Grantee Research Project Results
2015 Progress Report: Human Neural Stem Cell Metabolomic, Cellular and Organ Level Adverse Outcome Pathway Relationships for Endocrine Active Compounds
EPA Grant Number: R835551Title: Human Neural Stem Cell Metabolomic, Cellular and Organ Level Adverse Outcome Pathway Relationships for Endocrine Active Compounds
Investigators: Stice, Steve , Smith, Mary Alice , Lu, Kun , Zhao, Qun
Institution: University of Georgia
EPA Project Officer: Aja, Hayley
Project Period: September 1, 2013 through August 31, 2016 (Extended to August 31, 2017)
Project Period Covered by this Report: September 1, 2014 through August 31,2015
Project Amount: $799,938
RFA: Development and Use of Adverse Outcome Pathways that Predict Adverse Developmental Neurotoxicity (2012) RFA Text | Recipients Lists
Research Category: Human Health , Chemical Safety for Sustainability
Objective:
Our overall objective is to determine the effects of endocrine active compounds (EACs) on events and adverse outcome pathways, spanning key temporal windows of human neural development. We are using human neural cells for metabolomic, cell function (viability and neurite outgrowth), and in vivo chick embryo central nervous system assays to reach this objective and provide information on the mode of actions of EACs.
Progress Summary:
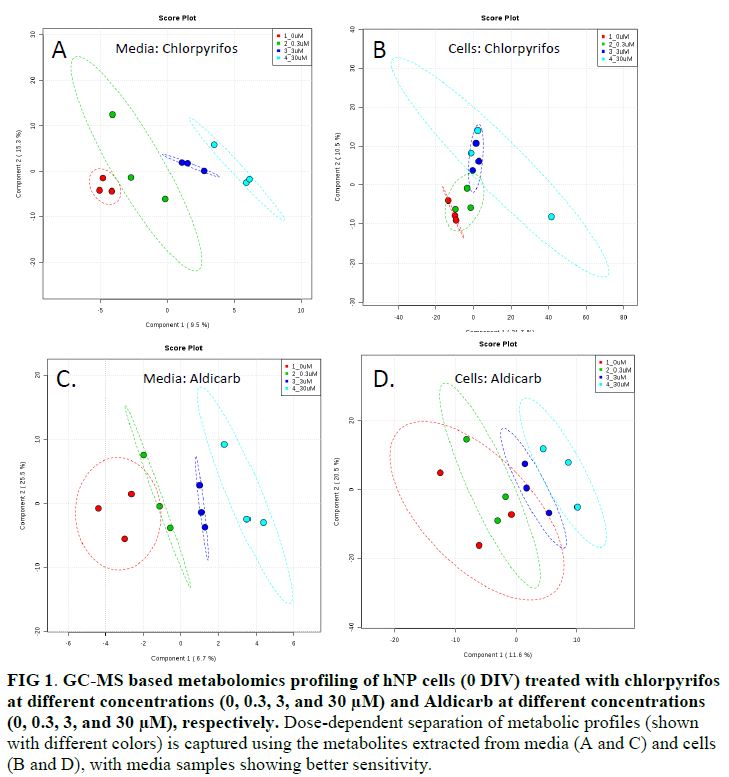
- Test EACs altered the metabolic profiles of hNP cells (FIG 1), as demonstrated with the GC-MS based metabolomics profiling results. As shown in the PLS-DA plots, control samples generally are well separated from cells treated with EACs (chlorpyrifos and aldicarb are illustrated in FIG 1).
- Metabolites extracted from both media and cells were used to examine the effect of exposure. Media samples generally offer better sensitivity to capture metabolic changes induced by test EACs, as shown by a good separation with the controls at lower doses (FIG 1).
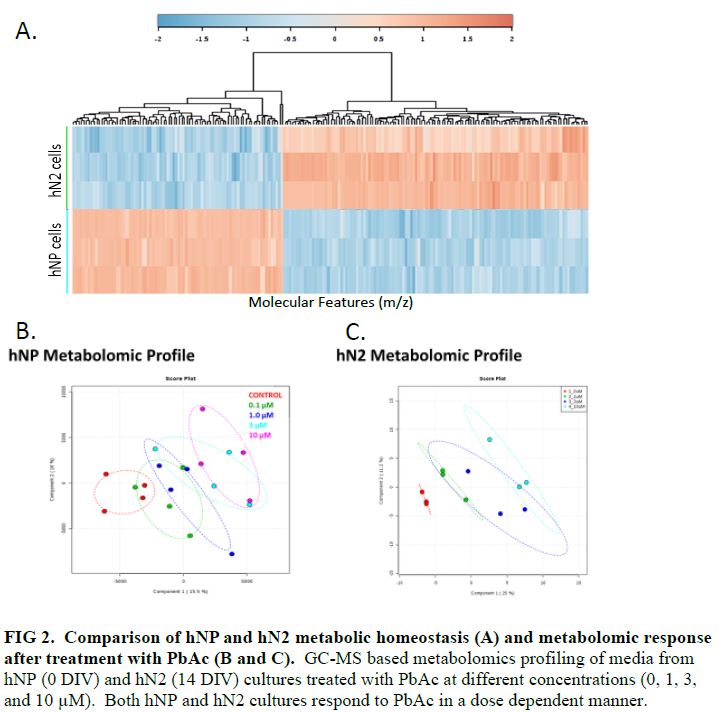
- hNP (DIV 0) and hN2 (DIV 14) cells have significantly different metabolic homeostasis (FIG 2A). Test EACs perturbed these metabolic profiles of both hNP and hN2 cells in a dose-dependent manner (FIG 2B and 2C).
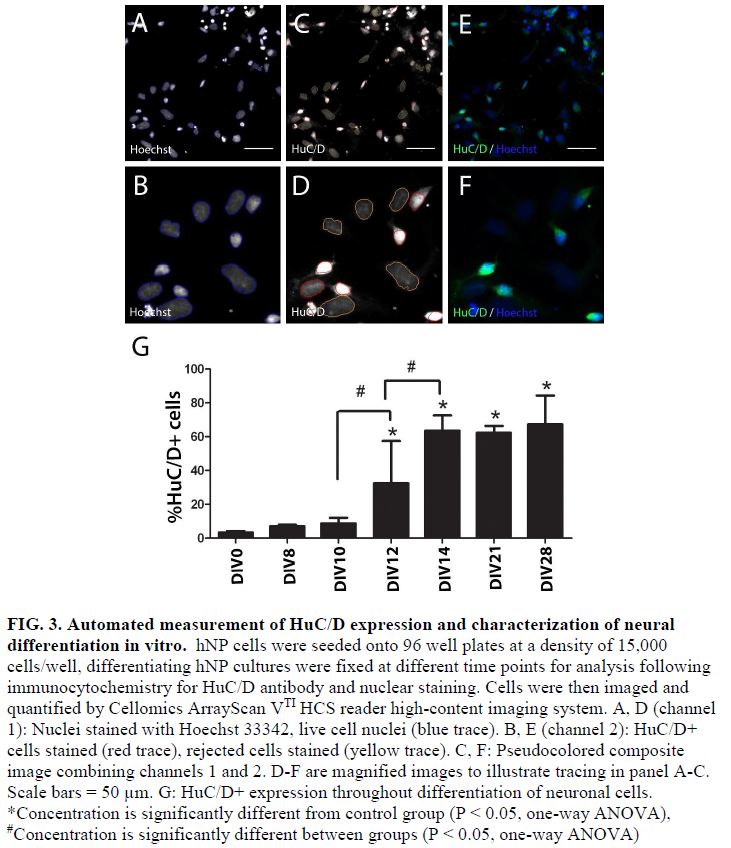
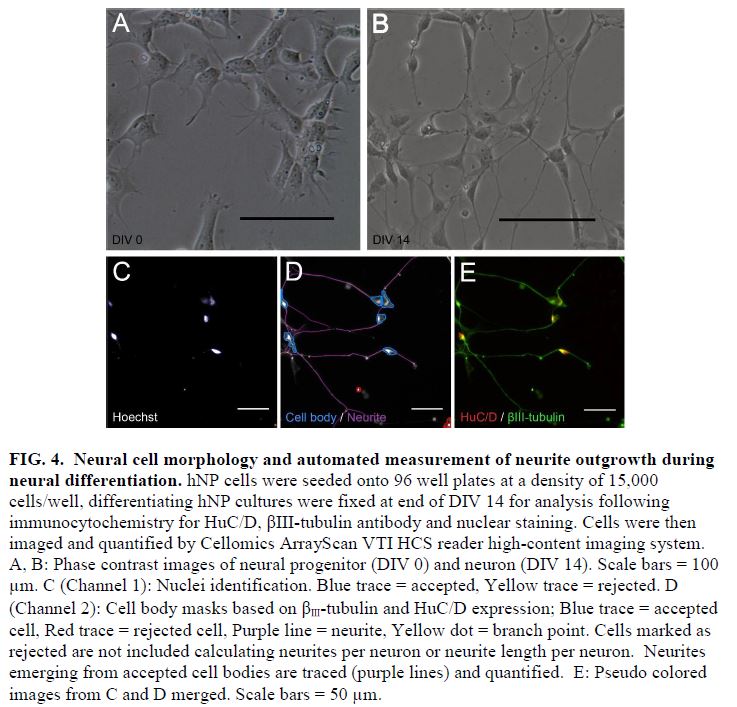
- During the normal course of maturation in vitro, expression of a post mitotic neuronal marker, HuC/D, increases over the initial 2 weeks of maturation, demarcating a timeframe for a DNT assay (FIG 3 and 4).
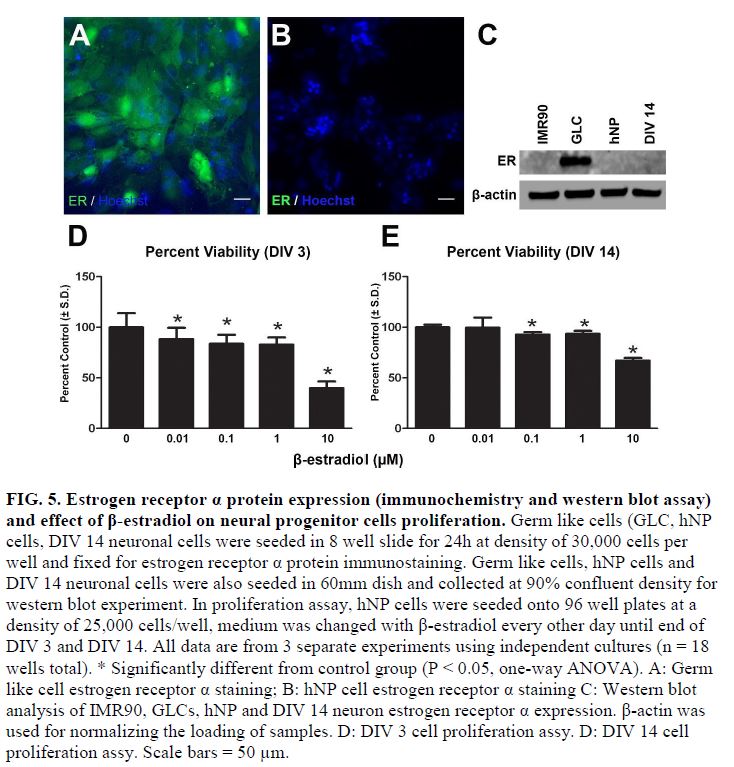
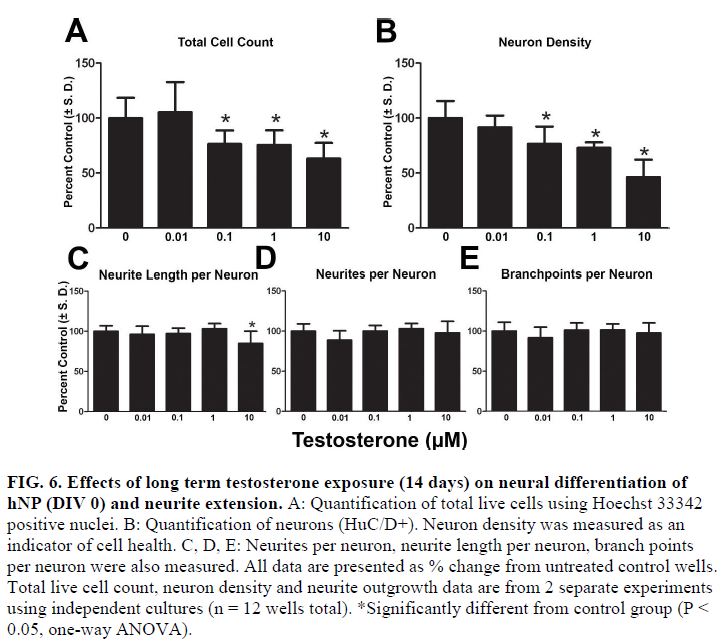
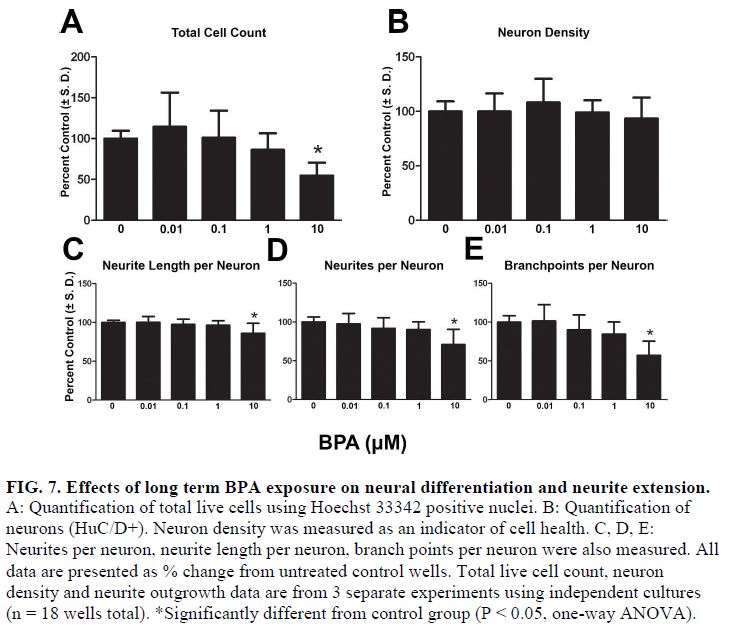
- Using this DNT model system, testosterone and β-estradiol inhibited neuronal maturation at micromolar levels while β-estradiol also disrupted neurite extension at 10 μM. Treating cells in this window with bisphenol A (BPA) significantly inhibited neurite outgrowth and branching in these continuum cultures but only at the highest concentrations tested (10 μM) (FIG 5, 6, and 7).
- Exploring neurotoxicity over 14 days provides new insights into the effects of EACs, suggesting β-estradiol specific cytotoxicity was higher than previously observed and manifested during the progenitor state through a non-estrogen receptor α (ER α) mediated effect on neurites over this DNT continuum (FIG 5).
- We conducted more experiments on tracking of human neural progenitor cell in chick embryos. SPIO-labeled NP cells can be reliably and repeatedly tracked throughout development after transplantation to the chicken embryo.
- Our first year’s work confirmed the feasibility of cooling the chick embryo for 1 hour decreased motion artifacts during MR imaging, without detriment to the developing chick. In the second year, we sought to longitudinally observe the growth of the chick central nervous system (CNS) by cooling and imaging the same embryos multiple times throughout development. We were able to image six individual embryos on day 10, 15, and 20 of embryonic development without retarding the chick's development or observing motion artifacts within our MR images. After imaging, MR image segmentation analysis allows us to calculate the volume of specific regions in the CNS. Key neuroanatomy including the telencephalon, lateral ventricles, cerebellum, and brainstem were all characterized at each of the three imaging time points. The volumetric characterization and trends were consistent between all six of the embryos imaged. These results validate our proposed procedure for longitudinal imaging and provide the basis for future toxicological studies that will dose the chick embryo in ova and longitudinally observe CNS growth under altered conditions.
We concluded:
- SPIO-labeled NP cells can be reliably and repeatedly tracked throughout development after transplantation to the chicken embryo.
- MR imaging is able to provide quantitative measurements, including 3D volume of key neuroanatomy and developmental biomarkers of chick embryo brains, without adverse effect from cooling embryos for 1 hour.
Future Activities:
Our major goal for the third year is to further characterize EACs in multiple key events (neurite outgrowth, metabolomics, and CNS development) using the unique assay systems established in the first year of this grant. We believe we can further understand mechanisms of action because we have initial data for chlorpyrifos and estrogen.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 32 publications | 7 publications in selected types | All 7 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Goodfellow FT, Simchick GA, Mortensen LJ, Stice SL, Zhao Q. Tracking and quantification of magetically labeled stem cells using magnetic resonance imaging. Advanced Functional Materials 2016;26(22):3899-3915. |
R835551 (2015) R835551 (2016) R835551 (Final) |
Exit Exit Exit |
Supplemental Keywords:
human neural progenitor cells, neurite outgrowth, window of susceptibility, metabolomics, endocrine active compounds, neural maturationRelevant Websites:
Regenerative Bioscience Center Exit
Stice Lab Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.