities under proposed Specific Objective 3. To date, we have accomplished the following:
• After identifying a major source of variation in deriving embryoid bodies for cardiomyocyte differentiation as the source of serum that was being used, we have started getting more consistent differentiation. We have been using this new media for several CC-derived iPSc lines and have also found variation in the extent of differentiation that is related to culture conditions. Using a spinner flask, the size and reproducibility has been improved. These are now being used in differentiation assays to test cardiotoxicants that we have already collected in vivo data on.
• In addition to iPSC derived cardiomyocytes, we have established primary cardiomyocyte cultures from CC lines with in vivo data with the goal of comparing iPSC derived cardiomyocytes, primary cardiomyocytes, and in vivo cardiomyocytes to cardiotoxicant response across at least 8 CC lines.
• To investigate the in vitro-to-in vivo predictive ability of OCM, we have completed echocardiography (Echo) using ECGenie and ECG using a Vevo High-Frequency Ultrasound for detailed baseline cardiac phenotypes for 55 CC lines using both male and female mice. These Echo measures were performed in both conscience and unconscious mice, with substantial strain-specific differences observed in many measurements that are far greater than anticipated.
Project 3:
During this no-cost extension year, most of the work in the past year focused on the activities under proposed Specific Objectives 2, 3, and 4.
To date, this year we have accomplished the following:
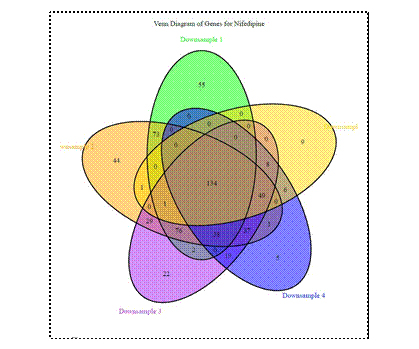
• Down-sampling analyses of the TempoSeq pipeline have been revealing in better understanding the role of replication in highthroughput dose-response modeling, when the total number of assays is held fixed. These issues are critically important for study design, and have an important role to play in toxicological design practice. These analyses show the important role of replication in obtaining robust results, and illustrate the tradeoff vs. coverage with multiple chemicals. Fig 3.1 illustrates with a Venn diagram of expression effects for Nifedipine in cardiomyocytes. An associated manuscript us underway and the results were to be presented at the 2020 SOT. Due to the high degree of feature replication in gene expression, the results also serve to provide important information on design for lowerthroughput assays, including physiological parameters.
FIgure 3.
• Our published improved pipeline for dose-response analysis has been brought to a streamlined level so that it has higher impact in toxicology, including for alternate cell models and alternate chemical agents.
• The dose-response pipeline has had additional added flags and corrections for control-sample bias, which can otherwise produce apparent dose-response effects. Briefly, Hill models are fit in which the mean response for control samples are “tied down” to a common value. We have also expanded the types of points of departure possible to be detected, including now those based on (i) standard deviations departures from control, (ii) departures on the fitted Hill model, and (iii) departures according to a specified percentage change.
• The annotation-informed analyses of population variation and association is furthered by the use of set-based approaches, for which we have developed the Marker Trait Complete Analysis approach. The general approach is generic, but is particularly useful in settings with collections of physiological phenotypes for cell assays with multiple predictive features.
• We have completed scripting of pipelines to more easily compare human genetic variation to phenotypes, automatically connecting analyses to LocusZoom output.
• We have completed scripting our analysis protocols using R/Markdown.
Fig 3.1 illustrates with a Venn diagram of expression effects for Nifedipine in cardiomyocytes
Future Activities:
Project 1:
• We will continue data analysis of experiments in 5 donors with 1,000+ chemicals.
• We will conduct data analysis for TempO-seq high-throughput transcriptomics on 650+ chemicals in the standard iPSC-cardiomyocyte donor.
• We will conduct a simulation analysis of the 43 donor/140 chemical set analyzing the power of different sample sizes (in terms of diverse donors) to characterize hazard and toxicodynamic variability.
• We will continue collecting protein binding and other kinetic data to enable in vitro-to-in vivo comparisons as needed.
• We will work closely with Project 3 staff to analyze the data from high-content screening and high-throughput transcriptomics.
Project 2:
• We will use existing iPS lines from the eight CC lines used to screen chloroquine and isoproterenol (previously tested in vivo) to test in vitro effects on cardiomyocyte function in collaboration with Project 1. These studies were interrupted with the COVID-19 shutdown but will soon re-start. These will be done in both iPSC derived cardiomyocytes and in primary cardiomyocytes.
• Analysis of baseline cardiac function using both EKG an Echo (conscience and unconscious) will be submitted for publication.
• By conclusion of the project, we anticipate having generated detailed baseline cardiac phenotypes for 55 CC lines, performed genetic analysis to identify the genetic architecture underlying cardiac phenotypic variation, tested 8 iPSC and corresponding CC lines and primary cardiomyocytes for several chemicals identified in Project 1 as candidate cardiotoxicants. Although not of the scale original envisioned, the results will still address the original primary goal of the project, are mice predictive of human cardiotoxicity and do in vitro OCM accurately recapitulate in vivo phenotypes.
Project 3:
• Completing the down-sampling analysis and providing a publication that assists the community in understanding the costs and benefits of various dose-response designs for cardiomyocyte studies, as well as other cell-based designs.
• Finishing analysis of the data from high-content screening and high-throughput transcriptomics, working closely with Project 1 and 2 personnel.
• Finalizing the developed protocol for variability analysis across cell lines. By using this protocol in concert with other developing chemical prioritization schemes, e.g. in ToxPi, we can extend the concept of the down-sampling analysis to its consequences in ToxPi summaries.
Journal Articles: 46 Displayed | Download in RIS Format
| Other center views: | All 151 publications | 46 publications in selected types | All 46 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Auerbach S, Filer D, Reif D, Walker V, Holloway AC, Schlezinger J, Srinivasan S, Svoboda D, Judson R, Bucher JR, Thayer KA. Prioritizing environmental chemicals for obesity and diabetes outcomes research: a screening approach using ToxCastTM high-throughput data. Environmental Health Perspectives 2016;124(8):1141-1154. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) R835802C003 (2015) |
|
|
|
Barton-Maclaren TS, Westphal M, Sarwar E, Mattison D, Chiu WA, Dix D, Kavlock R, Krewski D. Challenges and opportunities in the risk assessment of existing substances in Canada: lessons learned from the international community. International Journal of Risk Assessment and Management 2017;20;(1-3):261-283. |
R835802 (2016) R835802 (2017) R835802 (2018) |
Exit Exit Exit |
|
|
Blanchette AD, Grimm FA, Dalaijamts C, Hsieh NH, Ferguson K, Luo YS, Anson B, Rusyn I, Chiu WA. Thorough QT/QTc in a dish:An in vitro human model that accurately predicts clinical concentration-QTc relationships. Clinical Pharmacology and Therapeutics 2019;105:1175-1186. |
R835802 (2018) R835802C001 (2018) |
|
|
|
Blanchette A, Burnett S, Rusyn I, Chiu W. A tiered approach to population-based in vitro testing for cardiotoxicity:Balancing estimates of potency and variability. Journal of Pharmacological and Toxicological Methods 01;119(107154). |
R835802 (2020) |
Exit Exit |
|
|
Blanchette A, Burnett S, Grimm F, Rusyn I, Chiu W. A Bayesian Method for Population-wide Cardiotoxicity Hazard and Risk Characterization Using an In Vitro Human Model. Toxilogical Sciences 2020;178(2):391-403. |
R835802 (2019) |
Exit Exit |
|
|
Bokkers BGH, Mengelers MJ, Bakker MI, Chiu WA, Slob W. APROBA-Plus: a probabilistic tool to evaluate and express uncertainty in hazard characterization and exposure assessment of substances. Food and Chemical Toxicology. 2017;110:408-417. |
R835802 (2017) R835802 (2018) |
Exit Exit Exit |
|
|
Burnett S, Karmakar M, Murphy W, Chiu W, Rusyn I. A new approach method for characterizing inter-species toxicodynamic variability. Journal of Toxicology and Environmental Health, Part A 2021;. |
R835802 (2020) |
Exit Exit |
|
|
Ceballos D, Luo Y, Chen Z, Blanchette A, Zhou Y, Wright F, Baker E, Chiu W, Rusyn I. Relationships between constituents of energy drinks and beating parameters in human induced pluripotent stem cell (iPSC)-Derived cardiomyocytes. Food and Chemical Toxicology 2021;149:111979. |
R835802 (2019) |
Exit Exit |
|
|
Chiu WA, Wright FA, Rusyn I. A tiered, Bayesian approach to estimating population variability for regulatory decision-making. ALTEX 2017;34(3):377-388. |
R835802 (2016) R835802 (2017) R835802 (2018) R835166 (2016) R835166 (Final) |
Exit Exit |
|
|
Chiu WA, Axelrad DA, Dalaijamts C, Dockins C, Shao K, Shapiro AJ, Paoli G. Beyond the RfD:Broad application of a probabilistic approach to improve chemical dose-response assessments for noncancer effects. Environmental Health Perspective 2018;126(6):067009. |
R835802 (2018) R835802C001 (2018) |
|
|
|
Chiu WA, Guyton KZ, Martin MT, Reif DM, Rusyn I. Use of high-throughput in vitro toxicity screening data in cancer hazard evaluations by IARC Monograph Working Groups. ALTEX 2018;35(1):51-64. |
R835802 (2017) R835802 (2018) R835802C003 (2018) |
Exit Exit |
|
|
Chiu WA, Rusyn I. Advancing chemical risk assessment decision-making with population variability data: challenges and opportunities. Mammalian Genome 2018;29(1-2):182-189. |
R835802 (2017) R835802 (2018) |
Exit Exit |
|
|
Cote I, Andersen ME, Ankley GT, Barone S, Birnbaum LS, Boekelheide K, Bois FY, Burgoon LD, Chiu WA, Crawford-Brown D, Crofton KM, DeVito M, Devlin RB, Edwards SW, Guyton KZ, Hattis D, Judson RS, Knight D, Krewski D, Lambert J, Maull EA, Mendrick D, Paoli GM, Patel CJ, Perkins EJ, Poje G, Portier CJ, Rusyn I, Schulte PA, Simeonov A, Smith MT, Thayer KA, Thomas RS, Thomas R, Tice RR, Vandenberg JJ, Villeneuve DL, Wesselkamper S, Whelan M, Whittaker C, White R, Xia M, Yauk C, Zeise L, Zhao J, DeWoskin RS. The next generation of risk assessment multi-year study--highlights of findings, applications to risk assessment, and future directions. Environmental Health Perspectives 2016;124(11):1671-1682. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) R835802C001 (2015) R835166 (Final) |
|
|
|
Fantke P, Aylward L, Bare J, Chiu WA, Dodson R, Dwyer R, Ernstoff A, Howard B, Jantunen M, Jolliet O, Judson R, Kirchhübel N, Li D, Miller A, Paoli G, Price P, Rhomberg L, Shen B, Shin HM, Teeguarden J, Vallero D, Wambaugh J, Wetmore BA, Zaleski R, McKone TE. Advancements in life cycle human exposure and toxicity characterization. Environmetnal Health Perspective 2018;126:125001. |
R835802 (2018) R835802C001 (2018) |
|
|
|
Ford L, Lin H, Tsai H, Zhou Y, Wright F, Sedykh A, Shah R, Chiu W, Rusyn I. Hazard and risk characterization of 56 structurally diverse PFAS using a targeted battery of broad coverage assays using six human cell types. TOXICOLOGY 2024;503(153763). |
R835802 (Final) |
Exit |
|
|
Garbutt TA, Konneker TI, Konganti K, Hillhouse AE, Swift-Haire F, Jones A, Phelps D, Aylor DL, Threadgill D. Permissiveness to form pluripotent stem cells may be an evolutionarily derived characteristic in Mus musculus. Scientific Reports 2018;8:14706. |
R835802 (2018) R835802C002 (2018) |
Exit |
|
|
Grimm FA, Iwata Y, Sirenko O, Bittner M, Rusyn I. High-content assay multiplexing for toxicity screening in induced pluripotent stem cell-derived cardiomyocytes and hepatocytes. Assay and Drug Development Technologies 2015;13(9):529-546. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) R835802C001 (2015) |
Exit Exit Exit |
|
|
Grimm FA, Iwata Y, Sirenko O, Chappell GA, Wright FA, Reif DM, Braisted J, Gerhold DL, Yeakley JM, Shepard P, Seligmann B, Roy T, Boogaard PJ, Ketelslegers HB, Rohde AM, Rusyn I. A chemical-biological similarity-based grouping of complex substances as a prototype approach for evaluating chemical alternatives. Green Chemistry 2016;18(16):4407-4419. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) R835802C001 (2015) R835166 (Final) |
Exit Exit Exit |
|
|
Grimm FA, Blanchette A, House JS, Ferguson K, Hsieh NH, Dalaijamts C, Wright AA, Anson B, Wright FA, Chiu WA, Rusyn I. A human population-based organotypic in vitro model for cardiotoxicity screening. ALTEX 2018;35:441-452. |
R835802 (2018) R835802C001 (2018) R835802C003 (2018) |
Exit Exit |
|
|
Grimm FA, House JS, Wilson MR, Sirenko O, Iwata Y, Wright FA, Ball N, Rusyn I. Multi-Dimensional in Vitro Bioactivity Profiling for Grouping of Glycol Ethers. Regulatory Toxicology and Pharmacology 2019;101:91-102. |
R835802 (2018) R835802C001 (2018) R835166 (Final) |
Exit |
|
|
Grimm FA, Klaren WD, Li X, Lehmler HJ, Karmakar M, Robertson LW, Chiu WA, Rusyn I. Cardiovascular effects of polychlorinated biphenyls and their major metabolites.Environmental Health Perspectives 2020;128(7):077008. |
R835802 (Final) |
|
|
|
Grondin CJ, Davis AP, Wiegers TC, King BL, Wiegers JA, Reif DM, Hoppin JA, Mattingly CJ. Advancing exposure science through chemical data curation and integration in the Comparative Toxicogenomics Database. Environmental Health Perspectives 2016;124(10):1592-1599. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) R835802C003 (2015) |
|
|
|
Guyton KZ, Rusyn I, Chiu WA, Corpet DE, van den Berg M, Ross MK, Christiani DC, Beland FA, Smith MT. Application of the key characteristics of carcinogens in cancer hazard identification. Carcinogenesis 2018;39(4):614-622. |
R835802 (2017) R835802 (2018) |
Exit Exit Exit |
|
|
House JS, Grimm FA, Jima DD, Zhou Y-H, Rusyn I, Wright FA. A pipeline for high-throughput concentration response modeling of gene expression for toxicogenomics. Frontiers in Genetics 2017;8:168 (11 pp.). |
R835802 (2017) R835802 (2018) |
Exit Exit Exit |
|
|
Iwata Y, Klaren WD, Lebakken CS, Grimm FA, Rusyn I. High-content assay multiplexing for vascular toxicity screening in induced pluripotent stem cell-derived endothelial cells and human umbilical vein endothelial cells. Assay and Drug Development Technologies 2017;15(6):267-279. |
R835802 (2017) R835802 (2018) R835166 (Final) |
Exit Exit |
|
|
Judson R, Houck K, Martin M, Richard AM, Knudsen TB, Shah I, Little S, Wambaugh J, Woodrow Setzer R, Kothya P, Phuong J, Filer D, Smith D, Reif D, Rotroff D, Kleinstreuer N, Sipes N, Xia M, Huang R, Crofton K, Thomas RS. Editor's highlight: Analysis of the effects of cell stress and cytotoxicity on in vitro assay activity across a diverse chemical and assay space. Toxicological Sciences 2016;152(2):323-339. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) R835802C003 (2015) |
Exit Exit Exit |
|
|
Konganti K, Ehrlich A, Rusyn I, Threadgill DW. gQTL:a web application for QTL analysis using the collaborative cross mouse genetic reference population. G3:Genes, Genomes, Genetics 2018;8(8):2559-2562 |
R835802 (2017) R835802 (2018) R835802C001 (2018) R835802C002 (2018) |
Exit Exit |
|
|
Li G, Shabalin AA, Rusyn I, Wright FA, Nobel AB. An empirical Bayes approach for multiple tissue eQTL analysis. Biostatistics 2018;19(3):391-406. |
R835802 (2017) R835802 (2018) |
Exit Exit |
|
|
LInd L, Araujo J, Barchosky A, Belcher S, Berridge B, Chiamvimonvat N, Chiu W, Cogliano V, Elmore S, Farraj A. Key Characteristics of Cardiovascular Toxicants. Environmental Health Perspectives 2021;129(9). |
R835802 (2020) |
|
|
|
Lu E, Grimm F, Rusyn I, De Saeger S, De Bouvre M, Chiu W. Advancing probabilistic risk assessment by integrating human biomonitoring, new approach methods, and Bayesian modeling:A case study with the mycotoxin deoxynivalenol. ENVIRONMENT INTERNATIONAL 2023;182(108326). |
R835802 (Final) R835166 (Final) |
Exit |
|
|
Lu E, Ford L, Rusyn I, Chiu W. Reducing uncertainty in dose-response assessments by incorporating Bayesian benchmark dose modeling and in vitro data on population variability. RISK ANALYSIS 2024; |
R835802 (Final) R840450 (2023) |
Exit |
|
|
Marvel SW, To K, Grimm FA, Wright FA, Rusyn I, Reif DM. ToxPi Graphical User Interface 2.0: dynamic exploration, visualization, and sharing of integrated data models. BMC Bioinformatics 2018;19(1):80 (7 pp.). |
R835802 (2017) R835802 (2018) R835802C001 (2018) R835802C003 (2018) |
Exit Exit Exit |
|
|
Rusyn I, Greene N. The impact of novel assessment methodologies in toxicology on green chemistry and chemical alternatives. Toxicological Sciences 2018;161(2):276-284. |
R835802 (2017) R835802 (2018) |
Exit Exit |
|
|
Shah I, Setzer RW, Jack J, Houck KA, Judson RS, Knudsen TB, Liu J, Martin MT, Reif DM, Richard AM, Thomas RS, Crofton KM, Dix DJ, Kavlock RJ. Using ToxCast™ data to reconstruct dynamic cell state trajectories and estimate toxicological points of departure. Environmental Health Perspectives 2016;124(7):910-919. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) |
|
|
|
Sirenko O, Grimm FA, Ryan KR, Iwata Y, Chiu WA, Parham F, Wignall JA, Anson B, Cromwell EF, Behl M, Rusyn I, Tice RR. In vitro cardiotoxicity assessment of environmental chemicals using an organotypic human induced pluripotent stem cell-derived model. Toxicology and Applied Pharmacology 2017;322:60-74. |
R835802 (2016) R835802 (2017) R835802 (2018) R835166 (Final) |
Exit Exit Exit |
|
|
Tilley SK, Reif DM, Fry RC. Incorporating ToxCast and Tox21 datasets to rank biological activity of chemicals at Superfund sites in North Carolina. Environment International 2017;101:19-26. |
R835802 (2017) R835802 (2018) |
Exit Exit Exit |
|
|
Wignall JA, Muratov E, Sedykh A, Guyton KZ, Tropsha A, Rusyn I, Chiu WA. Conditional Toxicity Value (CTV) predictor: an in silico approach for generating quantitative risk estimates for chemicals. Environmental Health Perspectives 2018;126(5):057008 (13 pp.). |
R835802 (2017) R835802 (2018) |
|
|
|
Zhang G, Marvel S, Truong L, Tanguay RL, Reif DM. Aggregate entropy scoring for quantifying activity across endpoints with irregular correlation structure. Reproductive Toxicology 2016;62:92-99. |
R835802 (2015) R835802 (2016) R835802 (2017) R835802 (2018) R835802C003 (2015) R835168 (Final) R835796 (2017) |
Exit Exit Exit |
|
|
Zhang G, Roell KR, Truong L, Tanguay RL, Reif DM. A data-driven weighting scheme for multivariate phenotypic endpoints recapitulates zebrafish developmental cascades. Toxicology and Applied Pharmacology 2017;314:109-117. |
R835802 (2016) R835802 (2017) R835802 (2018) R835796 (2017) |
Exit Exit Exit |
|
|
Zhou Y-H, Marron JS, Wright FA. Computation of ancestry scores with mixed families and unrelated individuals. Biometrics 2018;74(1):155-164. |
R835802 (2016) R835802 (2017) R835802 (2018) |
Exit Exit Exit |
|
|
Kosnik MB, Strickland JD, Marvel SW, Wallis DJ, Wallace K, Richard AM, Reif DM, Shafer TJ. Concentration–response evaluation of ToxCast compounds for multivariate activity patterns of neural network function. ARCHIVES OF TOXICOLOGY 2013;94:469-484. |
R835802 (2019) |
Exit |
|
|
Li G, Jima D, Wright FA, Nobel AB. HT-eQTL:integrative expression quantitative trait loci analysis in a large number of human tissues. BMC Bioinformatics 2018;19:95. |
R835802 (2018) R835802C003 (2018) |
|
|
|
Kosnik MB, Reif DM. Determination of chemical-disease risk values to prioritize connections between environmental factors, genetic variants, and human diseases. Toxicology and Applied Pharmacology2019;379:114674. |
R835802C003 (2018) |
Exit Exit |
|
|
Kosnik MB, Planchart A, Marvel SW, Reif DM, Mattingly CJ. Integration of curated and high-throughput screening data to elucidate environmental influences on disease pathways. Computational Toxicology2019;12:100094. |
R835802C003 (2018) |
Exit Exit |
|
|
Chen Z, Liu Y, Wright FA, Chiu WA, Rusyn I. Rapid hazard characterization of environmental chemicals using a compendium of human cell lines from different organs. ALTEX-Alternatives to Animal Experimentation 2020; 37(4):623-638 |
R835802 (2019) |
Exit Exit |
|
|
Hsieh NH, Reisfeld, B, Bois FY, Chiu WA. Applying a global sensitivity analysis workflow to improve the computational efficiencies in physiologically-based pharmacokinetic modeling. Frontiers in Pharmacology 2018 9:588. |
R835802 (2018) R835802C001 (2018) |
Exit Exit |
Supplemental Keywords:
cardiovascular, stem cells, toxicity pathway, variability, pharmacokinetic model, dose-responseRelevant Websites:
ToxPi: Toxicological Prioritization Index Exit
Progress and Final Reports:
Original Abstract Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R835802C001 High-throughput Hazard,Dose-responseandPopulationVariabilityAssessmentofCardiotoxicity in aHumanInducedPluripotentStem Cell(iPSC)-derivedinvitro Culture Model
R835802C002 Linking in vitro-to-in vivoToxicity Testing Using
Genetically-matchedOrganoids and Mice from a Novel Genetic Reference Population
R835802C003 A Pipeline for in vitro-to-in vivo Extrapolation, Population Modeling,
& Prioritization
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2020 Progress Report
- 2018 Progress Report
- 2017 Progress Report
- 2016 Progress Report
- 2015 Progress Report
- Original Abstract
46 journal articles for this center