Grantee Research Project Results
2016 Progress Report: Project B: Exposure Project: Mn, DDT/E and PBDE Exposure to Farmworker Children
EPA Grant Number: R834513C002Subproject: this is subproject number 002 , established and managed by the Center Director under grant R834513
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Center for Research on Early Childhood Exposure and Development in Puerto Rico
Center Director: Alshawabkeh, Akram
Title: Project B: Exposure Project: Mn, DDT/E and PBDE Exposure to Farmworker Children
Investigators: Eskenazi, Brenda , Bradman, Asa
Current Investigators: Eskenazi, Brenda , Harley, Kim , Holland, Nina T. , Jerrett, Michael , Sjodin, Andreas , Arora, Manish , Smith, Donald , Eisen, Ellen , Molitor, John , Hubbard, Alan , Lustig, Robert
Institution: University of California - Berkeley
EPA Project Officer: Hahn, Intaek
Project Period: August 1, 2009 through July 31, 2014 (Extended to July 31, 2017)
Project Period Covered by this Report: June 1, 2015 through May 31,2016
RFA: Children's Environmental Health and Disease Prevention Research Centers (with NIEHS) (2009) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
In Project B, we are examining novel methods of examining prenatal exposure to manganese (Mn), PBDE, and DDT/E compounds. For Mn, we are developing methods to measure Mn in shed deciduous teeth and in hair collected when the children were 10.5 years of age. For PBDEs and DDT/E, we are developing models to back-extrapolate prenatal exposure levels from 9-year measured levels and other determinants of exposure.
Progress Summary:
Specific Aim 1. To measure Mn, PBDEs, and DDT/E in shed deciduous teeth. (Population: CHAMACOS children participating since birth.)
Mn: We have successfully developed a method for measuring Mn in human teeth by laser ablation ICP-mass spectrometry (LA/ICP/MS), including analysis of 307 teeth collected from the CHAMACOS cohort. Rather than calculate one estimate of cumulative exposure over the entire prenatal period, we have used a method to distinguish Mn exposure experienced in the 2nd trimester from that experienced in the 3rd trimester. Weobserved a significant association (rspearman=0.36, p=0.001, n=77) between Mn levels in the entire prenatally-formed dentine [as defined by 55Mn:43Ca area under the curve (AUC)] with floor dust Mn loading (µg Mn/m2 floor area) and a stronger relationship with Mn levels in dentine formed only in the 2nd trimester (rspearman=0.40; p=0.0005; n=72), likely because dust samples were collected in the 2nd trimester. We have also compared Mn levels in cord blood and mantle dentine. While there was no significant association of 55Mn:43Ca AUC of all prenatal sampling points with cord blood Mn (rspearman=-0.01; p=0.99), 55Mn:43Ca in the sampling point immediately adjacent to the neonatal line in each tooth showed a significant positive correlation with cord blood Mn (rspearman=0.70; p=0.003; n=16). These findings were published in Environmental Science & Technology (Arora et al. 2012) and the measurements were used in subsequent studies examining exposure and health outcomes (see below and Progress Report A).
PBDEs and DDT/E: As a pilot investigation, we assessed the feasibility of measuring persistent organic pollutants in deciduous teeth. Dr. Andreas Sjödin at the CDC in Atlanta attempted to measure PBDEs and DDT in anonymous deciduous teeth collected from dental patients demographically similar to the CHAMACOS cohort. Using whole tooth digestion, neither PBDEs nor DDT were detected. Additional anonymous deciduous teeth were obtained from a local clinic and Dr. Arora extracted the dentine from these teeth to determine if more sensitive analyses could be employed to measure PBDEs and organochlorine (OC) compounds including DDT. Initial results were not promising. Specifically, low detection frequencies for the typically prevalent congeners PBDE-47 and -99 and high concentrations of PBDE-183 for all samples suggested possible contamination with PBDE-183. One challenge in the interpretation of xenobiotic measurements in dentine is the likelihood that dentine tissue, which is vasculated, may be in equilibrium with child exposures when the teeth are shed and may not reflect prenatal exposures. Some compounds, such as metals, are incorporated into the chemical structure of the dentine, but many organic compounds are not. Additional research is needed to broadly screen for chemicals in dentine strata and validate measurements against maternal and child biomonitoring.
Specific Aim 2. To determine the relationship between Mn, PBDE, and DDT/E levels in teeth with other relevant biological measures.
For a subset of CHAMACOS participants, we conducted laboratory measurements of Mn in multiple biological samples, including 202 whole blood samples (maternal blood, cord blood, child’s blood), 128 urine samples (maternal at 26-week gestation and child at 24 months), and 62 teeth and examined the interrelationships of Mn levels in all matrices.
Mn levels (Mn:Ca ratio) were higher in prenatal than postnatal dentine (geometric mean (GM) = 0.51 vs. 0.16, p<0.0001). Maternal blood Mn concentrations increased from 26 weeks gestation to delivery (GM = 14.6 to 20.7 µg/L, p = 0.001) and child blood Mn concentrations decreased from cord blood to 24-month bloodsamples (39.9 vs. 25.0 µg/L, p = 0.005). Mn levels in tooth dentine during the 3rd trimester were positivelycorrelated with Mn concentrations in cord blood (rs = 0.31), while there was a negative correlation between prenatal Mn levels in enamel and concentrations in maternal blood at 26-weeks’ gestation (rs = -0.36). We did not find any significant differences in Mn urine concentrations over time and did not observe significant correlations between Mn levels in teeth and urine, suggesting that urinary concentrations are not a useful measure of environmental Mn exposure.
We observed significantly higher (p < 0.05) levels of Mn in prenatal dentine, prenatal maternal blood, and 24-month urine from children if there was a farm worker living in the home during the corresponding time period compared to no farmworker living in the home. Prenatal Mn levels in dentine and cord blood were also correlated with Mn loading in prenatal house dust samples (rs = 0.27 and 0.29, respectively; p < 0.1). Tooth dentine and blood Mn concentrations had the strongest associations with potential sources of Mn exposure in the home. These findings were published in Environmental Science & Technology (Gunier et al. 2014).
Specific Aim 3. To quantify the contribution of agricultural pesticide use to Mn levels in house dust, teeth, hair and child blood.
Mn in dust: We collected house dust samples from 378 CHAMACOS participant residences, with a second sample collected approximately nine months later from 90 of the residences. House dust samples were analyzed for Mn using inductively coupled plasma optical emission spectroscopy. Information from interviews, home inspections, and pesticide use 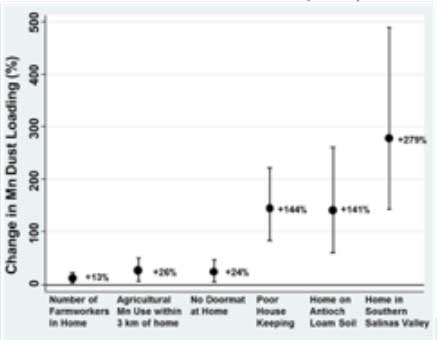
FIgure 1. Percentage change in manganese dust loading (µg/m2) for select
predictor variables estimated from multivariable mixed effect models (n=64).
| Table 1. Percent change of Mn in prenatala dentine and partial coefficient of determination (r2) for predictors in multivariable models for tooth Mn (n=206) and subset with tooth and dust Mn (n=130). | ||||||
|
| Children with Tooth Mn Levels | Children with Tooth and Dust Mn | ||||
|
Predictor Variable | % Changeb (95% CI) |
p-value | Partial r2 (%) | % Changeb (95% CI) | p- value | Partial r2 (%) |
| Maternal farmwork | 10.1 (0.1, 21) | 0.05 | 1.8 | 15.8 (1.9, 31.6) | 0.03 | 3.9 |
| (Prenatal yes vs. no) |
|
|
|
|
|
|
| Farm worker shoes in home | 8.1 (4.3, 12) | <0.001 | 8.4 | 6.4 (1.5, 11.4) | 0.01 | 5.2 |
| (Prenatal per worker) |
|
|
|
|
|
|
| Agricultural Fungicide Use Prenatal within 3 km |
4.9 (0.9, 9.1) |
0.02 |
2.8 |
3.4 (-1.6, 8.6) |
0.19 |
1.4 |
| (per IQRc = 809 kg ) |
|
|
|
|
|
|
| Soil Type | 15 (4.6, 27) | 0.004 | 4.0 | 21 (6.3, 37) | 0.004 | 6.4 |
| (Antioch Loam vs. other) |
|
|
|
|
|
|
| Mother smoked | -34 (-47, -18) | <0.001 | 6.7 | -40 (-55, -20) | 0.001 | 9.0 |
| (Prenatal yes vs. no) |
|
|
|
|
|
|
| Mn dust loading | - |
| - | 3.3 (0.3, 6.4) | 0.03 | 3.6 |
| (per IQRc = 1465 µg/m2) |
|
|
|
|
|
|
| R2 for Model |
|
| 22% |
|
| 29% |
| aPrenatal = 2nd and 3rd trimesters. bPercent change = (exp(β)-1)*100; cIQR = interquartile range. | ||||||
Mn in teeth: We examined environmental and lifestyle factors associated with prenatal tooth dentine Mn concentrations for 207 CHAMACOS children. We found that storage of farmworkers’ shoes in the home, maternal farm work, agricultural use of Mn-containing fungicides within 3 km of the residence, residence built on Antioch Loam soil and Mn dust loading (µg/m2 of floor area) during pregnancy were associated with higher Mn levels in prenatal dentine (p<0.05). Maternal smoking during pregnancy was inversely related to Mn levels in prenatal dentine (p<0.01).
Multivariable regression models explained 22 – 29% of the variability of Mn in prenatal dentine (Table 1). Our results suggest that Mn measured in prenatal dentine provides retrospective and time-specific levels of fetal exposure resulting from environmental and occupational sources. These findings were published in Environmental Science & Technology (Gunier et al. 2013).
Mn in hair: In the absence of a validated methodology for the accurate determination of hair Mn without external contamination, we developed and validated a method to analyze Mn in hair which included thorough sample cleanup, showing in the process that prior published studies of hair Mn are likely erroneous (Eastman et al 2013). We have collected hair samples from 455 children (145 boys and 310 girls). Due to costs associated with the extensive cleanup procedure necessary to analyze the hair properly, we were only able to analyze hair from a subsample of children. Dr. Smith at UCSC has analyzed samples from 135 participants. The Mn hair concentrations in our cohort (median=0.08 µg/g) were similar (median=0.07 µg/g) to the only other published study using the same cleaning methodology, which focused on children exposed to Mn through proximity to a ferroalloy plant (Eastman et al. 2013). In bivariate analyses, we found that Mn hair concentrations were related (p<0.2) to location of the hair sample (lower in samples from the top of the head), soil type at the child’s residence (higher in Antioch Loam), location of the residence in the Southern portion of Monterey County (higher in the South) and agricultural use of Mn fungicides within 3 km of the child’s residence during the 6 months prior to sample collection. Hair Mn concentrations were not related to demographic variables, the presence of agricultural workers in the home or the use of hair care products prior to sample collection. Location of hair sample (p=0.04), residence in Southern Monterey County (p=0.13) and agricultural use of Mn fungicides near the child’s residence (p=0.04) remained significant in multivariate models (Figure 2) and this model explained 12% of the variability in Mn hair concentrations. We did not observe an association between hair Mn concentrations and
-
- year IQ or behavior in models adjusted for child’s exact age, maternal education, poverty status, language of assessment and HOME score. We are currently preparing a manuscript for publication on our Mn hair analyses.
Specific Aim 4. To identify population correlates of PBDEs and DDT/E exposure in 9-year- old boys.
CDC has analyzed CHAMACOS blood samples for PBDEs, DDT/E and PCBs in 412 mothers during pregnancy, 277 children at the 7-year visit, and 145 mothers and 552 children at the 9-year visit. The geometric means for these compounds decreased significantly (between 8 and 13 percent per year) in the children from age seven to nine years (Table 2). We also showed that BDE-153, the PCBs and DDE all decreased significantly per kilogram weight gain in the child related to dilution from an increasing body mass with age. Dr. Andreas Sjödin prepared a manuscript (later published in 2018) examining determinants of PBDE and DDT/E exposures in 9-year-olds. The statistical analysis for this paper is complete and includes comparison of the CHAMACOS serum concentration data with a published cohort of Texas children (Sjodin et al. 2014). CHAMACOS participants had significantly higher exposures to PBDEs than children from Texas. Concentrations of PCB-153 were comparable in the CHAMACOS and Texas children while p,p’-DDE concentrations were higher in the primarily Mexican population of the CHAMCOS cohort.
Table 2. Percentage change between age seven and nine by year and kilogram of weight gain for selected PBDEs, PCBs, DDT and DDE for participants with repeated measurements only.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Analyte N
Change per Year Change per kilogram weight gain
Polychlorinated biphenyls (PCBs)
| PCB-74 | 210 | -3.7 | (-6.7,-0.7) | 0.02 | -0.5 | (-0.9,-0.1) | 0.02 |
| PCB-99 | 224 | -3.2 | (-6.1,-0.1) | 0.04 | -0.5 | (-0.9,-0.1) | 0.01 |
| PCB-118 | 209 | -2.1 | (-5.3,1.2) | 0.21 | -0.7 | (-1.1,-0.3) | <0.001 |
| PCB-138/158 | 224 | -3 | (-6.0,0.0) | 0.05 | -0.6 | (-0.9,-0.2) | <0.001 |
| PCB-153 | 224 | -2.9 | (-5.9,0.2) | 0.07 | -0.6 | (-1.0,-0.2) | <0.001 |
| PCB-180 | 224 | -3.2 | (-6.1,-0.1) | 0.04 | -0.6 | (-0.9,-0.2) | 0.01 |
Persistent Pesticides
DDE 228 -1.7 (-6.7,3.6) 0.53 -2.4 (-3.4,-1.4) <0.001
Abbreviations: BDE-28, 2,4,4'-tribromodiphenyl ether; BDE-47, 2,2',4,4'-tetraBDE; BDE-85, 2,2',3,4,4'-pentaBDE; BDE-99, 2,2',4,4',5-pentaBDE; BDE-100, 2,2',4,4',6-
pentaBDE; 2,2',4,4',5,5'-hexaBDE; BDE-154, 2,2',4,4',5,6'-hexaBDE; CB-74, 2,4,4',5-
tetrachlorobiphenyl; CB-99, 2,2',4,4',5-pentaCB; CB-118, 2,3',4,4',5-pentaCB; CB- 138/158, 2,2',3,4,4',5'-hexaCB and 2,3,3',4,4',6-hexaCB; CB-153, 2,2',4,4',5,5'-hexaCB;
CB-180, 2,2',3,4,4',5,5'-heptaCB; and p,p'-DDE, 2,2-Bis(4-chlorophenyl)-1,1- dichloroethene.
Back-extrapolation of DDT/E and PBDEs: We evaluated three modeling approaches (physiologically-based pharmacokinetic modeling (PBPK), deletion substitution algorithms, and SuperLearner) to estimate maternal DDT/E and PBDE exposures during pregnancy when measurements were collected from the mothers nine years after birth. To accomplish this, we used the models to predict serum levels measured at 26 weeks gestation in the pregnant mothers (n=161) from levels measured in the mothers and the child when the child was 9. For each model, we assessed whether the nine-year maternal (n=94) or child (n=161) measurements alone, or levels in both mothers and children (n=89) at age nine, predicted the levels in the mothers during pregnancy. Model performance was assessed using the root mean squared error (RMSE) and coefficient of determination (R2) of log10-transformed back- extrapolated versus measured levels. For all compounds and subsets, SuperLearner outperformed the other approaches with RMSEs and R2s ranging from 0.10-0.31 log10 ng/g and 0.58-0.97, respectively. Typically, model RMSEs were lower and R2s were higher for p,p’-DDT/E than PBDE congeners, and estimations using maternal levels (when their child was 9 years) were more accurate for back extrapolation to pregnancy levels than using child levels at 9 years. The PBPK model performed well but not as well as SuperLearner when back- extrapolating prenatal levels from maternal levels for compounds with longer half-lives such as
p,p’-DDE and BDE-153 (RMSEs= 0.21 and 0.28 log10 ng/g and R2s= 0.88 and 0.57, respectively). Overall, our results demonstrate the ability to accurately back-extrapolate prenatal levels from maternal levels 9 years after delivery, with SuperLearner performing the best based on our fit criteria. These findings were published in Environmental Science & Technology (Verner et al. 2015).
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other subproject views: | All 108 publications | 43 publications in selected types | All 42 journal articles |
|---|---|---|---|
| Other center views: | All 697 publications | 170 publications in selected types | All 169 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Bradman A, Quiros-Alcala L, Castorina R, Schall RA, Camacho J, Holland NT, Barr DB, Eskenazi B. Effect of organic diet intervention on pesticide exposures in young children living in low-income urban and agricultural communities. Environmental Health Perspectives 2015;123(10):1086-1093. |
R834513 (2014) R834513 (2016) R834513C002 (2014) R834513C002 (2015) R834513C002 (2016) R834513C003 (Final) |
|
|
|
Dave V, Street K, Francis S, Bradman A, Riley L, Eskenazi B, Holland N. Bacterial microbiome of breast milk and child saliva from low-income Mexican-American women and children. Pediatric Research 2016;79(6):846-854. |
R834513 (Final) R834513C002 (2016) R834513C003 (2015) |
Exit Exit Exit |
|
|
Salvatore AL, Castorina R, Camacho J, Morga N, Lopez J, Nishioka M, Barr DB, Eskenazi B, Bradman A. Home-based community health worker intervention to reduce pesticide exposures to farmworkers’ children: a randomized-controlled trial. Journal of Exposure Science & Environmental Epidemiology 2015;25(6):608-615. |
R834513 (2014) R834513 (2015) R834513 (2016) R834513C002 (2014) R834513C002 (2015) R834513C002 (2016) R834513C004 (2015) |
Exit |
Supplemental Keywords:
Health, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Health Risk Assessment, Environmental Policy, Biology, Children's Health, biological markers, harmful environmental agents, pesticide exposure, agricultural community, flame retardants, neurochemical effects, PBDE, children's vulnerablity, farmworkersProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R834513 Center for Research on Early Childhood Exposure and Development in Puerto Rico Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834513C001 CHAMACOS Cohort Project: Pesticides and PBDE on Neurobehavior and Puberty
R834513C002 Project B: Exposure Project: Mn, DDT/E and PBDE Exposure to Farmworker Children
R834513C003 Epigenetics Project
R834513C004 Community Outreach and Translation Core
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2015 Progress Report
- 2014 Progress Report
- 2013 Progress Report
- 2012 Progress Report
- 2011 Progress Report
- 2010 Progress Report
- Original Abstract
42 journal articles for this subproject
Main Center: R834513
697 publications for this center
169 journal articles for this center
