Grantee Research Project Results
2015 Progress Report: Cardiometabolic, Autonomic, and Airway Toxicity of Acute Exposures to PM2.5 from Multipollutant Atmospheres in the Great Lakes Region
EPA Grant Number: R834797C002Subproject: this is subproject number 002 , established and managed by the Center Director under grant R834797
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Health Effects Institute (2005 — 2010)
Center Director: Greenbaum, Daniel S.
Title: Cardiometabolic, Autonomic, and Airway Toxicity of Acute Exposures to PM2.5 from Multipollutant Atmospheres in the Great Lakes Region
Investigators: Harkema, Jack , Fink, Greg , Wagner, James
Institution: Michigan State University
EPA Project Officer: Chung, Serena
Project Period: December 1, 2010 through November 30, 2015 (Extended to December 31, 2016)
Project Period Covered by this Report: August 1, 2014 through July 31,2015
RFA: Clean Air Research Centers (2009) RFA Text | Recipients Lists
Research Category: Human Health , Air
Objective:
Our objectives in Project 2 arise out of GLACIER’s overarching hypothesis that the major air pollutants, fine particulate matter (PM2.5) and ozone (O3), are 1) capable of eliciting multiple important adverse cardiometabolic health effects that are dependent on 2) the local multipollutant milieu, 3) an individual’s pre-existing cardiovascular (CV) and metabolic condition (susceptibility factors), and 4) the interactive toxicity of PM2.5 and O3 coexposure. Goals of Project 2 are to determine the CV, autonomic nervous system (ANS), and airway toxicity in rats acutely exposed to concentrated ambient PM2.5 (CAP) from distinct multipollutant atmospheres commonly found in the Great Lakes Region of the United States. Our studies are extensions of our previous findings that CAP-induced alterations in heart rate variability are dependent on specific PM2.5 emission sources in distinct locations in the Great Lakes Region. We will use a mobile air research facility (AirCARE 1) that is fully equipped with inhalation toxicology and atmospheric monitoring labs to conduct toxicology studies of rats exposed to CAP derived from real-world PM2.5 in three distinct locations dominated by industrial/urban, transported/regional, or near-roadway/residential emission sources. Blood pressure, heart rate, heart rate variability, and direct measurements of autonomic nerve activity will be continuously monitored during CAP and/or O3 exposures in lean or obese rats with and without diet-induced facets of the cardiometabolic syndrome (CMS) (hypertension, insulin resistance, endothelial dysfunction), respectively. Acute functional responses will be measured by radiotelemetry and will be correlated with specific PM constituents and their emission sources determined for the same highly resolved 30-minute timeframes, thereby making associations of exposure and health effects especially robust. Studies will feature novel real-time sympathetic nerve recordings during PM2.5 and/or O3 inhalation exposure. In addition, our project will highlight the unique integrative capabilities of our research team to link specific health cardiovascular effects in a sensitive obese population with PM content by a combined technological expertise that is unavailable elsewhere. Our GLACIER project will extend and complement the research of lean and obese human subjects (Project 1), conducted at the same exposure sites, by making invasive and prolonged measurements that could not be practically or ethically done in humans (e.g., repeated CAP exposures, continuous recordings of CV and autonomic nerve function, and microscopic examination of multiple organs for exposure-related pathology). Our acute animal studies also will overlap and integrate scientifically with the animal toxicology study of long-term multipollutant exposures in Project 3.
Progress Summary:
There have been no changes to our study investigators in Year 5 of this project. Our objectives in Project 2 also have remained the same in Year 5. Studies have been designed to address GLACIER’s overarching hypothesis that the major criteria air pollutants, fine particulate matter (PM2.5) and ozone (O3), are 1) capable of eliciting multiple important adverse cardiometabolic health effects that are dependent on 2) the local multipollutant milieu, 3) an individual’s pre-existing cardiovascular (CV) and metabolic condition (susceptibility factors), and 4) the interactive toxicity of PM2.5 and O3 co-exposure. As in Year 4, research in Year 5 addressed all four of these hypotheses by conducting both field-exposure studies and mechanism-driven experiments. These studies were extensions of our previous findings in Years 1-3 that CAP-induced alterations in heart rate variability are dependent on specific PM2.5 emission sources in distinct locations in the Great Lakes Region. Our results in Years 1-5 have been designed to extend and complement the initial research of lean and obese human subjects (Project 1), by making invasive and prolonged measurements in laboratory rats/mice that could not be practically or ethically done in humans (e.g., repeated CAP exposures, semi-continuous cardiovascular recordings, and microscopic examination of multiple organs for exposure-related pathology). Our acute animal studies were also designed to contrast and complement the initial animal toxicology studies of long-term multipollutant exposures in Project 3.
In Year 5, we 1) continued our data analysis from the rat O3 and PM2.5 co-exposure studies that were conducted in a rural location (Dexter, MI; with transported rather than locally generated air pollution) and in an urban location (Dearborn, MI; with locally generated air pollution), 2) completed analysis of the pulmonary data garnered from our laboratory study that was designed to determine the metabolic and respiratory effects of O3 exposure in mice that are genetically prone to develop type two diabetes (T2D), and 4) completed another study that investigated the role of innate lymphoid cells in the development of O3-induced nasal type 2 immunity and eosinophilic rhinitis in mice. In addition, we continued our data analysis from an intraCLARC collaborative animal toxicology study with Harvard University that employed our rat model of diet-induced CMS and their inhalation exposure system to traffic emissions from the Boston Tunnel. A brief summary of specific Project 2 accomplishments in the last year and proposed future studies in the remainder of Year 5 (2015) and into our no-cost-extension year (2016) are briefly described below.
Study 1A: Rat inhalation toxicology study in a rural location (Dexter, MI; transported pollution). In June 2013, we initiated a rat inhalation toxicology study at our rural/regional exposure site in Dexter, MI, that was similar in design to that previously conducted in Dearborn MI. Repeated acute inhalation exposures of rats, fed normal (ND) or HFr-diets (HFrD), to O3/PM2.5, were performed for 19 consecutive weekdays, 8 h/day, in our mobile air research laboratory (AirCARE 1) parked in a rural setting in Dexter, MI. In contrast to the urban/industrial site in Dearborn, MI, with local traffic and industrial air pollutant emission sources, this Dexter exposure site is dominated by transferred air pollution from distant regional emission sources. Like the previous Dearborn study, cardiovascular responses (e.g., blood pressure, heart rate, heart rate variability) were measured every 5 minutes by radiotelemetry during and after daily exposures. The inhalation exposures were successfully completed in early July.
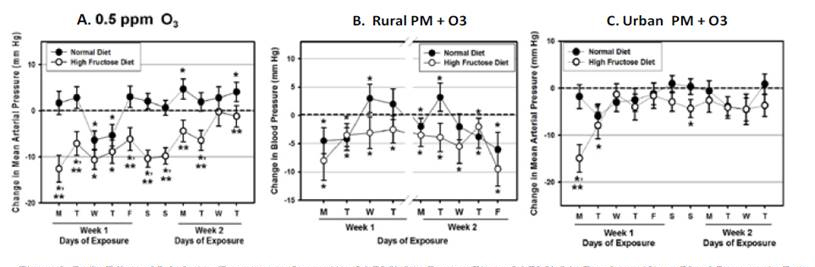
In Year 5 we focused on a re-analysis of blood pressure that we had previously reported in a preliminary form. Drs. Cathie Spino and Bin Nan from our Biostatistical and Data Management Core (BDM) at the University of Michigan completed a thorough quality control and statistical analysis of all the data collected from this study. We first used data from the first 9 days from this study to compare the responses to O3/PM2.5 in Dexter (Rural) to our previous data from rats exposed to O3 alone and to O3/PM2.5 in Dearborn (Urban). We found that inhalation of O3/PM2.5 in Dexter caused significant drops in mean arterial pressure (3-8 mm Hg) in rats fed HFrD that were maintained for most days of the exposures (Figure B, white circles). In contrast, both decreases and increases in blood pressure (3-5 mm Hg) were detected in ND rats during exposures (Figure 1B, black circles). These responses were similar to those observed in our O3/PM2.5 exposure study in urban/industrial Dearborn (Figure 1C) where ND rats had no exposure-related responses and HFrD quickly adapted after acute drops in blood pressure during the first day of exposure. In both rural Dexter and urban Dearborn, coexposure to PM2.5 diminished the vascular response induced by O3 exposure alone (compare Figures 1A, B to 1C).
Figure 1. Daily Effect of Inhalation Exposure to Ozone (a), O3/pm2.5 in Dexter, (b), or O3/pm2.5 in Dearborn (c) on Blood Pressure in Rats
Fed Normal (black circles) or High Fructose (white circles) Diets. Data is plotted as effect estimates on blood pressure (mmHg) of the indicated
xposure compared to filtered air (indicated by dotted line-zero axis). & indicates significant difference from Air-ezposed rats ont eh same diet;
** indicates differences from rats fed a Noram Diet.
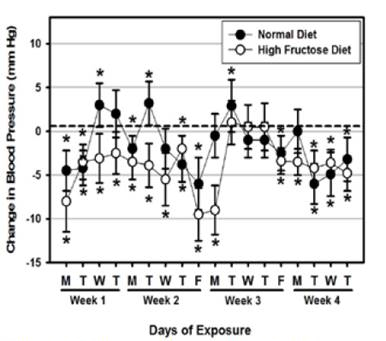
Exposure-related responses of decreased blood pressure persisted during a third and fourth week of exposure to O3/PM2.5 (Figure 2). Depression in blood pressure was similar in NF and HFrD rats (4-5 mm Hg) with increasing exposures by the end of Week 4.
Figure 2. daily effect of 4-week inhalation Exposure to O3/pm2.5
on BLood Pressure in Rats fed normal (black circles) or high fructose
(white circles) diets. Data is plotted as effect estimates on blood pressure
(mmHg) of the indicated exposure compared to filtered air (indicated by
dotted lines - zero axis). * Indicates significant difference from air-exposed
rats on teh same diet; ** Indicates significant difference from rats fed a normal diet
Study 1B: Associations of exposure/health outcomes in rats exposed to locally generated air pollution in an urban location (Dearborn, MI). With the help of Dr. Bin Nan in the Biostatistics and Data Management Core, and Dr. Morishita in the Exposure Core, cardiovascular responses and PM2.5 metrics (e.g., trace elements, gases, sources) collected over the same 30-minute timeframes were analyzed with linear mixed model approaches to determine the effect of PM2.5 emission sources on cardiovascular responses. Dr. Morishita determined that our field site at Salinas Elementary School in Dearborn, MI, was impacted by five major sources of PM2.5: secondary, urban dust, diesel/motor vehicle, refinery, and iron/steel. Our previous analyses showed that depression in blood pressure in both ND and HFrD rats was related to diesel/motor vehicle sources, and that traffic sources also were associated with decreased heart rate in HFrD (see details in Year 4 Annual Report). These analyses were extended in Year 5 to determine source-related outcomes in heart rate variability as measured by RMSSD, which reflects the integrity of vagus nerve-mediated autonomic control of the heart. PM2.5-related decreases in RMSSD were associated with traffic and refinery sources in ND rats, and with urban dust in HFrD rats (Figure 3). Assessment of 30-minute lag exposure effects further identified secondary sulfates to influence RMSSD in ND rats, whereas exposure-related effects were lost in HFrD rats. These data suggest that subjects with metabolic syndrome may be less sensitive to parasympathetic response induced by urban PM sources of PM2.5 than normal healthy subjects, and sensitivity for cardiovascular responses to specific PM sources is related to metabolic health.
| Element Associaations with HRV | ||
|---|---|---|
| Current | Lag | |
| HF diet | Mg, Fe | None |
| Normal diet | Ba, Mn, As | Bas, Mn, Sn, Ti |
| Source Associations with HRV | ||
| Current | Lag | |
| HF Diet | Urban Dust | None |
| Normal Diet | Vehicle, Refinery | Secondar, Vehicle, Refinery |
Study 2: Inhaled ozone caused more severe lung lesions in hyperglycemic, insulin-resistant KKAy mice as compared to normoglycemic, insulin-sensitive C57BL/6 mice. Epidemiological studies suggest that diabetics may be more susceptible to the adverse health effects of air pollution. The pathobiology underlying this susceptibility is yet unknown. In this study we tested the hypothesis that repeated inhalation exposures to a common gaseous air pollutant, ozone (O3), will induce more severe lung lesions in hyperglycemic, insulin-resistant mice as compared to normoglycemic, insulin-sensitive mice. Male C57BL/6, KK and KKAy mice were exposed to 0 ppm (filtered air; FA) or 0.5 ppm O3, 4h/day, for 13 consecutive weekdays. Two hours after the last exposure, mice were subjected to insulin tolerance tests (ITT) and then sacrificed 24 hours postexposure. Bronchoalveolar lavage fluid (BALF) was analyzed for inflammatory cells and cytokines, and lung tissues were processed for light microscopy, morphometry, and RT-PCR analysis.
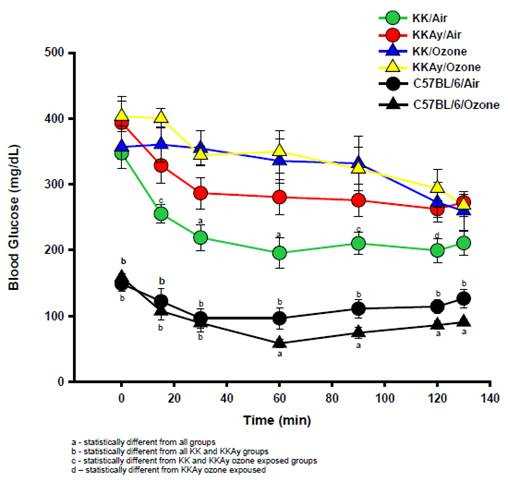
Normoglycemic, C57BL/6 mice exposed to FA or O3 had normal ITT indicating insulin sensitivity, while FA- and O3-exposed hyperglycemic KKAy mice had abnormal ITT demonstrating insulin resistance (IR). O3 induced IR in KK mice.
Figure 4. Blood glucose levels after intraperitoneal injection of insulin (insulin tolerance tests)
in KK, KKAy and C57BL/6 mice exposed to ozone or filtered air. Ozone exposure induced
insulin resistance in hypergycemic KK mice, but not in normoglycemic C57BL/6 mice. The insulin
resistance in KKAy mice exposed to air or ozone were similar in severity.
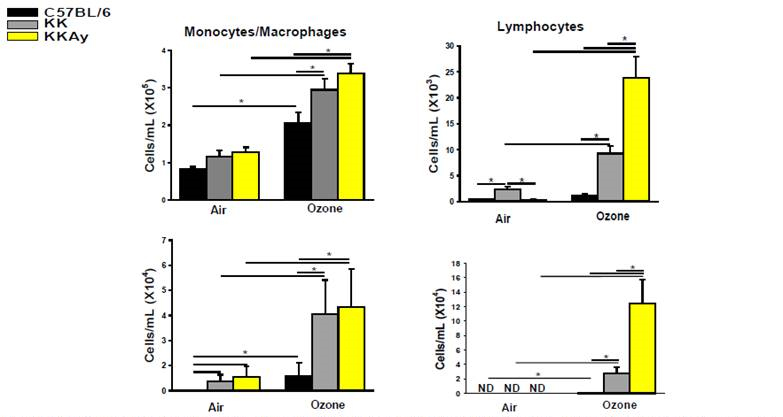
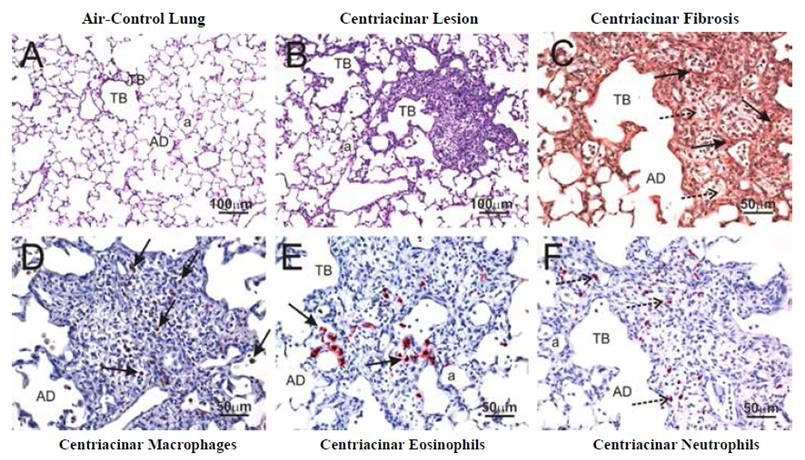
FA-exposed control mice had no exposure-related pulmonary pathology. FA-exposed C57BL/6 mice did, however, have slightly fewer neutrophils in BALF, as compared to FA-exposed KKAy and KK mice (Figure 5). In C57BL/6 mice, O3 caused modest increases in the number of BALF inflammatory cells (neutrophils, monocytes/macrophages) and minimal to mild centriacinar lung lesions without fibrosis. In contrast, O3 induced greater increases in BALF inflammatory cells (neutrophils, monocytes/macrophages, eosinophils, and lymphocytes) in KKAy and KK mice, as compared to similarly exposed C57BL/6 mice. In addition, O3 exposure induced an increase in IL-5 only in the BALF of KKAy mice. O3-exposed KKAy mice also had more severe centriacinar lung lesions that included marked eosinophilic and neutrophilic inflammation, epithelial hyperplasia, and peribronchiolar and alveolar fibrosis (Figure 6). O3-induced fibrotic lesions in KKAy mice were associated with conspicuous eosinophil infiltration and relative fold increases in mRNA expression of eotaxin, IL-13, and MMP-12, as compared to FA-exposed KKAy mice.
Figure 5. Inflammatory cells in the bronchoalveolar lavage fluid (BALF) of C57BL/6, KK and KKAy mice exposed to
ozone or filtered air. Ozone exposures caused an increase in BALF inflammatory cells in all strains of mice. Hyperglycemic
KKAy and KK mice had greater numbers of macrophages/monocytes, lymphocytes, neutrophils and eosinophilis in BALF
after ozone exposure as compared to similarly exposed nomoglycemic C57BL/6 mice.
Figure 6. Light phomicrographs of the lung from KKAy mice exposed to 0 ppm (A) or 0.5 ppm ozone (B-F) a, alveolus; AD, alveolar duct; TB,
terminal bronchiole. Ozone expsores induced fibrosis and marked neutrophilic and eosinophilic inflammation in the centriacinar regions of the lung.
In conclusion, hyperglycemic KKAy mice, that spontaneously develop type II diabetes, had significantly greater O3-induced lung lesions, including eosinophilic inflammation and fibrosis, as compared to similarly exposed, normoglycemic C57BL/6 mice. The lungs of KK mice also had greater pulmonary inflammation after ozone exposures as compared to similar exposed C57BL/6 mice, but the severity of the inflammation was not as severe as that in the KKAy mice. These results suggest that people with type II diabetes, or pre-diabetic conditions, may be more susceptible to the respiratory health effects caused by elevated concentrations of ambient O3.
Study 3: Ozone-induced eosinophilic rhinitis is dependent on innate lymphoid cells. Epidemiological studies suggest that elevated ambient concentrations of ozone are associated with activation of eosinophils in the nasal airways of atopic and nonatopic children. In a previous study, we reported that mice repeatedly exposed to ozone develop eosinophilic rhinitis and type 2 immune responses. In the present study we determined the role of innate lymphoid cells (ILCs) in the pathogenesis of ozone-induced eosinophilic rhinitis by using lymphoid-sufficient C57BL/6 mice, Rag2-/- mice that are devoid of T cells and B cells, and Rag2-/-Il2rg-/- mice that are depleted of all lymphoid cells including ILCs. Animals were exposed to 0 or 0.8 ppm ozone for 9 consecutive weekdays (4 h/day). Mice were sacrificed 24 h post-exposure and nasal tissues were selected for histopathology and gene expression analysis.
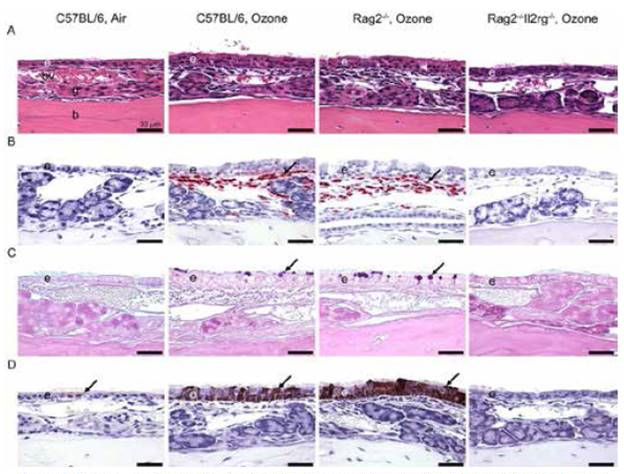
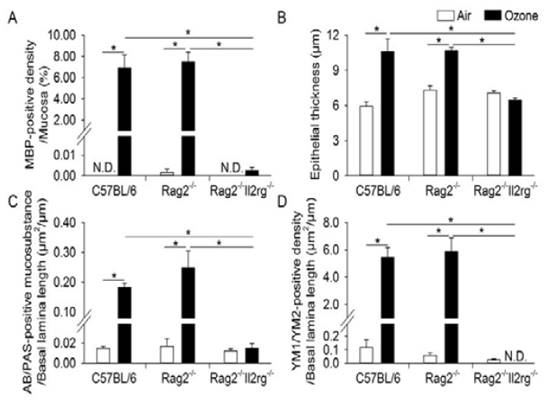
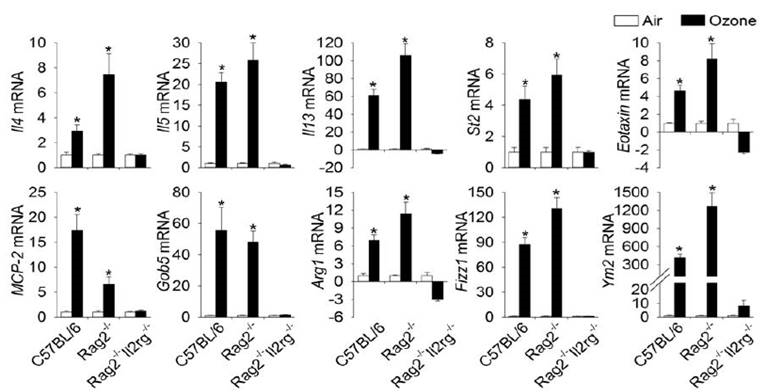
ILC-sufficient C57BL/6 and Rag2-/- mice exposed to ozone developed marked eosinophilic rhinitis and epithelial remodeling (e.g., epithelial hyperplasia and mucous cell metaplasia; Figures 7 and 8). Chitinase-like proteins (YM1/YM2) and alarmins (IL-33, IL-25, and TSLP) also were morphometrically increased in the nasal epithelium of ozone-exposed C57BL/6 and Rag2-/- mice. Ozone exposure elicited increased expression of Il4, Il5, Il13, St2, eotaxin, MCP-2, Gob5,Arg1, Fizz1, and Ym2 mRNA in C57BL/6 and Rag2-/- mice (Figure 9). In contrast, ozone-exposed ILC-deficient Rag2-/Il2rg-/- mice had no nasal lesions or overexpression of Th2- or ILC2-related transcripts. These results indicate that ozone-induced eosinophilic rhinitis, nasal epithelial remodeling, and type 2 immune activation are dependent on ILCs. This is the first study to demonstrate that ILCs play an important role in the nasal pathology induced by repeated ozone exposure.
(A) Hematoxylin and eosin (H&E) stain. Exposure of 0.8 ppm ozone for 9 days caused marked rhinitis and epithelial remodeling (i.e., hyperplasia, mucous cell metaplasia, and hyalinosis) in ILC-sufficient C57BL/6 mice and Rag2-/- mice. By contrast, ILC-deficient Rag2-/Il2rg-/- mice exposed to ozone had no nasal lesions.
(B) Immunohistochemical stain for major basic protein (MBP) in murine eosinophils. Large numbers of eosinophils were detected in the nasal mucosa of ozone-exposed C57BL/6 mice and Rag2-/- mice (arrows).
(C) Alcian Blue (pH 2.5)/Periodic Acid Schiff (AB/PAS) stain for acidic and neutral mucosubstances. C57BL/6 and Rag2-/- mice exposed to ozone stored AB/PAS-stained intraepithelial mucosubstances in the apical aspect of nonciliated epithelial cells (arrows).
(D) Immunohistochemical stain for chitinase-like 3 (YM1) and chitinase-like 4 (YM2) proteins. Air-exposed mice contained small amounts of YM1/YM2 proteins in the apical aspect of ciliated epithelial cells (arrows). YM1/YM2 proteins were found throughout the full thickness of the nasal transitional epithelium of ozone-exposed C57BL/6 mice and Rag2-/- mice (arrows). e, nasal epithelium; bv, blood vessel in lamina propria; g, gland in lamina propria; b, bone.
Figure 7. Light photomicrographs of eosinophilin inflammation and epithelial remodeling in the nasal mucosa
lining the lateral wall of the proximal nasal passages.
Figure 8. Morphometric evaluation for eostinophilic rhinitis and epithelial remodeling in the nasal
mucosa lining of the lateral wall of the proximal nasal passages. A. Eosinophil density in the nasal
mucosa (i.e., epithelium and lamina propria). B. Thickness of the nasal transitional epitheliom.
C. Volume density of AB/PAS-stained intrapethelial mucosubstances per length of basal lamina. D.
Volume density of YM1/YM2 proteines per lengs of basal lamina p valuers < 0.05. Data are express as
mean ± SEM (n = 6/group).
Figure 9. Gene expression analysis in the proximal nasal mucosa. Gene expression in the proximal nasal mucosa was analyzed for
eotaxin, St2, MCP-2, Gob5, Arg1 and Ym2 mRNA on SmartChip Real-Time PCR. System and for η4, η5, η13, and Fizz1 on ABI
PRISM 7900 HT Sequence Detection System. Genes showing statistical significances (air va. ozone/same strain, p values < 0.05).
Data are expressed as fold changes relative to their respective controls ± SEM (n = 6/group).
Collaborative Research Effort with Other Clean Air Research Centers
Toxicity of Traffic-Based Air Pollution in Rats with Diet-Induced Cardiometabolic Syndrome: In the Fall of 2012, investigators from GLACIER and Harvard University CLARC initiated an intraCLARC collaborative toxicology study under the principal direction of Drs. Jack Harkema (GLACIER) and John Godleski (Harvard University). Using our established high fructose-diet-fed rat model of CMS, the collaborative study was designed to determine if this dysfunctional cardiometabolic condition predisposes to the toxic effects of traffic-related air pollution and to identify underlying toxicological modes of action by which this may occur. Our goal is to discern if CMS renders the laboratory animal more susceptible to the cardiovascular, autonomic and airway toxicity of a multipollutant mixture of primary particles and secondary organic aerosols derived from traffic emissions in the Boston Tunnel. Data analysis is still underway and results from this study will be compared to similar studies in GLACIER’s Project 2. A brief description of the study design is presented below.
Animals: Twelve 200 gram Sprague-Dawley rats were obtained from Taconic Farms with implanted DSI telemeters capable of monitoring blood pressure, heart rate, and temperature. Another 36 animals were obtained from Taconic Farms without telemeters. All rats were fed a high fructose diet (Harlan TD.89247; 60% of calories comes from fructose) for 8 weeks prior to use in any experiments. The high fructose diet was provided by the GLACIER CLARC. Inhalation exposures were conducted at Harvard’s Boston Tunnel site (see below). Rats were weighed weekly, and 4 hrs of continuous data were collected from the telemeters in the animals at 2 week intervals. Rats were without food or water during the 5 hours of daily exposure, but when returned to their housing during nonexposure hours they were fed their specified diets.
Traffic-Related Urban Aerosol Particles (TRUAP) Exposure Protocol: Rats were continuously exposed to TRUAP or filtered air (FA) in single-animal plethysmographs for 5 hrs/day. TRUAP inhalation exposures are derived from the realtime ventilation exhaust of a moderate traffic density tunnel (with small positive road grade, approximately 2°), in the northeast United States. TRUAP consists of primary and secondary traffic-derived fine and ultrafine particles (1nm to 2.5 μm (PM2.5)). Twelve (12) animals (6/group TRAUP; 6/group filtered air all with implanted telemeters continuously monitoring blood pressure, heart rate, and temperature) were exposed each day, 4 days/week for three consecutive weeks. Another group of animals without telemeters, also will be exposed to filtered air or TRAUP for studies as described below. In prior TRUAP studies, concentrations averaged approximately 50 µg/m3 (combined primary and secondary particles), with standard deviations less than 25% of the mean for any component; however, during these fall exposures, the mass concentrations were unusually low averaging approximately 30 µg/m3. Exposures were initiated at the same time each day, limiting variability due to diurnal traffic patterns.
Outcomes: Analyses of the initial cardiopulmonary function analyses conducted by Dr. Godleski and his team are presented in detail in the Harvard CLARC annual progress report. In the remainder of Year 5, we at MSU will complete histopathology and biochemical assays. We will compare filtered air vs. TRUAP exposures in rats on a high fructose diet, which produces hypertension and other facets of the metabolic syndrome (e.g., insulin resistance, hyperglycemia, dyslipidemia). From the telemetered animals, Dr. Godleski’s team will complete the following outcomes: cardiovascular parameters, including blood pressure, heart rate, heart rate variability, and facets of the ECG that are all derived from the telemetry system; respiratory parameters including respiratory rate, times of inspiration, expiration, inspiratory pause, expiratory pause and relaxation, peak air flows during inspiration and expiration, average air flow during expiration, tidal and minute volumes, inspiratory duty cycle, and minute ventilation have been completed. In another group of non-telemetered animals, bronchoalveolar lavage fluid (BALF) was collected after 4 days of exposure and analyses were completed for total inflammatory cells and individual cell types (e.g., neutrophils) as well as total protein and ß-N-acetyl-glucuronidase. From the rats in which BALF was collected and analyzed, heart blood also was collected and analyzed for complete blood counts and differentials; blood chemistries (chem 17), including blood glucose, electrolytes, triglycerides, and hepatic function also were assessed.
Future Activities:
Our main goal in the remainder of Year 5 and in the no-cost-extension year (2016) of this project is to complete the ongoing studies and prepare manuscripts for peer-review publication. Important analyses that need to be completed are listed below along with a few small, short-term laboratory studies that need to be conducted to complete the body of work necessary for manuscripts to be of publication quality.
Ongoing Analyses:
- Cardiotelemetry data analysis: We will assess exposure-related arrhythmias and ECG abnormalities in rats fed a normal or high fructose diet and exposed to O3, PM2.5, and O3/PM2.5 (co-exposure) in both the industrial/urban and transported/rural inhalation exposure studies, conducted in Dearborn and Dexter, MI, respectively. These data will form the basis of one or two manuscripts.
- Associations of exposure/health outcomes: Working in close collaboration with colleagues in the Exposure Core and the Biostatistics/Data Management Core, we will continue the correlation analyses to determine associations between acute cardiovascular responses (e.g., heart rate, mean arterial pressure, heart rate variability) and PM2.5 metrics (e.g., trace elements, gases, sources). We will conduct these correlative analyses to determine modifications by ozone co-exposure on the PM2.5-induced cardiovascular health effects. These data will form the basis for at least one manuscript.
- Morphometric, biochemical, and molecular analyses: We will complete these analyses in the lungs of ozone-exposed mice to explore and elucidate the role innate lymphoid cells in the development of type 2 immunity and associated features of allergic host response (mucous cell metaplasia, eosinophilic inflammation). Based on preliminary results, we anticipate another manuscript that will demonstrate that innate lymphoid cells mediate ozone-induced type 2 immunity and other features of allergic airway disease (mucous cells metaplasia, eosinophilic inflammation).
Anticipated Accomplishments in the Remainder of 2015 and Early in 2016: We will finish the laboratory assays on all the rats, complete the statistical analyses, and prepare a joint report and manuscript for submission to EPA and to an appropriate scientific journal for publication, respectively.
Journal Articles on this Report : 18 Displayed | Download in RIS Format
| Other subproject views: | All 73 publications | 28 publications in selected types | All 28 journal articles |
|---|---|---|---|
| Other center views: | All 148 publications | 72 publications in selected types | All 72 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Balasubramanian P, Sirivelu MP, Weiss KA, Wagner JG, Harkema JR, Morishita M, Mohankumar PS, Mohankumar SM. Differential effects of inhalation exposure to PM 2.5 on hypothalamic monoamines and corticotrophin releasing hormone in lean and obese rats. NeuroToxicology 2013;36:106-111. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2012) R834797C002 (2013) R834797C002 (2014) R834797C002 (2015) R834797C002 (Final) |
Exit Exit Exit |
|
|
Brandenberger C, Li N, Jackson-Humbles DN, Rockwell CE, Wagner JG, Harkema JR. Enhanced allergic airway disease in old mice is associated with a Th17 response. Clinical & Experimental Allergy 2014;44(10):1282-1292. |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) |
Exit Exit |
|
|
Brook RD, Bard RL, Kaplan MJ, Yalavarthi S, Morishita M, Dvonch JT, Wang L, Yang H-Y, Spino C, Mukherjee B, Oral EA, Sun Q, Brook JR, Harkema J, Rajagopalan S. The effect of acute exposure to coarse particulate matter air pollution in a rural location on circulating endothelial progenitor cells: results from a randomized controlled study. Inhalation Toxicology 2013;25(10):587-592. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C001 (2013) R834797C001 (2014) R834797C001 (2015) R834797C001 (2016) R834797C001 (Final) R834797C002 (2013) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2013) R834797C003 (Final) R833740 (2012) R833740 (Final) |
Exit |
|
|
Brook RD, Xu X, Bard RL, Dvonch JT, Morishita M, Kaciroti N, Sun Q, Harkema J, Rajagopalan S. Reduced metabolic insulin sensitivity following sub-acute exposures to low levels of ambient fine particulate matter air pollution. The Science of the Total Environment 2013;448:66-71. |
R834797 (2012) R834797 (2013) R834797 (2014) R834797 (2015) R834797 (Final) R834797C001 (2012) R834797C001 (2013) R834797C001 (2014) R834797C001 (2015) R834797C001 (2016) R834797C001 (Final) R834797C002 (2012) R834797C002 (2013) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (Final) |
Exit Exit Exit |
|
|
Brook RD, Bard RL, Morishita M, Dvonch JT, Wang L, Yang HY, Spino C, Mukherjee B, Kaplan MJ, Yalavarthi S, Oral EA, Ajluni N, Sun Q, Brook JR, Harkema J, Rajagopalan S. Hemodynamic, autonomic, and vascular effects of exposure to coarse particulate matter air pollution from a rural location. Environmental Health Perspectives 2014;122(6):624-630. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C001 (2013) R834797C001 (2014) R834797C001 (2015) R834797C001 (2016) R834797C001 (Final) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R833740 (2012) R833740 (Final) |
|
|
|
Liu C, Ying Z, Harkema J, Sun Q, Rajagopalan S. Epidemiological and experimental links between air pollution and type 2 diabetes. Toxicologic Pathology 2013;41(2):361-373. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2012) R834797C002 (2013) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2013) R834797C003 (2014) R834797C003 (2015) R834797C003 (Final) |
Exit Exit Exit |
|
|
Liu C, Xu X, Bai Y, Wang TY, Rao X, Wang A, Sun L, Ying Z, Gushchina L, Maiseyeu A, Morishita M, Sun Q, Harkema JR, Rajagopalan S. Air pollution-mediated susceptibility to inflammation and insulin resistance:influence of CCR2 pathways in mice. Environmental Health Perspectives 2014;122(1):17-26. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2014) R834797C003 (2015) R834797C003 (Final) |
|
|
|
Liu C, Bai Y, Xu X, Sun L, Wang A, Wang TY, Maurya SK, Periasamy M, Morishita M, Harkema J, Ying Z, Sun Q, Rajagopalan S. Exaggerated effects of particulate matter air pollution in genetic type II diabetes mellitus. Particle and Fibre Toxicology 2014;11:27. |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2014) R834797C003 (2015) R834797C003 (Final) |
Exit Exit Exit |
|
|
Maiseyeu A, Yang H-Y, Ramanathan G, Yin F, Bard RL, Morishita M, Dvonch JT, Wang L, Spino C, Mukherjee B, Badgeley MA, Barajas-Espinosa A, Sun Q, Harkema J, Rajagopalan S, Araujo JA, Brook RD. No effect of acute exposure to coarse particulate matter air pollution in a rural location on high-density lipoprotein function. Inhalation Toxicology 2014;26(1):23-29. |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C001 (2014) R834797C001 (2015) R834797C001 (2016) R834797C001 (Final) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R833740 (Final) |
Exit Exit |
|
|
Morishita M, Bard RL, Kaciroti N, Fitzner CA, Dvonch T, Harkema JR, Rajagopalan S, Brook RD. Exploration of the composition and sources of urban fine particulate matter associated with same-day cardiovascular health effects in Dearborn, Michigan. Journal of Exposure Science & Environmental Epidemiology 2015;25(2):145-152. |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C001 (2014) R834797C001 (2015) R834797C001 (2016) R834797C001 (Final) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2015) R834797C003 (Final) |
Exit Exit |
|
|
Morishita M, Bard RL, Wang L, Das R, Dvonch JT, Spino C, Mukherjee B, Sun Q, Harkema JR, Rajagopalan S, Brook RD. The characteristics of coarse particulate matter air pollution associated with alterations in blood pressure and heart rate during controlled exposures. Journal of Exposure Science & Environmental Epidemiology 2015;25(2):153-159. |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C001 (2014) R834797C001 (2015) R834797C001 (2016) R834797C001 (Final) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2015) R834797C003 (Final) R833740 (Final) |
Exit Exit |
|
|
Ong CB, Kumagai K, Brooks PT, Brandenberger C, Lewandowski RP, Jackson-Humbles DN, Nault R, Zacharewski TR, Wagner JG, Harkema JR. Ozone-induced type 2 immunity in nasal airways. Development and lymphoid cell dependence in mice. American Journal of Respiratory Cell and Molecular Biology 2016;54(3):331-340. |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) |
Exit Exit |
|
|
Rao X, Zhong J, Maiseyeu A, Gopalakrishnan B, Villamena FA, Chen LC, Harkema JR, Sun Q, Rajagopalan S. CD36-dependent 7-ketocholesterol accumulation in macrophages mediates progression of atherosclerosis in response to chronic air pollution exposure. Circulation Research 2014;115(9):770-780. |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2015) R834797C002 (Final) R834797C003 (2014) R834797C003 (2015) R834797C003 (Final) |
Exit Exit Exit |
|
|
Sun L, Liu C, Xu X, Ying Z, Maiseyeu A, Wang A, Allen K, Lewandowski RP, Bramble LA, Morishita M, Wagner JG, Dvonch JT, Sun Z, Yan X, Brook RD, Rajagopalan S, Harkema JR, Sun Q, Fan Z. Ambient fine particulate matter and ozone exposures induce inflammation in epicardial and perirenal adipose tissues in rats fed a high fructose diet. Particle and Fibre Toxicology 2013;10:43. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (Final) R834797C001 (2013) R834797C002 (2013) R834797C002 (2014) R834797C002 (2015) R834797C002 (Final) R834797C003 (2013) R834797C003 (2014) R834797C003 (Final) |
Exit Exit Exit |
|
|
Vital M, Harkema JR, Rizzo M, Tiedje J, Brandenberger C. Alterations of the murine gut microbiome with age and allergic airway disease. Journal of Immunology Research 2015;2015:892568 (8 pp.). |
R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) |
Exit Exit Exit |
|
|
Wagner JG, Allen K, Yang HY, Nan B, Morishita M, Mukherjee B, Dvonch JT, Spino C, Fink GD, Rajagopalan S, Sun Q, Brook RD, Harkema JR. Cardiovascular depression in rats exposed to inhaled particulate matter and ozone: effects of diet-induced metabolic syndrome. Environmental Health Perspectives 2014;122(1):27-33. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C001 (2013) R834797C001 (Final) R834797C002 (2013) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2013) R834797C003 (Final) |
Exit |
|
|
Wagner JG, Kamal AS, Morishita M, Dvonch JT, Harkema JR, Rohr AC. PM2.5-induced cardiovascular dysregulation in rats is associated with elemental carbon and temperature-resolved carbon subfractions. Particle and Fibre Toxicology 2014;11:25 (10 pp.). |
R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) |
Exit Exit Exit |
|
|
Ying Z, Xu X, Bai Y, Zhong J, Chen M, Liang Y, Zhao J, Liu D, Morishita M, Sun Q, Spino C, Brook RD, Harkema JR, Rajagopalan S. Long-term exposure to concentrated ambient PM2.5 increases mouse blood pressure through abnormal activation of sympathetic nervous system:a role for hypothalamic inflammation. Environmental Health Perspectives 2014;122(1):79-86. |
R834797 (2013) R834797 (2014) R834797 (2015) R834797 (2016) R834797 (Final) R834797C002 (2013) R834797C002 (2014) R834797C002 (2015) R834797C002 (2016) R834797C002 (Final) R834797C003 (2013) R834797C003 (2014) R834797C003 (2015) R834797C003 (Final) |
|
Supplemental Keywords:
inhalation toxicology, acute multipollutant exposures, high-fructose diet, rats, PM, ozone, cardiometabolic syndrome, Air, Scientific Discipline, ENVIRONMENTAL MANAGEMENT, Health Risk Assessment, Biology, Biochemistry, air toxics, Risk Assessment, aerosol particles, human health, physiology, human exposure, particulate matter, cardiotoxicity, susceptible populations, acute cardiovascualr effects, ambient air quality, cardiopulmonaryProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R834797 Health Effects Institute (2005 — 2010) Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834797C001 Cardiometabolic Effects of Exposure to Differing Mixtures and Concentrations of PM2.5 in Obese and Lean Adults
R834797C002 Cardiometabolic, Autonomic, and Airway Toxicity of Acute Exposures to PM2.5 from Multipollutant Atmospheres in the Great Lakes Region
R834797C003 Long Term Metabolic Consequences of Exposures to Multipollutant Atmospheres in the Great Lakes Region
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2016 Progress Report
- 2014 Progress Report
- 2013 Progress Report
- 2012 Progress Report
- 2011 Progress Report
- Original Abstract
28 journal articles for this subproject
Main Center: R834797
148 publications for this center
72 journal articles for this center