Grantee Research Project Results
2014 Progress Report: Human Neural Stem Cell Metabolomic, Cellular and Organ Level Adverse Outcome Pathway Relationships for Endocrine Active Compounds
EPA Grant Number: R835551Title: Human Neural Stem Cell Metabolomic, Cellular and Organ Level Adverse Outcome Pathway Relationships for Endocrine Active Compounds
Investigators: Stice, Steve , Smith, Mary Alice , Lu, Kun , Zhao, Qun
Institution: University of Georgia
EPA Project Officer: Aja, Hayley
Project Period: September 1, 2013 through August 31, 2016 (Extended to August 31, 2017)
Project Period Covered by this Report: September 1, 2013 through August 31,2014
Project Amount: $799,938
RFA: Development and Use of Adverse Outcome Pathways that Predict Adverse Developmental Neurotoxicity (2012) RFA Text | Recipients Lists
Research Category: Human Health , Chemical Safety for Sustainability
Objective:
Our overall objective is to determine the effects of endocrine active compounds (EACs) on events and adverse outcome pathways, spanning key temporal windows of human neural development. We are using human neural cells for metabolomic, cell function (viability and neurite outgrowth), and in vivo chick embryo central nervous system assays to reach this objective and provide information on EACs mode of actions.
- Characterize metabolomic profiles during windows of susceptibility (WOS) represented by neural tube stage neural progenitor (NP) cells and differentiated NP cells exposed to escalating doses of endocrine active compounds. These cell types represent significant windows of susceptibility (WOS) during neural development and builds on our previous study, EACs affecting human stem cell-derived germ cell progenitors.
- Determine endocrine active compound-induced alterations in key human neural cellular events and establish their associations with the metabolic changes. Differentiation (maturation), neurite outgrowth, and cell viability will be evaluated during early developmental neural windows of susceptibility (WOS) using human NP cells.
- Quantify the WOS vulnerability of the developing human neural cell chick chimera model. We will develop an innovative human NP cell/chick embryo model embryo spanning neural tube development through organogenesis.
Progress Summary:
OBJ 1: Characterize metabolomic profiles during windows of susceptibility (WOS) represented by neural tube stage neural progenitor (NP) cells and differentiated NP cells exposed to escalating doses of endocrine active compounds.
To determine the metabolomic profiles during WOS, we treated (0 DIV) hNP and 14 DIV (hN2) cells with varying doses of test chemicals to determine their sensitivity to increasing doses of endocrine active compounds. Examples of our results are shown below.
As shown in the PLS-DA in Figure 1, increasing concentrations of the positive control Bis-1 in hN2 cells analyzed for metabolomics changes in both treated cells and resulting media demonstrate the increasing metabolomic response in a dose dependent manner. However there was better separation of doses in the medium than the cells, suggesting that the medium is a more sensitive indicator of the metabolomics response than cells alone. hNP data not shown but is similar to the hN2 data.
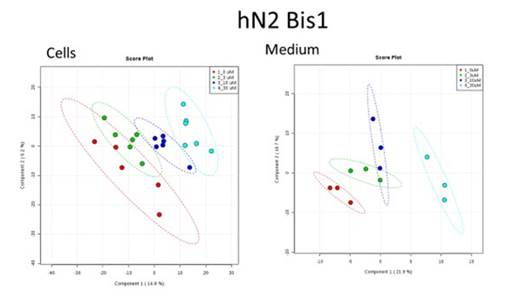
Figure 1. PLS-DA scores plot of hN2 cells after treatment with positive control Bis-1
at doses of 0,3,10 and 30 µM. Separation of dose groups (shown in different colors)
indicates different metabolomics response based on dose.
OBJ 2: Determine endocrine active compound-induced alterations in key human neural cellular events and establish their associations with the metabolic changes.
To determine whether metabolic changes after treatment with EACs are more sensitive indicators of toxicity than cellular events, cell viability and metabolic changes were determined in hNP and hN2 (Figures 2) cells for Bis-1. Metabolomic response to treatment with Bis-1 is at least as sensitive as cell viability in hNP cells.
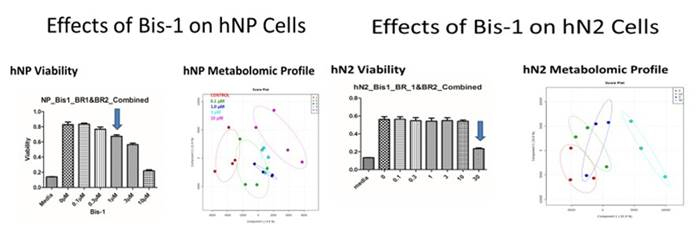
Figure 2. Sensitivity of cell viability compared to matabolomics response after treatment of hNP and hN2 cells with Bis-1.
BLue arrow indicates the lowest dose at which there is a statistical decrease in cell viability. For metobolomics response,
Neural maturation and Neurite outgrowth: Our objective here was to mimic early human neural development starting with proliferative neural progenitor (hNP) cells at 0 DIV and progressing through development of post mitotic, neurite extending neurons to closely represent the developmental continuum in an in vitro system suitable for high content imaging, in a format suitable for high throughput DNT screening 14 DIV and 28 DIV. Here we establish the developmental timeline from mitotic neural progenitors to postmitotic neurons and further investigate if this developmental phase is more sensitive to the compound Bis-1, previously demonstrated as detrimental to post thaw neurite recovery in cryopreserved mature neurons.
Our results show that in the normal course of maturation, expression of a post mitotic neuronal marker, HuC/D increase during the 14 DIV maturation period, with no further significant increase after that. We also show that chronic exposure of differentiating cells progenitor populations elicit toxicity at significantly lower doses than previously determined as toxic by neurite recovery assays. However the effects are limited to reduced neurite outgrowth and no significant lowering of neuronal population is observed at the end of the 14 DIV differentiation period. Thus we have developed a multiplex high content assay to determine the effects of toxicant in cells representative of the developing human neural tube and for the first time demonstrated that Bis-1 specifically affects neurite outgrowth at lower concentrations than can be detected using acute neurite recovery assays.
For objective 2, we have shown that metabolomics is at least as sensitive as cell viability in determining potential for toxic effects in both WOS as presented by hNP and hN2 cells. We are currently testing additional endocrine active chemicals in this system.
OBJ 3: Quantify the WOS vulnerability of the developing human neural cell chick chimera model. Although this objective was not scheduled to start until the second year we have made significant progress in the first year. A next-generation SPIO contrast agent Molday ION Rhodamine B (MIRB) allows for multimodal stem cell tracking with fluorescent microscopy and magnetic resonance imaging (MRI). Human neural progenitors NP cells were cultured with MIRB with in the proliferation medium. MIRB was observed in cytoplasmic vacuoles and Prussian blue staining confirming the presence of the iron nanoparticle after 48 hour incubation with MIRB. MIRB+ hNP cells were differentiated for 14 days in vitro (DIV). Upon 14 DIV differentiation, hNP cells adopted a more mature neuronal phenotype while retaining the MIRB nanoparticle. Additionally, MRI was employed to investigate anatomical and functional changes of developing neural systems of normal chick embryos, which will be used as benchmark for comparisons with those embryos affected by endocrine active compounds.
We concluded:
-
NP cells can be efficiently labeled with SPIO without harming the biological integrity of stem cells
-
SPIO-labeled NP cells can be tracked throughout development after transplantation to the chicken embryo
-
Experimental design is easily scaled up for toxicological studies
-
Foundation for a novel method of in vivo toxicology modeling
-
MRI is able to provide quantitative measurements, including 3D volume and developmental biomarkers (MR relaxation time, T1 and T2) of chick embryo brains.
Future Activities:
Journal Articles:
No journal articles submitted with this report: View all 32 publications for this projectSupplemental Keywords:
Relevant Websites:
Regenerative Research Bioscience Center Exit
The University of Georgia Stice Lab Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
