Grantee Research Project Results
Final Report: Water Innovation Network for Sustainable Small Systems (WINSSS)
EPA Grant Number: R835602Center: Water Innovation Network for Sustainable Small Systems
Center Director: Reckhow, David A.
Title: Water Innovation Network for Sustainable Small Systems (WINSSS)
Investigators: Reckhow, David A. , Lawler, Desmond , Kinney, Kerry A. , Speitel, Gerald E. , Katz, Lynn , Kirisits, Mary Jo , Ray, Chittaranjan , Tobiason, John , Boyer, Treavor H. , Zhang, Qiong , Butler, Caitlin S. , Park, Chul , Shenoy, Prashant , Saleh, Navid , Dvorak, Bruce I. , Wilson, Steven , Brown, Jess , Lai, Rebecca
Institution: University of Massachusetts - Amherst , The University of Texas at Austin , University of Florida , University of Illinois Urbana-Champaign , University of Nebraska at Lincoln , University of South Florida , Carollo Engineers
EPA Project Officer: Aja, Hayley
Project Period: August 1, 2014 through July 31, 2017 (Extended to July 31, 2019)
Project Amount: $4,100,000
RFA: National Centers for Innovation in Small Drinking Water Systems (2013) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
The Water Innovation Network for Sustainable Small Systems (WINSSS) brings together a national team of experts to transform drinking water treatment for small water systems (SWS) to meet the urgent need for state-of-the-art innovation, development, demonstration, and the implementation of treatment, information, and process technologies. This is accomplished in part by leveraging existing relationships with industry through the Massachusetts Water Cluster.
Conclusions:
Education and Outreach
During its initial 3-yr project period, the WINSSS Center maintained an active outreach program that focused on providing credible and engaging information to help advance the state of the science. This allowed for the research, findings, and data developed from the Center to be disseminated by multiple pathways, thereby increasing knowledge and positively impacting small system sustainability and public health protection. Center outreach provides nationwide exposure of WINSSS Center products and other applicable technology-related information. A number of approaches have been used to create a resource that improves access to technology information and fosters communication among stakeholders (small systems, consulting engineers, state personnel, Technology Assistance [TA] providers, and scientists). During the life of the Center, members of the WINSSS Executive Committee collaborated with the DeRISK Center Executive Committee to coordinate outreach and educational activities. This includes creating a joint monthly e-newsletter, a shared website (drinkingwatercenters.org) that describes the work being done for each center with links to each center’s respective site, and collaborating on outreach presentations, such as at the AWWA Annual Conference.
Emerging Technologies
Service Unit 3 for the WINSSS Center is focused on identifying and supporting emerging technologies that were not included in the WINSSS Center proposal. WINSSS Center leadership recognized that researchers outside of the WINSSS and DeRISK Center awards are likely to have some excellent ideas for viable innovative technologies for small water systems. Thus, a competition to offer a total of $200,000 in seed funding to support fundamental work on technologies that are appropriate for small systems and could be ready for use within 10 years was included in the WINSSS Center proposal. Brief summaries of the findings of the 4 projects are presented below.
- Removal of Nitrate from Groundwater without Co-Production of High Concentration of Disposable Brine. Arup SenGupta- and Hang Dong, Lehigh University
The project is summarized below in the abstract from a draft journal article prepared by Hang Dong and Arup SenGupta; the paper is entitled, “CO2-driven hybrid ion exchange nitrate removal process (CHIX-N) with simultaneous partial desalination plus co-removal of fluoride and no brine waste disposal.”
The number of groundwater sources contaminated with nitrate has increased significantly and EPA has set a maximum contamination level of 10 ppm as N for decades. Ion exchange process with standard or nitrate selective resins is commonly applied for nitrate removal. However, intensive brine regeneration (10% - 15% NaCl) and the disposal of such brine-laden regeneration waste poses maximum hardship during practice. In addition, traditional ion exchange process cannot achieve partial desalination and efficient trace ligand co-contaminant removal, ex. fluoride, from the complex groundwater. In this study, we present a novel carbon dioxide driven hybrid ion exchange process (CHIX-N) that does not require intensive chemical consumption and poses no brine waste for nitrate removal. Carbon dioxide (CO2) is the only chemical needed to sustain the process. In contrast to traditional brine (12% NaCl) regeneration, we have demonstrated that CHIX-N process could perform nitrate removal without brine consumption and eliminate the burden of brine waste, plus achieving efficient fluoride removal and more than 50% partial desalination. A hybrid nitrate selective resin loaded with hydrated zirconium oxide nanoparticles (NSR-Zr) and a shell-core weak acid cation exchange resin (SC-WAC) form the heart of the process and both of them could be regenerated by using pressurized carbon dioxide as the sole regenerant. Groundwater from nitrate treatment facility (Lancaster, Pennsylvania) was used to validate the process.
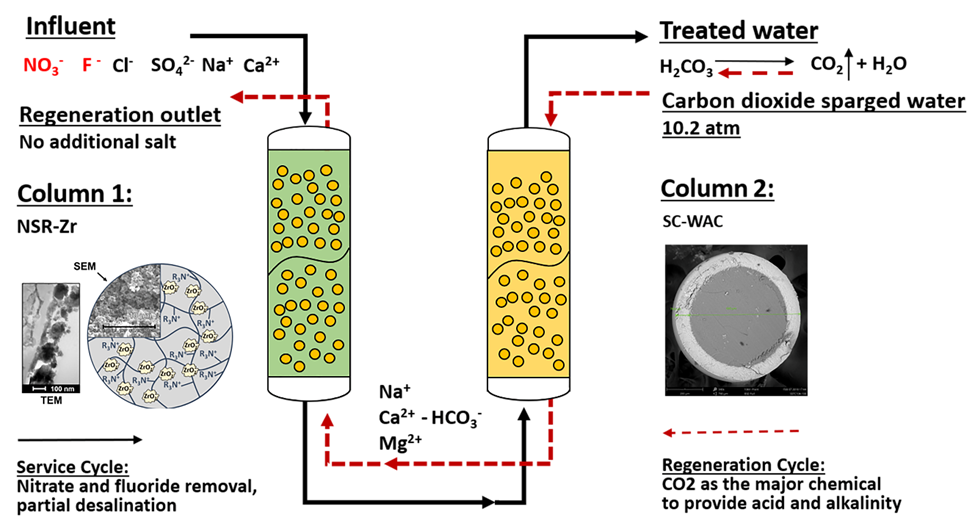
Figure 1 from the draft journal article describes the treatment process components.
Figure 1. Schematic of CHIX-N process illustrating both service cycle (nitrate/fluoride removal and partial desalination) and carbon dioxide regeneration.
Executive Summary:
This project evaluated the efficacy of electrocoagulation (EC) and electrooxidation (EO) for the mitigation of organic carbon (disinfection byproduct precursor), microbes (E. coli and Cryptosporidium), and trace organic compounds (acyclovir, trimethoprim, and benzldimethyldecylammonium chloride) in drinking water. EC and EO were tested independently and sequentially using surface and ground waters, including both synthetic model waters and actual environmental waters. In general, the EC-EO sequence improved removal of the target contaminants, providing more energy efficient treatment in comparison to EC or EO alone, albeit to varying extents. In particular, EC-EO improved system efficacy for treatment of surface waters (characterized by higher organic matter), whereas the relatively clean groundwater matrices did not substantially improve using EC ahead of EO. Electrochemical water treatment technologies may be particularly amenable to small drinking water systems as they offer a number of advantages over conventional water treatment, including avoiding the addition of corrosive chemicals, easy operation and automation, no alkalinity consumption, small footprint, and portability for water treatment during emergencies and in remote settings.
- Biological Denitrification for Small Water Systems Using Iron-Sulfur Minerals. Sarina Ergas and Jeffrey Cunningham- University of South Florida
Treatment of nitrate contaminated groundwater using sulfur-bearing minerals as electron donors for autotrophic denitrification
John Sutton, Erica Dasi, Laura Rodriguez-Gonzalez, Jeffrey Cunningham, Sarina J. Ergas
Department of Civil & Environmental Engineering, University of South Florida
Anthropogenic nitrate contamination of drinking sources causes potential health problems, especially for pregnant and nursing women and infants. The most common technologies for removing nitrate from drinking water rely on physical-chemical processes that can be prohibitively expensive for implementation in small community water systems (CWS). As an alternative, sulfur-oxidizing denitrification has been shown in previous studies to achieve high nitrate removal rates with minimal excess sludge production. However, the use of elemental sulfur as an electron donor results in high alkalinity consumption and sulfate by-product production. Prior studies have shown that the use of sulfide-bearing minerals, such as pyrite (FeS2), may yield similar nitrate removal efficiencies as elemental sulfur with lower by-product generation. In this study, five minerals were initially examined for their suitability as electron donors for autotrophic denitrification through batch microcosm studies. Sphalerite ((Zn,Fe)S) and pyrite provided adequate removal of nitrate with minimal sulfur by-product production compared with pyrrhotite (Fe1-xS¬2) and molybdenite (MoS2). The use of iron (II)-sulfide as an electron donor resulted in ammonia generation, possibly due to dissimilatory nitrate reduction to ammonia (DNRA), which would not be favorable for use in CWS. Based on these studies, sphalerite and pyrite were selected for use in up-flow packed-bed reactors to further examine their ability to promote denitrification. During a 35-day period with a hydraulic residence time (HRT) of 14 hours, removal of total inorganic nitrogen (TIN = NO3--N + NO2--N + NH4+-N) was 28% for sphalerite and 31% for pyrite. After increasing the HRT to 28 hrs, TIN removal increased to 54% for sphalerite and 56% for pyrite, with very low by-product generation. Our current work is directed at further investigations of nitrate removal mechanisms and microbial community dynamics. This is the first study to show that sphalerite is a potential electron donor for denitrification of contaminated groundwater: however, the long HRTs required may result in high reactor volume requirements.
- Reactive Electrochemical Membranes for Simultaneous Removal of Multiple Classes of Contaminants of Concern in Small Drinking Water Systems. Brian Chaplin- University of Illinois at Chicago, Wenqing Xu- Villanova University
The project PIs and graduate students have submitted a journal article to Environmental Science and Technology which has been accepted for publication. The full citation will be included in the WINSSS final report. A summary of the project is found below.
Project Summary
The objectives of this project were: 1) to further improve the performance of Ti4O7-based reactive electrochemical membranes (REMs) by adding carbon adsorptive materials to the membrane matrix and thus increase trace level contaminant adsorption and subsequent electrochemical destruction; and 2) to validate the technical feasibility of using the novel REM for the removal of disinfection byproducts. To that end, powder activated carbon (PAC) or multi-walled carbon nanotubes (MWCNTs) were added to Ti4O7 ceramic REMs. These composite REMs were tested for simultaneous adsorption and electrochemical reduction of N-Nitrosodimethylamine (NDMA). The carbon-Ti4O7 composite REMs had high electrical conductivities (1832 to 2991 S m-1), where the carbon materials and Ti4O7 ceramics were in direct electrical contact. Results indicated that the addition of the carbonaceous materials significantly increased the residence times of NDMA in the REMs by a factor of 3.8 to 5.4, thereby allowing near complete electrochemical NDMA reduction to occur with the application of a -1.1 V/SHE cathodic potential. Synthetic electrolyte solutions with 10 µM NDMA achieved > 4-log NDMA removal in single pass through the PAC-REM and MWCNT-REM, with liquid residence times of 11 to 22 s. Under these experimental conditions the permeate concentrations were between 18 and 80 ng L-1. The treatment of a surface water sample spiked with 6.7 nM NDMA and under similar operating conditions (liquid residence time of 22 s), achieved 92 to 97% removal with permeate concentrations between 16 and 40 ng L-1. Density functional theory calculations were used to interpret the experimental data and simulations indicated that the rate-limiting step was a direct electron transfer reaction followed by an activationless proton transfer. These results were recently published in Environmental Science and Technology (DOI: 10.1021/acs.est.8b05933). This same approach is currently being applied for the removal of haloacetic acids and trihalomethanes and the reaction mechanisms are being investigated by density functional theory. We will leverage the results of this work to apply for future funding from the National Science Foundation.
Center Sustainment
As stated in the WINSSS proposal, our vision for the Center is of an ongoing entity that provides innovations for small systems on a long-term basis, extending well beyond the initial funding period (i.e., beyond 2019). This requires that the Center have a long-term funding plan as well as a plan to keep its contributions relevant and vigorous.
Funding from intellectual property (IP) licensing and validation exercises is a potential source related to technologies developed by the university PIs working under the Center and from Water Cluster activities related to the Center. As new technologies are developed, it has been our intention to establish agreements on IP crafted in a way that will return a fraction of any income to the Center.
In addition, WINSSS has been playing a role in third-party testing and evaluation of new water treatment technologies. A fee for such testing is charged to cover costs of piloting and associated water quality analysis, as well as administrative costs. This testing is an opportunity of great interest to NEWIN and to the Massachusetts Clean Energy Center (CEC). Partly for this reason, the CEC pledged $100,000 to UMass in support of a mobile pilot-testing facility. The mobile pilot-testing trailer was carefully developed on the Amherst campus to make maximum use of existing resources and donated materials. The intent has been for the trailer to be used in the piloting projects within the Center as well as for cluster-related third-party testing and validation.
Direct funding by utilities might proceed via the North Carolina Urban Water Consortium (UWC) model. While small utilities are not generally able to support national technology efforts, medium to large utilities might have the interest and resources to help. Many have recognized that it is in their self-interest to support organizations such as the UWC to help address regional problems related to water quality and treatment. The UWC model incorporates a board of advisors from NC water utilities who make decisions on funding using pooled resources. On a national scale, the Water Research Foundation (WRF, formerly AWWARF) runs a research program partly through subscriber utility fees. The WRF subscribers are heavily weighted toward the largest utilities in the US. Many medium sized systems have elected not to subscribe because of perceptions that WRF’s research activities are driven by the needs of a relatively small number of big utilities. This situation has helped create some space for local organizations such as the NC UWC. The Center has proposed to work with the National Institutes for Water Resources centers, who have expressed an interest, to help them develop a local UWC model for interested states. In return, the National Center would be given some authority to help steer the funding toward issues that have national as well as regional significance and are appropriate to small systems as well as medium or large systems.
National and regional industry or trade groups (e.g., NRWA, AWWA, NEIWPCC) and their local sections are strong supporters of this Center. This support may not translate to any direct financial assistance. It may however, lead to collaborative education and technology transfer activities that are mutually beneficial. This type of collaboration has developed in many parts of the US, such as with the NEWWA coalition. The National Center may be able to continue its outreach activities at a greatly reduced cost by virtue of such collaborations.
State primacy agencies should benefit substantially from the Center’s activities. New technologies and associated training will help small systems comply with state and federal regulations, which will lift some of the burdens under which primacy agencies are working. Since the state agencies would be a chief beneficiary of this work, it makes sense to look to them for assistance to continue the Center funding. Many state agencies managing water programs are financially challenged. Nevertheless, we may be able to make a convincing argument that supporting the Center is a good investment and ultimately relieves regulatory pressures, helps with training of state regulatory personnel, and helps with interstate regulatory coordination (Project B1).
Long-term Innovation
Sustainability of the Center’s intellectual vigor is another issue that requires careful planning. To be successful in the long-term, it is critical to embrace new ideas and approaches. A dynamic and fertile atmosphere for innovation must be maintained at the Center. All of the Center PIs actively engage in research, immerse themselves in the scientific literature, interact with industry on new water technologies, and regularly participate in national and international conferences as well as in panels to evaluate proposals for national and international funding agencies. Nevertheless, to best serve national needs, the flow of new ideas must come from a broader range of researchers than those directly funded by this Center. Many new ideas worth pursuing will not come from the Center PIs (i.e., will not be the Center’s intellectual property) but from other researchers not currently associated with the Center who will be best suited to advance them. As such, one activity of the Center in 2017 and 2018 was the four awards of $50,000 each we made under the Service Unit 3 Emerging Technologies program (as listed above).
Mobile Water Innovation Laboratory The WINSSS Center benefits from seed funding for a mobile treatment facility that has been provided by the Massachusetts Clean Energy Center. The UMass Mobile Water Innovation Laboratory (MWIL) is a 36-ft trailer that has been customized to allow simulation of both an experimental and a parallel control water treatment train. Each train is able to treat up to 10 gallons per minute (gpm) of water using a wide range of conventional and advanced technologies.
The original purpose of the UMass MWIL was twofold: (1) to support the EPA center (WINSSS) by testing WINSSS-associated technologies in pilot scale and (2) to support NEWIN-associated companies by testing other technologies as requested in pilot scale. The need for a mobile unit is based on the site-specific nature of raw water quality and the large amounts of water used for testing at this scale, making transport of the requisite amount of raw water impractical.
Testing and calibration of the trailer units and sensors have been completed. The treatment scheme of the trailer consists of conventional technologies including a pre-oxidation contactor, up-flow roughing filtration, intermediate oxidation contactor, dual media sand-anthracite filtration, and a post-oxidation contactor. The installed components are designed in such a way that numerous operational and flow configurations are possible to evaluate a wide variety of treatment options. Advanced treatment technologies including ferrate oxidation, ion-exchange, electro-chemical oxidation, and ultra-filtration are currently being evaluated for installation during this current year as needed.
The first deployment for the trailer occurred on May 17-18, 2018 in Boston. After this initial event, we will use the trailer in support of: (1) testing WINSSS technologies such as ferrate under pilot scale conditions, and (2) direct assistance to small systems
Direct Assistance to Small Systems
With about 150,000 small systems in the US, it would be impossible for an entity like WINSSS to provide direct assistance to even a small fraction of all the sall s. However, WINSSS can help develop models for academic assistance and instruction much like RES’EAU has done in Canada with their Community Circle initiative. A key step in this direction has been taken with the offering of a special topics class at UMass on “Potable Water for Small and Disadvantaged Communities” the Fall 2017 semester.
Project A1: Implementing ferrate treatment of drinking water in the U.S.
Up to this point, project A1 has focused on bench-scale and small lab pilot-scale testing of ferrate. Most of this work was done on synthetic waters and about a dozen raw water sources. Two of these (Amherst and Gloucester, MA) have been studied in greater detail using a continuous flow treatment system. On-site piloting of ferrate treatment has proceeded. In year 4, the PIs met with MA regulatory officials and officials from two candidate piloting sites. Tentative approval was given for a 3-season pilot study at Plainville MA (groundwater) and Gloucester MA (surface water). Both locations are treating water with high DBP precursor levels and could benefit from the use of a non-chlorine pre-oxidant such as ferrate. The on-site piloting would be done with the WINSSS mobile lab (MWIL).
Project A2: Simultaneous Removal of Inorganic Contaminants, DBP Precursors, and Particles in Alum and Ferric Coagulation
This research outcome indicates the effect of the interaction between inorganic and organic contaminant and the interaction between the contaminant ions and the precipitates. The inorganic ions can interact with NOM in various ways (e.g., competitive, neutral, and synergistic). It was also revealed the interaction with the precipitate can affect the extent of precipitation. NOM presence enhanced the precipitation resulting in more precipitation occurred while As(III) retarded the iron flocculation process. However, it is still necessary to examine the zeta potential changes under different condition. Zeta potential will be measured in kinetic experiments and it will be an important step to demonstrate the hypothesis of flocculation characteristics. Additionally, dry-precipitate analysis will be performed such as scanning electron microscopy (SEM), x-ray diffractometry (XRD), and Fourier-transform infrared spectroscopy (FTIR). This analysis will provide an opportunity to understand the precipitate characteristic changes in chemical structural aspect.
Overall, the results are quite important to the operation of small utilities who might consider the use of precipitative coagulation with either aluminum or iron salts to remove both NOM and one of the inorganic contaminants studied (chromium arsenic, and fluoride). In general, the results show that one or the other coagulant can successfully remove, to a substantial degree, concentrations of both NOM and the inorganic constituent of interest. As the research develops further, we expect to develop guidelines for small utilities to assist them in choosing operating parameters, including the type of coagulant, its dose, and the optimal range of pH values, to simultaneously remove NOM and one of the inorganic contaminants.
Project A3: Contaminant reduction, life cycle impacts, and life cycle costs of ion exchange treatment and regeneration
Project A3 was complete as of the end of Year 3. Please refer to prior WINSSS annual reports for a description of this project’s results and conclusions.
Project A4: Natural Filtration Impacts on Post Disinfection Water Quality in Small Systems
Auburn and Nebraska City, two small communities in Nebraska, were selected. The towns of Auburn and Nebraska City draw their drinking water from a series of wells located on the bank of the Little Nemaha River, and on the bank of the Missouri River, respectively. The monitoring started in May 2016. Bi-weekly samples were only collected in May 2016 (moderately high flow period), while monthly samples were collected throughout the study (low flow period).
Preliminary results, May 2016 to July 2017, showed the ability of the two RBF systems to improve the quality of the source water. Turbidity, total and dissolved organic carbon, total coliform and E. coli significantly decreased after the two RBF systems. Turbidity decreased from 122-536 NTU to < 1 NTU at both RBF systems. In addition, total coliforms and E. coli were completely removed after filtration. Higher removal of dissolved organic carbon was observed at the RBF system in Auburn compare to the RBF system in Nebraska City. Dissolved organic carbon decrease from 4.2-6.1 to 0.5-0.7 mg/L in Auburn and from 3.8-4.8 mg/L to 2.1-2.6 mg/L in Nebraska City.
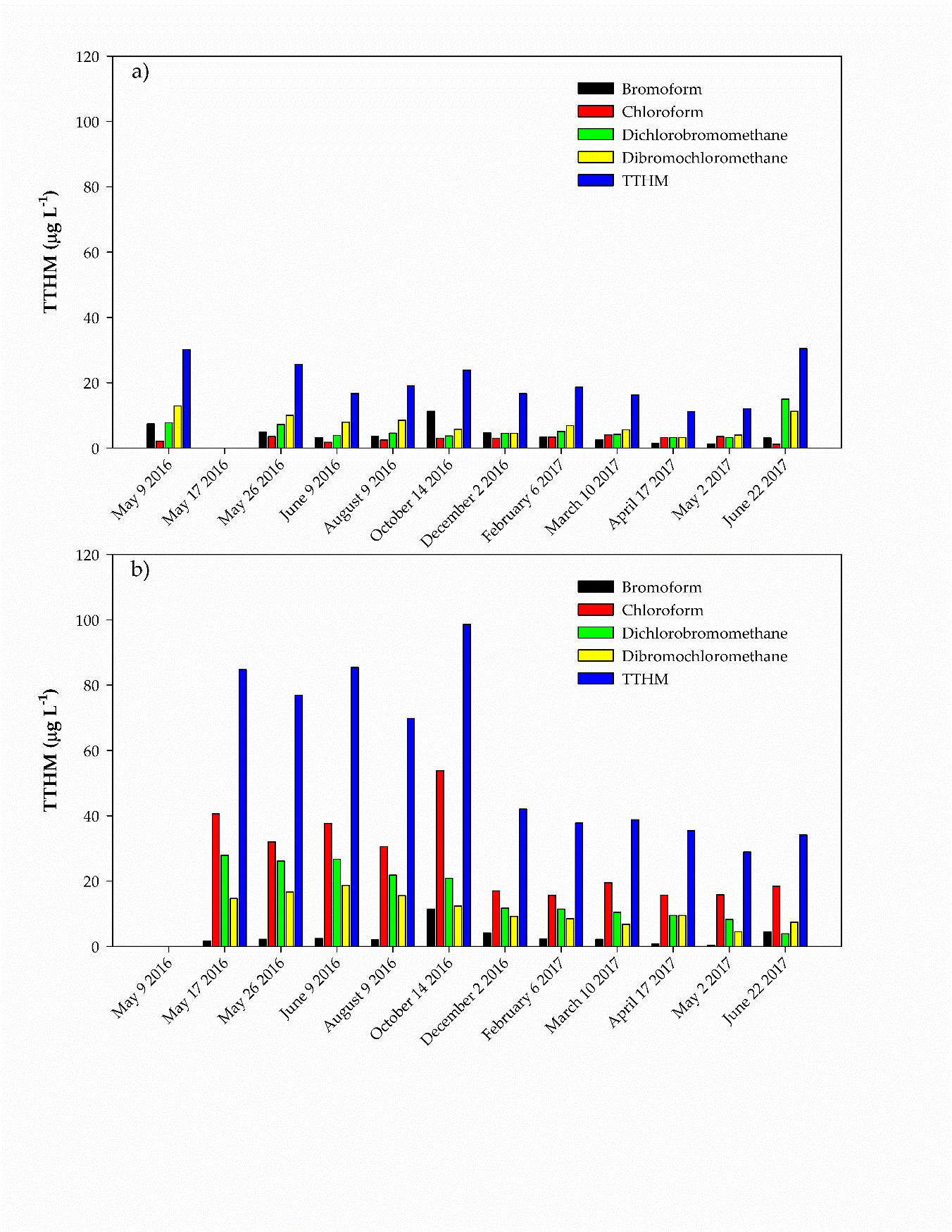
Among the trihalomethanes (THMs) investigated, bromoform ranged between 0.37 and 18.44 µg/L at both facilities, while higher concentrations of chloroform, dibromochloromethane, and dichlorobromomethane were detected in Nebraska City compared to Auburn. Chloroform, ranging between 15.0 and 53.82 µg/L, was the most abundant THM. The total concentration of THMs ranged between 11.17 and 30.15 µg/L in Auburn and between 22.88 and 98.54 µg/L. This high level of THM in Nebraska City can be related to the higher concentration of dissolved organic carbon observed after filtration in Nebraska City compare to Auburn. In addition, the high level of THM observed during Year 1 in Nebraska City appeared to be related to different groundwater flow regimes and well operational strategies in Year 1 as compared to Year 2. These resulted in higher levels of organic carbon observed during the first year and thus the higher THM levels.
Figure 2. Seasonal variations of Chloroform at Nebraska City (NC) and Auburn (AU).
The two RBF facilities represent valuable and cost-effective alternatives to enhance the quality and the safety of the feed water. Besides being able to remove turbidity and bacteria, i.e., total coliforms and E. coli, the two RBF facilities show a positive impact on THMs removal by reducing TOC and DOC levels.
The communities that are being monitored are in the Missouri Valley region, where surface water treatment plants typically must use enhanced coagulation to remove sufficient NOM to avoid DBP rule violations. The primary data helps document the fact that in many cases an RBF can provide water that results in DBP concentrations that meet Stage 1 standards.
Table 2 summarizes how RBF compares to competing processes for small water treatment plants
| Problem or Contaminant(s) addressed | Use of natural filtration of a surface water to remove contaminants, including natural organic matter that can result in DBP formation, to result in a higher quality water. |
| Size of Plant Applicability | Natural filtration is a treatment technology that has been used for communities of various sizes to fully treat or pre-treat the surface water before supply. River bank filtration is ideal for small communities that are located near a surface water. |
| Comparison to competing technologies | Competing technologies for surface water treatment include coagulation followed by conventional treatment (sedimentation, filtration) to remove NOM in the water. |
| Operator level Required (1 to 5) | RBF systems offer the potential to minimize the additional water treatment required beyond disinfection, potentially lowering the required operator level to 1 or 2. In some cases additional treatment including filtration and iron and manganese removal may still be required. |
| Problems with intermittent operation | RBF is a good process for intermittent operation based on the relative simplicity to the above ground system operation. |
| Residuals Management. | No residuals produced by RBF. |
| Energy Use | Electricity for the pumping will be dependent upon the well characteristics. In many cases, the electricity use will be less than a conventional water treatment plant because all unit operations in a conventional treatment plant may not be needed in RBF. |
| Cost Comparison | Requires electric power and maintenance, but less labor, chemical, and residual costs as compared to a conventional treatment system. |
| Health Benefits | Reducing disinfection byproducts such as THMs and haloacetic acids (HAAs) leads to less chance of carcinogenic substances in the water |
| Monitoring Required | Monitoring of regulated chemicals and microbial indicators in the river water and bank filtrate is required recommended. |
| Regulatory Issues | None expected. Typically determining if a water is under the influence of a surface water will be required. |
WINSSS Project B: Non-Treatment Innovations: Overcoming Regulatory Barriers, Software for Improved Asset Management, and Distributed Sensing Networks
Project B1: A Standardized Approach to Technology Approval
The results from the survey of state regulatory agencies was completed and the study was published in the August 2017 issue of the Journal of American Water Works Association. The paper is titled “State Barriers to Approval of Drinking Water Technologies for Small Systems.” The DOI is: https://doi.org/10.5942/jawwa.2017.109.0096.
This paper was selected by AWWA as the best paper in the small system division, and was recognized at the 2018 AWWA Annual Conference.
The workgroup has made significant progress toward engaging the states, as well as other stakeholders, in the idea of developing a national system for technology acceptance. The workgroup is following two distinct tracks forward: shared technology acceptance and data sharing/portal.
They have created a sub-group to explore the potential of working with the Interstate Technology and Regulatory Commission (ITRC), using their model of national teams of stakeholders who will evaluate specific technologies, where appropriate. This process is generally a 3-year program that leads to development of best practices and best available technologies for the specific technology type being evaluated.
Recognizing that the ITRC approach will likely not be appropriate for all new technologies, the workgroup focused on determining an acceptable approach and technology type that would be best suited to the ITRC model. Darrell Osterhoudt of ASDWA led a sub-group of seven states in developing a proposal for ITRC. This sub-group came up with a concept proposal related to an initial technology, micro- and ultrafiltration, that systems would put in to meet the requirements of LT2. The proposal was submitted to ITRC on July 7, 2017 and made it through their first cut of review. Discussions between this sub-group and the ITRC concerning the proposal and potential financial support for the proposed tasks are on-going. In addition, the workgroup has discussed collaborating with ECOS to develop a new sister organization to ITRC related to drinking water technology acceptance.
The New England workgroup, facilitated in part by Dave Reckhow, has been working with the Massachusetts Water Cluster, EPA Region 1, and ASDWA, toward developing a common set of standards acceptable to all the New England states. The New England workgroup has focused on accommodating demands for approval of ultraviolet treatment, especially for meeting the 4-log viral inactivation requirements of the Ground Water Rule. The challenge is that without a widely approved program for certifying UV validation facilities or national standards for the validation process, the states are faced with the challenge of independently reviewing and approving UV validation reports.
The workgroup, which has national representation, has developed momentum and will likely continue to meet into the foreseeable future. We anticipate the workgroup going well beyond the initial goals for this project, becoming a focal point for the industry in affecting real changes in the way technology is accepted at the state level to the benefit of small systems.
The public health impact of this task will be to help standardize the process of the approval of innovative technologies by state regulatory agencies. This may have the impact of a more rapid implementation into actual small systems of a wide range of technologies that reduce risk to public health.
Project B2: Simplified Data Entry System
A version of a smart phone app to allow for data collection and data entry with both android and iOS versions has been developed by the Center for Educational Software Development (CESD) at the University of Massachusetts, Amherst. During the summer of 2016 and then during the spring of 2017, undergraduate students tested the app as they collected asset data for real utilities. Brief case studies based on the use of the app have been posted to the WINSSS website. A final version of the app was prepared during the fall of 2017. It is available at no cost in android and iOS versions through the app stores.
By making it easier for small communities to use asset management tools, this offers the potential to gain the benefits expected from asset management. These benefits include better financial planning and improved routine maintenance of facilities. A public water supplier with a strong financial basis and good routine maintenance is less likely to experience deficiencies that result in health-based MCL violations; thus it is reasonable to expect that this new app will have positive public health impacts.
Project B3: A Distributed Sensing and Monitoring System
Sensors ideal for use in small water systems should be user-friendly, cost-effective, sensitive, specific, and capable of real-time and continuous monitoring of the target analyte. While there are many new sensing technologies available for detection of nitrate, few can simultaneously fulfill these stringent requirements. Driven by this need, we recently developed an electrochemical nitrate sensor that can potentially be used in small systems. The sensing mechanism is based on the electrocatalytic reduction of nitrate on a silver-modified electrode. The first generation sensor was fabricated on silver and carbon fiber ultramicroelectrodes and the results were published in Analytica Chimica Acta in 2015 [1]. Although there are merits in employing ultramicroelectrodes in nitrate analysis, it is more user-friendly and cost-effective to fabricate the sensor on paper-based electrodes. The second generation paper-based nitrate sensor was fabricated via electrodeposition of silver nanoparticles on a screen printed carbon electrode. The results were obtained using either a benchtop or handheld potentiostat and with a custom-built electrochemical cell with a low volume requirement (<10 µL). The as-fabricated sensor showed good response to nitrate in 0.1 M sodium sulfate under standard lab conditions. While the initial findings were encouraging, it was necessary to systematically optimize the sensor and evaluate its response to nitrate in complex samples such as soil extracts.
The focus of our research in Year 4 was to expand the use of this sensor for the analysis of nitrate in soil samples. Results suggest that amount of extraction solvent used can affect the nitrate extraction efficiency. To ensure proper extraction of nitrate from complex solid samples such as soil, the mass/volume ratio between the sample and the extraction medium has to be optimized.
WINSSS Project C: Assessment and Development of Treatment Processes for DBP Precursor and DBP Removal in Small Water Systems
ED Pretreatment. ED pretreatment was conducted with TH100-SNOM and TH100-LHA feed waters for 80% and 90% water recovery with 80% and 90% conductivity removal. Table 5 shows NOM mass loss (%) and specific energy consumption (SEC) for all eight conditions at which experiments were performed. NOM mass loss in the ED is due to NOM adsorption onto the membrane surface. Between SNOM and LHA, SNOM mass loss is less than LHA loss in all experimental conditions. The hydrophobic NOM (LHA) shows more adsorption onto the membrane surface in this research. NOM mass loss increases at higher water recovery and higher conductivity removal. Increasing water recovery and degree of conductivity removal requires extended operation of the ED system. Therefore, NOM has more chance to absorb onto the membrane surface. Besides, the ionic strength in the concentrate increases as ED separates more ions, so NOM adsorption increases due to the NOM configuration becoming coiled and compacted in high ionic strength condition. Specific energy consumption increases when the target water recovery and conductivity removal goal increase. ED pretreatment should achieve a certain degree of inorganic ion removal with minimal NOM adsorption and low energy consumption to minimize fouling in the NF process. The experiments with 80% water recovery and 80% conductivity removal showed the least NOM mass loss with proper inorganic ion removal; therefore, the subsequent NF experiments used this condition as ED pretreatment for both types of NOM.
| Type of NOM | Water Recovery | Conductivity Removal (%) | NOM Mass Loss (%) | SEC (kWh/m3) |
| SNOM | 80 | 80 | 1.20 | 0.212 |
| 90 | 2.88 | 0.215 | ||
| SNOM | 90 | 80 | 2.15 | 0.242 |
| 90 | 3.63 | 0.248 | ||
| LHA | 80 | 80 | 4.56 | 0.213 |
| 90 | 5.67 | 0.218 | ||
| LHA | 90 | 80 | 14.8 | 0.246 |
| 90 | 16.34 | 0.254 |
A coupled ED and NF/RO system
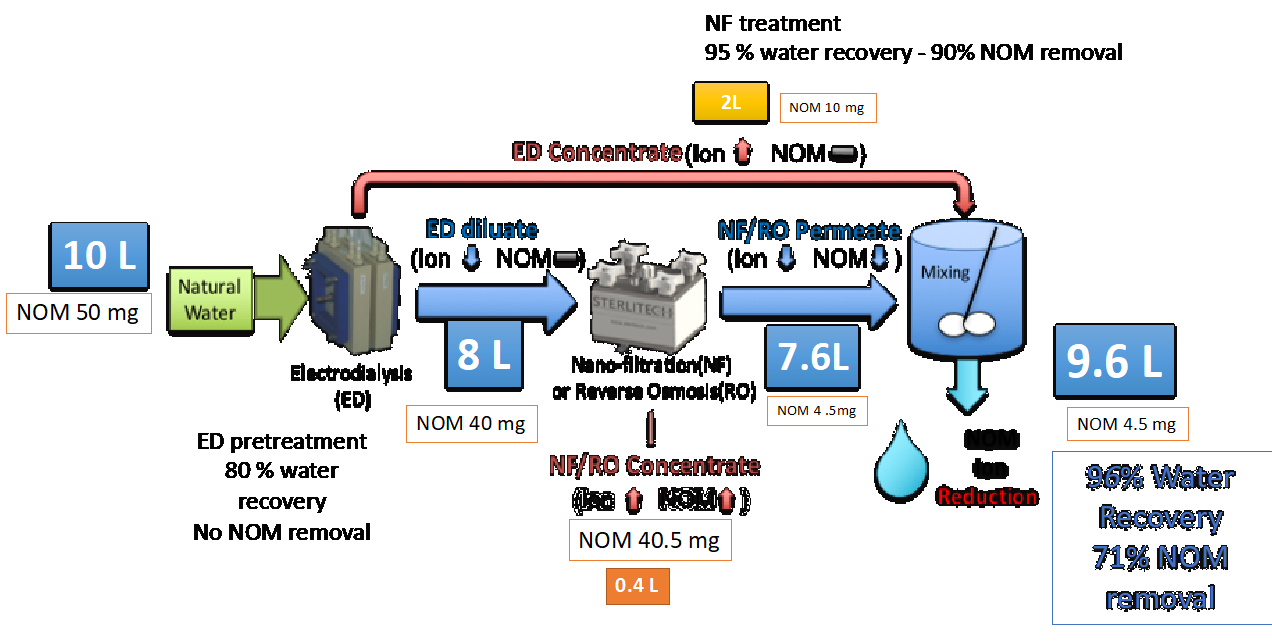
Based on ED pretreatment and NF/RO experiment results, a coupled ED-NF/RO system can achieve over 70% of NOM removal while retaining more than 80% of ions from the original feed water (Figure 23). The NOM removal efficiency and water recovery can be controlled based on the system design, type of NF or RO membranes, and ED operation condition.
Figure 23. Example calculation based on experimental results
The proposed hybrid ED/NF system for NOM removal is particularly well-suited for small utilities that have difficulty meeting the current regulations for DBPs and NOM removal. Membrane systems are cost-effective for small water systems because they can be operated and monitored remotely. Because chemical based treatment systems tend to exhibit strong economies of scale whereas membrane systems do not, the economics of membrane systems favors small water systems. Finally, although they are a new technology, membranes are easier to operate and require less knowledge to operate than chemical based treatment system, thereby furthering the value of this ED/NF hybrid treatment system for small water systems.
Journal Articles: 19 Displayed | Download in RIS Format
| Other center views: | All 48 publications | 19 publications in selected types | All 19 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Almassi S, Li Z, Xu W, Pu C, Zeng T, Chaplin B. Simultaneous Adsorption and Electrochemical Reduction of N-Nitrosodimethylamine Using Carbon-Ti4O7 Composite Reactive Electrochemical Membranes. ENVIRONMENTAL SCIENCE & TECHNOLOGY 2019;53(2):928-937. |
R835602 (Final) |
Exit Exit |
|
|
Almassi S, Samonte P, Li Z, Xu W, Chaplain B. Mechanistic Investigation of Haloacetic Acid Reduction Using Carbon-Ti4O7 Composite Reactive Electrochemical Membranes. ENVIRONMENTAL SCIENCE & TECHNOLOGY 2020;54(3):1982-1991. |
R835602 (Final) |
Exit Exit |
|
|
Dasi E, Cunningham J, Talla E, Ergas S. Autotrophic denitrification supported by sphalerite and oyster shells:Chemical and microbiome analysis. BIORESOURCE TECHNOLOGY 2023;375(128820). |
R835602 (Final) |
Exit |
|
|
Devi, P., Thakur, A., Lai, R. Y., Saini, S., Jain, R., Kumar, P. Progress in the Materials for Optical Detection of Arsenic in Water.Trends in Analytical Chemistry 2018; 110: 97-115 |
R835602 (2018) |
Exit |
|
|
Goodwill JE, Jiang Y, Reckhow DA, Gikonyo J, Tobiason JE. Characterization of particles from ferrate preoxidation. Environmental Science & Technology 2015;49(8):4955-4962. |
R835602 (2016) |
Exit |
|
|
Goodwill JE, Jiang Y, Reckhow DA, Tobiason JE. Laboratory assessment of ferrate for drinking water treatment. Journal: American Water Works Association 2016;108(3):E164-E174. |
R835602 (2015) R835602 (2016) R835172 (Final) |
Exit Exit |
|
|
Goodwill JE, Mai X, Jiang Y, Reckhow DA, Tobiason JE. Oxidation of manganese(II) with ferrate: stoichiometry, kinetics, products and impact of organic carbon. Chemosphere 2016;159:457-464. |
R835602 (2015) R835602 (2016) R835172 (Final) |
Exit Exit Exit |
|
|
Jiang Y, Goodwill JE, Tobiason JE, Reckhow DA. Effect of different solutes, natural organic matter, and particulate Fe(III) on ferrate(VI) decomposition in aqueous solutions. Environmental Science & Technology 2015;49(5):2841-2848. |
R835602 (2016) R835172 (Final) |
Exit Exit Exit |
|
|
Jiang Y, Goodwill JE, Tobiason JE, Reckhow DA. Bromide oxidation by ferrate(VI): the formation of active bromine and bromate. Water Research 2016;96:188-197. |
R835602 (2016) R835172 (Final) |
Exit Exit Exit |
|
|
Jiang Y, Goodwill JE, Tobiason JE, Reckhow DA. Impacts of ferrate oxidation on natural organic matter and disinfection byproduct precursors. Water Research 2016;96:114-125. |
R835602 (2016) R835172 (Final) |
Exit Exit Exit |
|
|
Li, F., Yu, Z., Han, X., Lai, R. Y. Electrochemical Aptamer-based Sensors for Food and Water Analysis:A Review Analytica Chimica Acta, 2018. |
R835602 (2018) |
Exit |
|
|
Lofti HR, Zhad Z, Lai RY. Hexavalent Chromium as an Electrocatalyst in DNA Sensing. Analytical Chemistry 2017;89(24):13342-13348. |
R835602 (2018) |
Exit Exit |
|
|
Lynn W, Heffron J, Mayer B. Electrocoagulation as a Pretreatment for Electroxidation of E. coli. WATER 2019;11(12). |
R835602 (Final) |
Exit Exit |
|
|
Plazas-Tuttle J, Das D, Sabaraya I, Saleh N. Harnessing the power of microwaves for inactivating Pseudomonas aeruginosa with nanohybrids. ENVIRONMENTAL SCIENCE-NANO 2018;5(1):72-82. |
R835602 (Final) |
Exit Exit |
|
|
Ringenberg, D., Wilson, S., and Dvorak, B. (2017) “State Barriers to Approval of Drinking Water Technologies for Small Systems,” Journal of the American Water Works Association, 109 (8) E343-E352, August, https://doi.org/10.5942/jawwa.2017.109.0096. |
R835602 (2017) R835602 (2018) |
Exit |
|
|
Ryan D, Maher E, Heffron J, Mayer B, McNamara P. Electrocoagulation-electrooxidation for mitigating trace organic compounds in model drinking water sources. CHEMOSPHERE 2021;273. |
R835602 (Final) |
Exit Exit |
|
|
Zhang J, Tejada-Martinez AE, Lei H, Zhang Q. Indicators for technological, environmental and economic sustainability of ozone contactors. Water Research 2016;101:606-616. |
R835602 (2015) R835602 (2016) |
Exit Exit Exit |
|
|
Zhang, J., A. E. Tejada-Martinez, H. Lei, Q. Zhang, 2016. Indicators for technological, environmental and economic sustainability of ozone contactors, Water Research, 101, 606-616. |
R835602 (2017) |
not available |
|
|
Jiang Y, Goodwill JE, Tobiason JE, Reckhow DA. Comparison of the effects of ferrate and ozone pre-oxidation on disinfection byproduct formation potentials. Water Research 2019; 156: 110-124. |
R835602 (2018) |
Exit |
Supplemental Keywords:
Drinking Water, Small systemsRelevant Websites:
Twitter account @tech4smallwater Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- 2018 Progress Report
- 2017 Progress Report
- 2016 Progress Report
- 2015 Progress Report
- Original Abstract
19 journal articles for this center