Grantee Research Project Results
Final Report: The Columbia Center for Children’s Environmental Health
EPA Grant Number: R834509Center: The Columbia Center for Children’s Environmental Health
Center Director: Perera, Frederica P.
Title: The Columbia Center for Children’s Environmental Health
Investigators: Perera, Frederica P. , Whyatt, Robin M. , Miller, Rachel L. , Evans, David , Rauh, Virginia , Andrews, Howard F. , Champagne, Frances , Rundle, Andrew , Shepard, Peggy
Institution: Columbia University in the City of New York , Resources for the Future , West Harlem Environmental Action (WE ACT for Environmental Justice) , Columbia University Mailman School of Public Health
EPA Project Officer: Callan, Richard
Project Period: September 24, 2009 through September 23, 2014 (Extended to September 23, 2015)
Project Amount: $3,953,320
RFA: Children's Environmental Health and Disease Prevention Research Centers (with NIEHS) (2009) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
Obesity Project:
1) To test whether prenatal and early-life exposures to the endocrine disrupters polycyclic aromatic hydrocarbon (PAH) and bisphenol A (BPA) predict body size growth trajectories and childhood obesity at age 8-10 years. This will be accomplished by following our ongoing birth cohort to age 8-10 years, measuring height and weight at ages 5, 7, 8-10, body composition at age 7, 8-10, and metabolic syndrome components at age 8-10 years. This work takes advantage of sophisticated geographic information system based data on the children's neighborhoods to control for social (e.g., poverty and socio-demographic composition) and physical factors (e.g., playgrounds, parks, fast-food vendors) likely to predict childhood obesity.
2) To determine whether differences in the methylation status of key genes involved in adipogenesis (PPARv2, C/EBPq, C/EBPli, C/EBP6 and DLK1), appetite control (FTO) mediate the association between xenobiotic exposures and childhood obesity outcomes. Methylation of these genes will be measured in cord white blood cell DNA by pyrosequencing.
Neurodevelopmental Project:
1) To determine whether prenatal exposures to the endocrine disrupters, PAH and BPA, are associated with adverse neurobehavioral outcomes in peri-pubertal children, as measured by diagnostic assessment of child psychopathology and cognitive functioning.
2) To determine whether prenatal exposure to PAH or BPA is associated with epigenetic changes in umbilical cord white blood cells (DNA methylation validated by gene expression) in candidate genes/pathways is associated with endocrine disruption and immune dysregulation, and whether altered methylation and gene expression in these candidates is associated with the neurobehavioral outcomes.
3) To determine the extent to which neighborhood-level conditions contribute to neurobehavioral outcomes and/or moderate the individual-level associations between exposure to PAH or BPA and child neurodevelopment using GIS.
Mechanistic Core:
1) To examine the consequence of prenatal oral BPA exposure on neurobehavioral, obesity and immune dysfunction in Balb/c mice by determining whether prenatal BPA exposure is associated with abnormal brain cytoarchitecture; impaired social, anxiety-like and cognitive performance; greater adult body weight; body fat composition; and organ fat and immune dysfunction in the adult offspring or grand offspring.
2) To examine the consequence of prenatal oral BPA exposure for tissue-specific molecular modifications in mice by determining prenatal BPA exposure-induced changes in DNA methylation in genes sensitive to endocrine disruption and immune dysregulation in the brain (hippocampus, hypothalamus, cortex), adipocytes and blood of the prenatally BPA exposed offspring and grand-offspring at gestation day 19 and adulthood (PND 60). Determine corresponding changes in gene expression, neurobehavioral, obesity and immunologic outcomes, and whether changes in DNA methylation and gene expression of genes critical for adipogenesis persist in blood of F1 male offspring from PND 28 to adulthood.
3) To examine the consequence of prenatal inhaled PAH exposure at current levels determined to exist in New York City's Northern Manhattan/South Bronx on neurodevelopment and obesity in Balb/c mice by determining whether prenatal PAH exposure is associated with abnormal brain cytoarchitecture, impaired anxiety-like and cognitive performance, greater body weight through weaning to adulthood, body fat composition, and organ fat content in adult offspring and grand offspring.
4) To examine the consequence of prenatal inhaled PAH exposure for tissue specific molecular modifications (DNA methylation) in mice of genes sensitive to endocrine disruption and immune dysregulation in the brain, adipocytes and blood of the prenatally exposed offspring and grand offspring at gestation day 19 and PND 60. To determine corresponding changes in gene expression, neurobehavioral, obesity and immunologic outcomes, and whether changes in DNA methylation and gene expression of genes critical for adipogenesis persist in blood of F1 male offspring from PND 28 to adulthood.
Data Management and Statistics Core:
1) To provide the centralized resource for all data management needs of the CCCEH projects, including database design, implementation and modification; data entry; and data retrieval for statistical analysis.
2) To implement procedures to maintain data quality, data security and confidentiality
3) To coordinate and oversee design and implementation of all statistical analyses for testing primary and secondary hypotheses, and provide statistical review of all publications for the research projects
4) To integrate geographic information systems, including acquisition and updating of community-level data, geocoding, mapping, and application of multi-level modeling, as required by the cohort-based Neurodevelopmental and Obesity projects.
Community Outreach and Research Translation Core:
1) To engage and expand the Community Advisory and Stakeholder Board.
2) To communicate the Center’s research findings through the development of educational materials designed for local residents, community organizations, healthcare providers, and other local stakeholders.
3) To facilitate education of public health and clinical professionals working in low-income communities of color in collaboration with the Center’s Pediatric Health Specialist.
4) To disseminate the Center’s findings using several communication methods.
5) Through our partnership with WE ACT, expand the capacity of low-income communities of color to advocate for improved environmental conditions by using the Center’s scientific findings and related findings by other investigators.
Summary/Accomplishments (Outputs/Outcomes):
Obesity Project:
Aim 1: PAH exposure
We have completed study subject enrollment with 400 cohort children having been followed-up through ages 8.5 to 12 years of age. Children provided fasting blood samples and took part in a clinical examination to assess blood lipids, blood glucose, insulin levels, blood pressure, pubertal development, and height, weight and waist circumference.
Blood samples were immediately sent to the Columbia Biomarkers Core Facility for measurements of blood cholesterol and glucose. Appointments for the children to visit the CCCEH clinical office were scheduled for 2-4 weeks after the blood collection. At the clinical visit, the child's height, weight, waist circumference, body composition, and blood pressure were measured. The child underwent a Tanner staging test, and completed a Fels physical activity questionnaire and the modified children's Block FFQ. In addition, a pediatrician provides feedback to the parent on the results of the blood tests, anthropometric measures, and the Tanner staging. Appropriate medical referrals are made for children whose clinical results are outside of norms.
We collaborated with NutritionQuest, the provider of the Block Food Frequency Questionnaire (FFQ), to modify their standard Spanish FFQ for children to include food items from the Dominican diet. Our FFQ is implemented via an online interface.
Because the modified Block FFQ is a new tool, initial statistical analyses of dietary patterns were completed on the first 78 collected data sets using the principle component analyses methods of Hu and colleagues. Two prominent principle components were identified, one loading high on fruits, low fat milk, whole grains, juices, vegetables, and legumes explained 22% of the variance in the data and the second loading high on sandwiches/hamburgers, cold breakfast cereal, processed meats, refined grains and desserts and explained 10% of the variance in the data. These results are consistent with the literature on dietary pattern analyses and the modified Block FFQ thus appears to be collecting valid data.
Analyses of mothers’ exposures to ambient air polycyclic aromatic hydrocarbons (PAHs) and obesity outcomes of their children at ages 5 and 7 were completed. There was a significant (p<0.05) association between high prenatal exposure to PAH and higher BMI Z-score and obesity risk at age 5 and with BMI Z-score, obesity risk and fat mass at age 7 (see Table 1). Differences in lean mass (e.g., body mass from organs, bone, muscle tissue) are unrelated to prenatal PAH exposures (Rundle, et al., 2012).
Table 1: Associations between maternal exposure to ambient air PAH during pregnancy and the child’s obesity related outcomes.
| BMI Z-score age 5 Beta1, p-value | BMI Z-score age 7 Beta1, p-value | % body fat Beta1, p-value | Fat mass (Kg) Beta1, p-value | Fat Free Mass (Kg) Beta1, p-value | |
|---|---|---|---|---|---|
| PAH exposure per unit log transformed exposure | 0.20, 0.03 | 0.18, 0.03 | 1.04, 0.03 | 0.60, 0.04 | 0.19, 0.49 |
1Adjusting for birth weight, ethnicity, mother’s receipt of public assistance during pregnancy, sex of the child, and maternal pre-pregnancy obesity.
As part of our work studying the effects of prenatal PAH exposures and childhood obesity, we developed and implemented inverse probability weighting (IPW) and marginal structural models (MSM) based methods to assess and adjust for potential biases occurring due to loss to follow-up in the cohort. These methods are being deployed across our studies of obesity, asthma and neurodevelopmental outcomes in the Center cohort. The methods were published as an online supplement to our American Journal of Epidemiology paper (Rundle, et al., 2012).
Following up on this 2012 publication showing that prenatal exposures to PAH were associated with higher BMI Z-scores at ages 5 and 7 and with higher fat mass at age 7, we have completed repeated measures analyses of BMI-score at age 5, 7, 9 and 11. Mixed linear growth model analyses show that there is a significant (p< 0.05) effect on the BMI by age intercept at age 5 (0.44 BMI units at age 5 per unit increase in Ln PAH exposure, 95% CI 0.13, 0.75) and no effect on the BMI by age slope. These results indicate that prenatal PAH exposure is associated with higher BMI at age 5, and this higher BMI status continues forward through time without acceleration or flattening of the trajectory. We also have assessed fat mass index (FMI – fat mass in Kg/height in meters2) measured at the 7, 9 and 11 year follow-up waves as a more direct measure of adiposity. A linear mixed model of FMI-by-age failed to converge, perhaps because only three waves of data were available. However, using generalized estimating equation analyses with repeated measures of FMI, we found that higher prenatal PAH exposures were associated with higher FMI (0.30 unit increase in FMI per unit Ln PAH, p=0.03) and the effects of prenatal PAH exposure on FMI did not vary by wave of follow-up. Similar to the BMI-by-age trajectory results, these analyses suggest that prenatal PAH is associated with FMI attained by age 7, and this higher FMI continues forward through time without a significant acceleration or flattening of the trajectory.
We also have applied latent class growth modeling (LCGM) analyses to the BMI data using the SAS PROJ TRAJ macro. LCGM revealed three major classes of growth classes: Class 1 closely reflects the growth curve of a child at the 50th percentile on the CDC growth charts at age 5 and staying at that percentile through age 11 (55% of children); Class 2 reflects children who were overweight at age 5 and whose growth curve accelerated into the obese category by age 12 (“Overweight-to-obese” trajectory class - 35% of children); and Class 3 reflects children who already were obese at age 5 and whose growth accelerates upwards through age 12 (“Obese-to-highly obese” trajectory class - 10% of children). After adjusting for child sex and potential confounders, children exposed to higher prenatal PAH concentrations were significantly more likely to have growth curves classified as Class 3 as compared to Class 1 (OR = 1.76 per unit Ln PAH, 95% CI 1.07, 2.88).
In terms of dissemination work on the PAH findings, we sent out a series of coordinated press releases when the 2012 AJE paper was published, have made presentations regarding the findings at community forums, and have presented the work in several academic forums. Dr. Rundle participated in the NIEHS Virtual Forum: Childhood Obesity and the Environment. The more recent results looking at growth trajectories have been presented as part of the February 2015 NIEHS/EPA Children’s Centers Webinar series.
Aim 1: BPA exposure
We have completed cross-sectional analysis of predictors and correlates of bisphenol A (BPA) levels in urine samples collected from the mothers during pregnancy and from the children at ages 3, 5 and 7. BPA levels in maternal and child age 5 and 7 urine samples are significantly higher for African Americans as compared to Dominicans. BPA levels were highest in the urine samples collected during summer months from the children at ages 3, 5 and 7. BPA levels in the prenatal, age 3 and 5 urine samples are correlated with eight phthalate metabolites measured in the urine samples (Hoepner, et al., 2013). Analyses of maternal urinary BPA levels and birthweight are complete and no associations were observed. Prenatal urinary concentrations of BPA were not associated with child BMI Z scores at age 5 and 7, but were associated with higher waist circumference and FMI at age 7. This work has been submitted to Environmental Health Perspectives and the peer reviewers have recommended that the manuscript be accepted for publication. We are waiting for final confirmation of its acceptance from the editorial office.
Since the launch of our study, there has been growing concern in the academic and lay literature about exposures to phthalates and childhood obesity. We have shown that urinary concentrations of phthalates are correlated with urinary concentrations of BPA and thus, phthalates have the potential to confound the associations of BPA with the outcomes of concern. Under separate funding, maternal and child urine samples have been analyzed for nine phthalate metabolites and Dr. Rundle oversaw a T32 Medical Fellow in Pediatric Endocrinology, Dr. Michelle Maresca, in analyzing the phthalate metabolite data and child body size. This work is aligned with our hypotheses that exposures to endocrine disrupting chemicals affect childhood body size, we include the results here in our progress report.
We have completed analyses of urinary phthalate concentrations in maternal and child (age 3 and 5) urine samples and child anthropometric outcomes at age 5 and 7. Child anthropometric outcomes are not associated with child urinary metabolite concentrations. However, higher concentrations of non-DEHP metabolites in maternal urine are associated with lower BMI Z scores at ages 5 and 7 and with lower FMI and waist circumference at age 7 among boys only. This work was e-published ahead of print in June 2015 in Environmental Health Perspectives, we expect it to appear in print soon. We also have collaborated with several of the other Children’s Centers to pool our urinary phthalate and child anthropometric outcome data. A paper based on the pooled analyses has been accepted for publication in the journal Epidemiology.
Airborne PAH concentrations as measured by the personal air-monitors carried by the mothers were not correlated with concentrations of BPA and phthalate metabolites in urine samples collected from the mothers nor with concentrations of these chemicals in urine samples collected from the children. The effects of prenatal PAH exposures on childhood growth are not confounded by exposures to these chemicals.
Aim 2
We completed the assessment of methylation status using pyrosequencing for the PPAR-Gamma and DLK1 genes in cord-blood DNA. Percent methylation scores across 16 CpG sites in PPAR Gamma were associated with child sex, but were not associated with prenatal exposure to PAH measured in air, or with BPA or phthalate metabolite concentrations in maternal urine samples. PPAR Gamma methylation status also was not associated with child anthropometric outcomes. Similarly, CpG methylation in DLK1 was not associated with exposures or outcomes.
Additional Work
Linking with the P01 Neurodevelopment Project (PI Rauh) we have assessed whether trajectories of development of emotional self-regulation as measured by the Child Behavior Checklist (CBCL) are associated with child obesity risk. The CBCL was administered to the cohort children at ages 3 to 5, 7, 9 and 11 years. The index of deficient self-regulation (DESR) was calculated by combining t-scores on three scales from CBCL: intense emotions (Anxiety/ Depression Scale), aggression (Aggression Scale), and impulsive behavior (Attention Scale). LCGM then were applied to repeated measures of Self-Regulation. Analyses identified two distinct and stable classes of Self-Regulation trajectories, a latent class reflecting “High and stable self-regulation throughout childhood” (82% of children) and a latent class that reflected “Poor self-regulation throughout childhood” (18% of children). Children in the “Poor self-regulation throughout childhood” trajectory class were significantly more likely also to be members of the “Overweight-to-obese” and the “Obese-to-highly obese” trajectory classes identified in the childhood anthropometric data (p=0.03). These analyses show that unhealthy weight gain trajectories are significantly associated with poorer developmental trajectories for emotional self-regulation. In addition, we assessed the link between DESR trajectory classes and child physical activity between ages 8.5 and 12 years as measured in the obesity project using the Fels physical activity questionnaire. Children who belonged to the “Poor self-regulation throughout childhood” latent class had significantly lower activity scores for the Fels Total Activity Scale (0.66 scale score points lower, p<0.01) as compared to participants who belonged to the “High and stable self-regulation throughout childhood” latent class. The deficit in physical activity was largely due to lower activity levels in leisure time; 0.48 scale score points lower on the Fels Leisure Time Scale (p<0.001).
We have completed analyses of child anthropometric outcomes and maternal use of antibiotics during pregnancy and mode of delivery and find that C-section birth and antibiotic use are associated with higher BMI Z scores at age 7 (Mueller, et al., 2014). We also have completed analyses showing that excessive weight gain during pregnancy is associated with higher BMI Z scores and fat mass in the child at ages 5 and 7 (Widen, et al., 2015a). We recently published a paper in the American Journal of Clinical Nutrition showing that excessive gestational weight gain among women who were normal weight or mildly overweight before pregnancy is associated with higher weight and % body fat 7 years postpartum (Widen, et al., 2015b).
Neurodevelopmental Project:
Aim 1: PAH exposure
During this grant period we have continued our analysis on the impact of prenatal PAH exposure on the neurodevelopment of our cohort as they grow and develop. At age 5 years, using the Wechsler Preschool and Primary Scale of Intelligence (WPPSI), after adjustment for potential confounders, we found that children highly exposed to PAH (> the median of 2.26 ng/m3) had Full-scale and Verbal IQ scores that were 4.31 and 4.67 points lower, respectively, compared to less exposed children (Perera, et al., 2010). These reductions are similar in magnitude to the effects of low-level lead exposure and show that prenatal exposure to PAH at levels encountered in NYC air adversely affected child IQ scores at 5 years of age.
Using a more informative scale for older children, the Weschler Intelligence Scale for Children (WISC), the same children were tested for IQ at ages 7 and 9. After adjusting for potential confounders, high PAH exposure was inversely associated with Full-scale IQ and Working Memory at ages 7 and 9 and with Verbal IQ and Processing Speed at age 9. It is known that toxic exposures are higher in communities of color, and frequently co-occur with chronic economic deprivation. With this knowledge, we examined the interaction between prenatal PAH exposure, measured by presence or absence of adducts in cord blood, and material hardship, based on questions regarding housing, food, electricity, and clothing given prenatally, and child age 6 months, and 1, 2, 3, and 5 years, on child IQ at 7 years. After adjustment for potential confounders, we found significant (p<0.05) inverse effects of PAH exposure on Full-scale IQ , Perceptual Reasoning, and Working Memory, in children of mothers with material hardship during pregnancy or recurring material hardship during the child’s early years, and not in children whose mothers did not experience material hardship. Additionally, there was a significant interaction (p<0.05) between high cord adducts and prenatal hardship on Working Memory and between high cord adducts and recurrent material hardship (Vishnevetsky, et al., 2015). These results indicate a need for a multifaceted approach to intervention.
Continuing our work on PAH exposure and neurodevelopmental outcomes, we found that high PAH exposure, measured as number of DNA adducts specific to benzo[a]pyrene (BaP), a representative PAH, in cord blood, was significantly (p<0.05) positively associated with symptoms of anxiety/depression at age 4.8 and with attentional problems at ages 4.8 and 7 years on the Child Behavior Checklist (CBCL) after adjusting for potential confounders. Additionally, higher PAH exposure was associated with 8.30 times the odds of having a borderline or clinical classification on the Diagnostic and Statistical Manual of Mental Disorders (DSM)-oriented Anxiety Problem Scale at age 4.8 years (p< 0.037) (Perera, et al., 2011).
Further examining the relationship between prenatal PAH exposure and symptoms of Anxiety/Depression and Attentional Problems on the CBCL, we used both prenatal air monitoring data and measurement of DNA adducts specific to BaP, a representative PAH, in both maternal and cord blood to determine if the relationship persisted using additional markers of PAH exposure. In our model, after adjusting for potential confounders, we found that high prenatal air monitoring, maternal BaP adducts and cord BaP adducts were significantly (p<0.05) positively associated with symptoms of anxiety/depression and attentional problems on the CBCL measured at ages 6-7. High PAH measured via prenatal air monitoring was associated with 4.59 times the odds of having borderline or clinical classification on the DSM (p<0.009) (Perera, et al., 2012). These consistent results provide evidence that environmental PAH at levels encountered in NYC air can adversely affect child behavior, especially internalizing problems, including both anxiety/depression, and attention problems that could affect school performance.
These results are consistent with those in the Center's parallel experimental studies that have observed effects of prenatal PAH on anxiety-like behaviors in mice (Project 3).
High prenatal PAH exposure, represented by maternal adducts, also was significantly related to attention deficit hyperactivity problems in children at age 9. After adjusting for relevant confounders, high PAH exposure was significantly (p<0.05) associated with increases on the DSM-IV total, DSM-IV Hyperactive-Impulsive and DSM-IV Inattentive subscales of the Conners’ Parent Rating Scale (CPRS) in addition to increased scores on the Conners’ ADHD Global Index (Perera, et al., 2014). These analyses adjusted for important potential variables, including characteristics of the home environment, maternal ADHD and comorbid anxiety/depression in the children.
In an analysis of MRI brain scans in 40 cohort children, 20 with high PAH exposure and 20 with low, we assessed the effects of prenatal PAH exposure on brain structure and the cognitive and behavioral correlates of those abnormalities at ages 7-9. There was a dose-response relationship between increased prenatal PAH exposure, air monitoring measured in the third trimester, and reductions of white matter surface in later childhood. These reductions were significantly (p<0.05) associated with slower information processing speed, on the WISC-IV and numerous CBCL measures, including externalizing problems and DSM ADHD symptoms (Peterson, et al., 2015).
Building on previous results from the cohort showing associations between PAH exposure and Anxiety/Depression symptoms on the CBCL, we evaluated the relationship between PAH, measured via adducts in maternal blood, and deficient emotional self-regulation (DESR) defined as moderate elevations on three specific scales of the CBCL (Anxious/Depressed, Aggressive Behavior, and Attention Problems) and social competence (measured by the Social Responsiveness Scale) at multiple time points between ages 3 and 11. Additionally, we measured the association of impaired self-regulation with social competence and whether impairment in self-regulation mediates the association of prenatal exposure to PAH with social competence. Using Generalized Estimating Equations to assess PAH on DESR, there was a significant interaction between exposure and time (p<0.05) that showed the developmental trajectory of self-regulatory capacity was delayed in the exposed children. Multiple linear regression revealed a positive association between presence of PAH adducts and problems with social competence (p<0.04), level of dysregulation and problems with social competence (p<0.0001), and evidence that self-regulation mediates the association of prenatal exposure to PAH with SRS (p<0.0007) (Margolis, et al., submitted to JCPP).
Aim 1: BPA exposure
We analyzed the association between maternal prenatal bisphenol A (BPA) exposure (dichotomized into high/low groups at the upper quartile) and child behavior at ages 3-4 (assessed via the CBCL), using generalized linear models, adjusting for postnatal BPA exposure and other potential confounders. We observed significant interactions (p<0.05) between prenatal BPA urinary concentrations and child sex, on Emotionally Reactive, Aggressive Behavior, and Internalizing Problems scales of the CBCL. After stratifying on sex, the BPA effects were positive and significant among boys on Emotionally Reactive and Aggressive Behavior indicating that boys with prenatal BPA exposure in the highest concentration quartile had, on average, more reported symptoms of problems in these areas. In contrast, among girls, high BPA exposure was associated with lower scores for Anxious/Depressed and Aggressive Behavior (p<0.05), and Internalizing Problems (p<0.1), indicating that girls in the high prenatal BPA exposure group had, on average, fewer reported problems in these areas than girls in the low exposure group. Postnatal BPA urinary concentration alone only had a significant negative effect only on Emotionally Reactive within the entire sample. Comparison of results before and after adjusting for postnatal BPA exposure found the effect estimates to be similar, suggesting that the prenatal period may be a more sensitive window for BPA exposure.
We built on these findings by analyzing the effect of maternal prenatal BPA (dichotomized into high/low groups at the upper tertile) and child behavior at ages 7-9 years (assessed by the CBCL) to determine if the association of sex-specific behavior differences persisted. Using a stratified model, adjusted for relevant confounders, including postnatal BPA, boys in the highest BPA tertile had significantly (p<0.05) higher Internalizing and Externalizing composite scores, and a higher score on their corresponding syndrome scales. Girls in the highest tertile showed a significant decrease (p<0.05) in the Internalizing composite score (Roen, et al., 2015). Together these results suggest BPA exposure may affect childhood behavioral outcomes in a sex-specific manner, and differently depending on timing of exposure.
Our recent results at ages 10-12, using outcome measures specific to anxiety and depression, the Revised Children’s Manifest Anxiety Scale (RCMAS) and Children’s Depression Rating Scale (CDRS), respectively, found that maternal prenatal BPA exposure (dichotomized into high/low groups at the upper tertile) is significantly associated (p<0.05) with increased RCMAS total score, RCMAS social concern scale, and CDRS total scale in boys, but there are no significant associations in girls. These results indicate the persistence of sex-specific effects of prenatal BPA exposure on the symptoms of anxiety and depression (Perera, et al., manuscript in prep.).
Aim 2
We have successfully completed pyrosequencing assays to assess the methylation of COX-2, ERα, CEBPα, PDE4D, and TH. We currently are analyzing the association between prenatal PAH and COX-2 and between prenatal BPA and ERα, CEBPα, PDE4D, and TH, respectively. We also are analyzing associations between the methylation of CpG sites on these genes and neurodevelopmental endpoints including mental and psychomotor developmental indices (from the Bayley Scales at ages 12, 24, and 36 months), IQ (from the WISC at age 7) and child behavior (from the CBCL at ages 3, 5, 7, and 11). We currently are working to optimize the analysis of additional CpG sites on the genes listed above and to develop pyrosequencing assays for the remaining gene candidates (NMDAR2b, CCL17, AhR, INF-γ, and THRβ).
We subsequently added brain-derived neurotrophic factor (BDNF) to our list of candidate genes based on experimental studies in Project 3 (see progress report for Project 3) and results of our parallel studies in a Chinese cohort showing associations of BDNF with prenatal PAH exposure and cognitive development. Our new approach synchronizes our human and animal data to select additional CpG sites of interest, taking advantage of our parallel experimental animal studies that allow us to assess the concordance between CpG-specific methylation related to either PAH or BPA and gene expression in target tissue, which is not accessible in the human cohort. After checking the homology between mice and humans, we map the sequence of interest and identify ‘indicator’ CpG sites located within or around this region that have been assayed using two genome-wide techniques (Infinium 450K and HELP-tagging) in a subset of our cord blood samples from the cohort. Using these indicator CpGs, we can assess the association between methylation and PAH or BPA exposure, as well as neurodevelopmental outcomes. If we identify a signal between either exposure or neurodevelopment at these indicator CpGs and demonstrate that there is sufficient variation in cord blood at these locations for follow-up analysis, we then develop pyrosequencing assays covering the CpG sites of interest to measure the methylation status of these CpGs in the cord blood samples of the full cohort. This analysis is done on a gene-by-gene basis; thus far, we have completed BDNF and are following a similar approach for ERα. We plan to complete remaining genes and data analysis in the coming year.
It is widely recognized that because DNA methylation is cell specific, failure to account for cell distribution when examining methylation in a mixed cell population can confound hypothesized exposure-to-methylation associations, leading to spurious results or failure to detect true relationships. Methods have been developed using a regression calibration approach to adjust for cellular mixture requiring reference methylation data. Currently, two reference datasets are available, both derived from adult blood. Cells represented in these datasets may not reflect the most prevalent cellular components of cord blood, leading some to question whether they are optimal when applied to cord blood. We have generated a 450K reference dataset using cord blood samples and currently are evaluating whether this reference dataset works better at adjusting for cell mixture in a cord blood matrix than the reference datasets from adult blood. This work was presented at the PPTOX meeting and a manuscript currently is in preparation.
Aim 3
The proportion of households below the federal poverty line in a participant's neighborhood (1-km street network buffer) is significantly (p<0.05) associated with reduced scores on the psychomotor development index (PDI) and mental development index (MDI) on the Bayley Scales of Infant Development at age 3 (Lovasi, et al., 2010).
We found that the neighborhood context variables, (given by self-report and geocoded into a 1-km network buffer around the home) building dilapidation and linguistic isolation were significantly (p<0.05) associated with lower performance IQ scores (measured by the Wechsler Pre-school and Primary Scale of Intelligence-Revised) at age 5 (Lovasi, et al., 2014). These data demonstrate that early life PAH and neighborhood-level linguistic isolation exposures independently predict lower cognitive abilities at age 5.
We geocoded 841 prenatal addresses from the study area in Northern Manhattan and The Bronx and are continuing the process for later ages (currently 4,693 addresses geocoded). In preliminary analyses of our GIS work, we found that neighborhood percent poverty confounds the effect of cumulative PAH exposure on hyperactive-impulsive behaviors at age 9. The effect of cumulative PAH exposure is underestimated without adjusting for neighborhood percent poverty, as neighborhood % poverty is positively associated with hyperactive-impulsive behaviors at age 9 (p<0.01) and negatively associated with cumulative PAH exposure (p=0.19). The effect of cumulative PAH exposure on hyperactive-impulsive behaviors at age 9 is modified by neighborhood-level educational attainment (p=0.67) in the low educational attainment stratum and (p=0.02) in the high educational attainment stratum. However, a formal test of interaction does not reach statistical significance (p=0.12).
We are using the results from the GIS work to inform our future analyses of the associations between endocrine disruptors and neurobehavioral outcomes. These neighborhood-level factors are potential effect modifiers and confounders; and our previous results allow us to build the most accurate models possible.
Mechanistic Core:
Aims 1 & 2
Studies were conducted by personnel trained in rodent phenotyping and behavioral neuroscience (Kathryn Gudsnuk) and in molecular epigenetics (Dr. Maria Kundakovic). The oral daily administration of BPA in mice was an effective strategy in achieving dose-dependent variation in both behavioral and molecular outcomes in mice and is viewed as a more optimal strategy for daily gestational exposure than more stress-inducing procedures such as gavage and injection. Urine levels confirmed the dose-dependent increases in BPA exposure, corresponding to a vehicle, 2 µg/kg, 20 µg/kg, and 200 µg/kg dosing regime. Initial analyses focused on behavioral and brain gene expression changes with follow-up analyses of DNA methylation in identified target genes. Due to the inclusion of multiple doses and both male and female offspring, our analyses were able to reveal a dose-dependent sex by BPA interaction on hypothalamic expression of estrogen receptor isoforms (alpha and beta). This same pattern of effect was observed in the expression of DNA methyltransferases. The curvilinear dosage effects were evident in the hypothalamus and cortex but less evident in other brain structures, such as the hippocampus. Epigenetic changes in the estrogen receptor alpha (Esr1) gene promoter revealed that BPA-induced changes in DNA methylation in the brain varied by sex and by brain region. Behavioral effects of prenatal BPA included increased social investigation and reduced aggression at the low dose exposure, with increased aggression at the 200 µg/kg dose in both males and females. This work was published in Proceedings of the National Academy of Sciences in 2013 (PNAS, 110(24):9956-9961). This paper also included findings that there are BPA-induced changes in the maternal behavior of treated dams, and our analyses have confirmed that these postnatal influences may impact specific behavioral and epigenetic outcomes. We currently are working with human developmental psychologists and environmental health researchers to determine whether these BPA-induced effects on mother-infant interactions are relevant in human studies.
Consistent with human studies of prenatal BPA-induced effects, on some measures we observe more severe behavioral impairments in male offspring in mice. Working memory was found to be impaired in male offspring that had received 200 µg/kg BPA, with no impairments observed in female offspring. Consistent with this behavioral finding, we determined reductions in hippocampal expression of brain derived neurotrophic factor (BDNF) – a critical mechanism regulating working memory – in male offspring. Epigenetic analyses revealed that in the hippocampus, male offspring exposed to prenatal BPA had increased DNA methylation of the BDNF gene promoter (Figure 1). These epigenetic changes also were observed in blood samples – indicating that peripheral DNA can be used to predict epigenetic changes in the brain. Analyses of human cord blood samples from the human cohort revealed that the BPA-associated epigenetic effects observed in mice were likewise observed in humans (Figure 2), suggesting a possible biomarker of exposure and neurobehavioral outcomes. This work was recently published in Proceedings of the National Academy of Sciences (PNAS, 112(22):6807-6813.).
Analyses of growth trajectories indicated significant weight gains in males exposed to 2 µg/kg BPA (compared to vehicle) – an effect evident at weaning that persisted until adulthood. However, we did not observe BPA-induced changes in body fat weights. We currently are exploring the expression of genes within the placenta that regulate growth and neurodevelopment to determine how these are impacted by prenatal BPA. These analyses may lead to additional studies in which human placental samples can be analyzed.
Figure 1. BPA-induced changes in a) memory, b) expression of BDNF, and c) BDNF CpG methylation.
Figure 2. BPA-associated changes in BDNF CpG methylation in human cord blood samples.
Aims 3 & 4
The components of the PAH aerosol have been published by us previously (Chu, J. Toxicol., 2012) Measured levels of the particulate fraction of PAH collected from the filters in the PAH mouse exposure chamber suggested good exposure of mice during experimental, but not during negative control, delivery conditions (Table 1).
| Exposure | N | Mean ± SE | Range |
|---|---|---|---|
| Normal Air | 11 | 3.03 ± 0.90 | 0-9.00 |
| PAH | 11 | 23.24 ± 3.05 | 7.38 - 40.00 |
Table 1. Pyrene concentrations (ng/m3) in exposure chamber
Monitors to measure ambient PAH exposures in the mouse housing units indicated no airborne contaminating PAH exposure.
Female BALB/c mice shortly after mating were exposed to PAH versus nebulized negative control solution alone in the PAH-chamber unit 5 hours a day, 5 days a week, over several rounds of exposure as shown below (Figure 3).
Figure 3. Experimental design.
Mice were weighed every other day. In addition, epigenetic and clinical (i.e., open field, light dark box) neurological outcomes were assessed at PND 60.
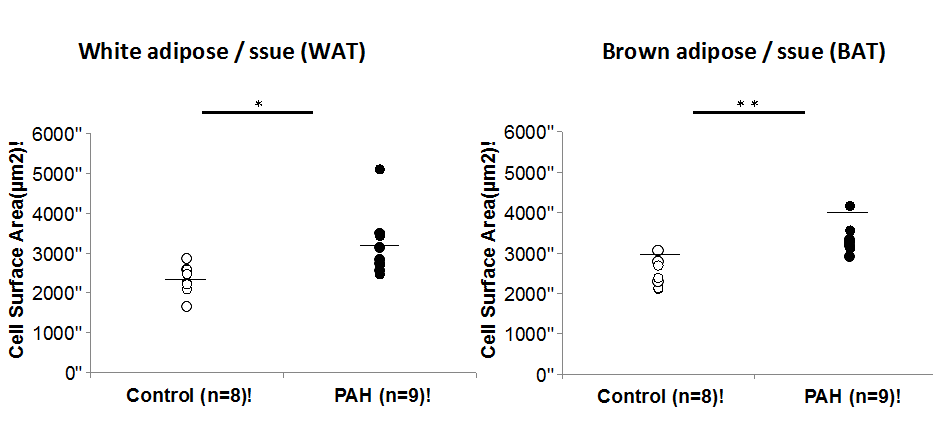
We found that offspring of dams exposed to greater PAH during gestation had increased weight (Figure 4, left), fat mass (Figure 4, right), as well as higher gene expression of PPARg, C/EBPa, Cox2, FAS and adiponectin and lower DNA methylation of PPARg (Figure 5).
Figure 4. Offspring mice were heavier following prenatal PAH exposure (left), and developed bigger adipocytes (mean surface area, white and brown fat) (right).
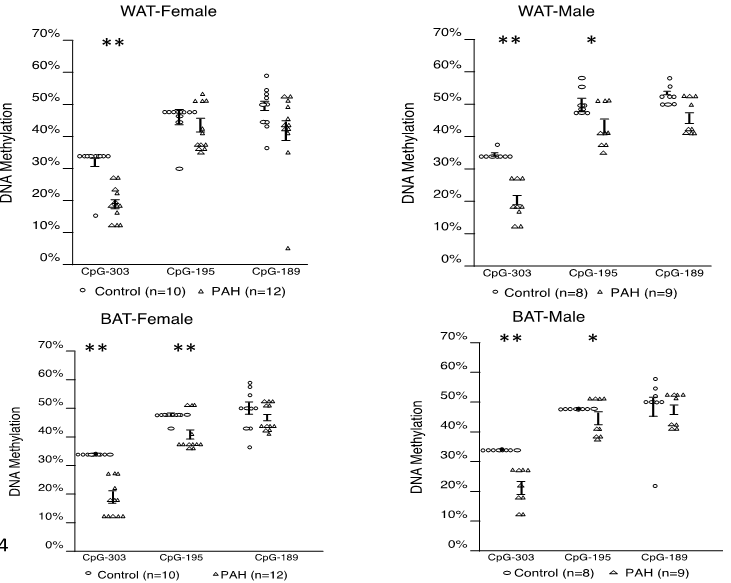
Figure 5. Prenatal PAH was associated with decreased DNA methylation PPARg promoter at CpG-303, -195, -189.
Of note, PPARg promoter adipose tissue methylation and expression negatively correlated in offspring, consistent with methylation-induced gene silencing. Moreover, the average methylation level of three CpG sites in PPARg promoter correlated negatively with body weight and body mass index at PND 60 (r=-0.43 and -0.62, 95% CI -0.68, -0.17 and 95% CI -0.78, -0.38, p=0.003 and p=0.00003, respectively, n=38) and cell surface area at PND 60 (r=-0.61, 95% CI-0.84, -0.18, p=0.01, n=17). Similar differences in phenotype, DNA methylation and gene expression extended through the grand offspring mice.
Further, prenatal PAH exposure was associated with more activity, and anxiety-like behaviors (Table 2), and decreased relative expression of BDNF and NRDAR2b in the mouse cerebral cortex (Table 3). High methylation of BDNF at exon 4 was found as well.
Table 2. Prenatal exposure was associated with less time idle and more sniffing.
| Time Idle | Sniff face/body | Sniff anoregion | Number of fecal boli | |
|---|---|---|---|---|
| Negative Control Air | 3.43 ± 0.46 (n=20 mice) | 1.66 ± 0.12 (n=20 mice) | 1.17 ± 0.10 (n=20 mice) | 4.39 ± 0.27 (n=56 mice) |
| PAH | 2.03 ± 0.28 (n=8 mice) | 2.52 ± 0.33 (n=8 mice) | 1.59 ± 0.15 (n=8 mice) | 5.65 ± 0.48 (n=47 mice) |
| P value | 0.05 | 0.01 | 0.025 | 0.054 |
Table 3. Prenatal PAH exposure was associated with reduced BDNF expression in the cortex.
| Control (n=10) | PAH (n=10) | P-value | |
|---|---|---|---|
| BDNF Exon 3 | 3.14 ±0.48 | 0.95 ± 0.11 | <0.0001 |
| BDNF Exon 4 | 3.36 ± 0.50 | 1.01 ± 0.13 | <0.0001 |
| NMDAR2b | 5.86 ± 1.15 | 1.34 ± 0.13 | <0.0001 |
Our findings suggest that prenatal exposure to BPA or PAH can have a lasting impact on tissue-specific gene expression patterns and that this effect is associated with epigenetic changes within the regulatory regions of target genes. We believe that these molecular alterations mediate the behavioral, neurodevelopmental and metabolic effects associated with BPA or PAH exposure. In the case of BPA, we have determined that there are non-linear dosage effects of BPA on outcomes – an important challenge to the “increased dose – increased disruption” assumption in toxicology and environmental health research. We also have determined that social experiences (mother-infant interactions) may moderate or mediate the effects of BPA. This finding has implications for future studies in humans and intervention strategies that focus on the interplay between toxin exposure and the social environment. We also have determined that human epigenetic biomarkers can be used to detect the impact of BPA and PAH exposure. Future studies can use these epigenetic biomarkers to determine whether long-term neurobehavioral outcomes associated with BPA and PAH exposure in humans are predicted by this epigenetic change.
Data Management and Statistics Core:
All Columbia Center for Children's Environmental Health (CCCEH) project data are housed in a Scientific Information Retrieval (SIR) database management system that is maintained by the Data Management, Statistics and Community Modeling (DSM) Core.
The DSM Core also is the central resource for statistical consultation and data analysis related to the development of manuscripts. Data security is ensured at the level of the server, the user, and the database. All data are fully compliant with Health Information Portability and Accountability Act (HIPAA) regulations. The DSM Core holds a meeting every Thursday at 10:30 am. The meeting is open to any project investigator and provides consultation with respect to the development of data requests, statistical analysis, interpretation of findings, and manuscript review. Dr. Gina Lovasi heads the community modeling component of the DSM. Lori Hoepner, DSM project coordinator, maintains the central database, which now contains a distinct record type (table) containing data for each of the scheduled interviews through the 11th year of life. Project-specific updates are outlined below.
Project 1–Endocrine Disruptors & Obesity Among Inner-City Children
Project 1 has enrolled 400 cohort subjects. Fasting blood lipids, insulin, and blood glucose have been analyzed for 400 subjects. Tanner staging and blood pressure assessment by a clinician has been performed on 393 subjects and food frequency questionnaires have been administered to 386 subjects. James Quinn, GIS expert, has geocoded the 4693 unique current addresses at the time of interview for all CCCEH subjects through age 11 years.
Project 2–Endocrine Disrupters, Epigenetic Mechanisms & Neurodevelopment
The neurodevelopment assessment component of the database has been developed and data has been entered for 387 cohort subjects. Data elements include, but are not limited to: the NEPSY-2, Purdue, KSADS, Social Responsiveness Scale, Social Communication Scale and the Weather Task Questionnaire.
Project 3–Molecular/Disease Consequences of Prenatal BPA, PAH Exposure Across Generations
The DSM has devised plans for statistical review and data storage for this project once data become available.
Community Outreach and Research Translation Core:
Aim 1
In 2010, CCCEH, working with WE ACT, restructured and expanded the Community Advisory and Stakeholder Board (CASB) to include representatives from a local community board, community-based organizations with an active focus on obesity and child development, and government agencies. In collaboration with WE ACT, CCCEH defined roles for the members and created a list of candidates. CCCEH and WE ACT invited organizations from the final candidate list to join the CASB and the first meeting was held in September 17, 2010.
In 2011, the CASB held six meetings to maintain engagement and added members from Northern Manhattan Perinatal Partnership and Nos Quedamos. In 2012, the CASB expanded to include representatives from Autism Speaks, East Harlem Asthma Center of Excellence, Isabella Geriatric Center, and New York Presbyterian Hospital Women, Infant, and Children (WIC) Program.
In 2013, CCCEH expanded the CASB to include active members from the community and governmental agencies engaged in advocacy and community awareness of environmental health issues—emphasizing the significance of endocrine disrupting chemicals and child development. Active engagement of the CASB was maintained through quarterly meetings and incorporation of their feedback into the Center's education and outreach strategies. Following the collaborative development of materials for the Healthy Home Healthy Child (HHHC) educational campaign in 2013, CASB member organizations have taken an active role in requesting and disseminating the HHHC materials to their own participants.
Since the first CASB meeting on September 17, 2010, the Board has grown in membership from 9 to 17 members in 2015. The current list of CASB members includes representatives from A.I.R. NYC, Autism Science Foundation, Bronx Borough President’s Office, Columbia University Head Start, Columbia Community Partnership for Health, East Harlem Asthma Center of Excellence (NYC Department of Health and Mental Hygiene), EPA Region 2, Harlem Children’s Zone, Harlem Health Promotion, Mom’s Clean Air Force, New York – Presbyterian Hospital WIC Program, Northern Manhattan Improvement Corporation, Northern Manhattan Perinatal Partnership, Nos Quedamos and Urban Health Plan, Inc.
The CASB met twice in 2015 and plans to continue meeting on a quarterly basis.
Aim 2
Over the grant period, COTC staff designed an integrative pest management brochure, a tip-sheet on reducing Bisphenol A (BPA) exposure, and a holistic children’s environmental health campaign.
In 2010, Core staff produced a newsletter about asthma prevention and management for the parents in our cohort, and for the wider Northern Manhattan and South Bronx community. Printed in Spanish and English, the newsletter provides resources for families seeking help in managing their children’s asthma, and information about joining clean air campaigns and other community initiatives. COTC staff distributed these materials to local OB/GYN, pediatric clinics, and community health workers, informing pregnant women and parents about asthma, and tips to reduce exposures to common environmental triggers. Core staff also worked to finalize brochures on non-toxic cleaners and Integrated Pest Management targeted to community members.
In 2011, Core staff produced an informative brochure explaining integrated pest management (IPM)—a pesticide free method to pest control. Printed in Spanish and English, COTC designed the brochure for the parents in the Center’s NYC cohorts, and the wider Northern Manhattan/ South Bronx communities. COTC staff distributed these and past educational materials to local community-based organizations, faith-based organizations, and OB/GYN and pediatric clinics and private practices, alerting pregnant women and parents to health risks for children exposed to common environmental pollutants. Core staff began working on developing a tip sheet highlighting best methods to reduce BPA exposure, with input from CASB members and community residents.
In 2012, Core staff conducted three focus groups to help inform the development of a tip sheet on reducing exposure to BPA. The finalized tip sheet was tailored for parents in the main cohort and the wider Northern Manhattan/South Bronx communities and was printed in Spanish and English. To educate caregivers about health risks to children, COTC staff distributed our materials on BPA, IPM, and asthma to local community-based organizations, and medical practices. Core staff and the CASB began developing a holistic educational campaign highlighting positive messages to promote environmental health through adoption of healthy habits, such as green cleaning and IPM that reduce exposure risks.
Also in 2012, COTC developed a role-modeling health education project for Center research workers to promote healthy lifestyle changes through personal testimony on how they incorporate tips to reduce environmental health exposures in their daily routine. When study participants arrived at the Center, they could view a photo of their research worker along with two action steps aimed at reducing exposures to environmental toxicants.
In 2013, COTC staff and the CASB completed work on a holistic educational campaign promoting environmental health. The COTC and the CASB identified and developed content for the following six themes: Clean Indoor Air, Green Cleaning, Eat Fresh, Create Change, Reduce Stress, and Toxic-Free Shopping. In July 2013, the Center completed the set of these six health tips aimed at women and children in Northern Manhattan and the South Bronx. These materials utilize the Center’s new focus on providing holistic strategies to reduce exposure to a range of environmental toxicants rather than focusing on one chemical or health outcome.
Since September 2013, the Center has distributed more than 20,000 HHHC holistic campaign brochures to the Northern Manhattan and South Bronx communities through CASB members, local nonprofit organizations and direct outreach. The COTC tracks the number of brochures distributed, the requesting organizations, and the organizations’ intended use of the brochures. The tracking records allow the COTC to gather information about the reach of the distribution and collect information about how organizations are incorporating these materials into their health education programming. Requesting organizations include New York Presbyterian Hospital WIC Program, Harlem Health Promotion Center, Harlem Children’s Zone, Department of Health and Human Services Administration for Children and Families, Columbia Community Partnership for Health, WE ACT for Environmental Justice and the NYC Department of Health and Mental Hygiene.
As a part of the HHHC campaign, the Center created a children’s activity book that incorporates the six holistic themes into imaging and activities intended to engage both child and parent. The COTC coordinator distributes these books at health fairs and other outreach activities.
Workshops: COTC staff used the educational messaging of the HHHC campaign to develop workshops over the grant period. In 2012, the COTC coordinator hosted three workshops: two for CASB member organizations (NYC Department of Health and the Isabella Geriatric Center), and one for Columbia University staff.
Monthly Tips: In September 2012, COTC staff started creating monthly tips, which offer guidance on limiting exposures to environmental harms. Monthly tips are disseminated through the Center website and social media (Facebook and Twitter). Other Center educational materials have been disseminated to the community through multiple avenues, including distribution of materials at local farmers' markets, health fairs, and partner community organization.
Outreach to Cohort: In October 2009, COTC staff organized a family event for study participants. The purpose of the event was to encourage retention and to provide health education resources to CCCEH Northern Manhattan/South Bronx cohort families. Representatives from the Cornell University Cooperative Extension, WIN for Asthma, and WE ACT were on hand to provide families with information and environmental health resources, and to answer questions about how to reduce environmental health threats.
On April 26, 2014, the Center, WE ACT, and Community League of the Heights' (CLOTH) Community Health Academy cohosted an environmental health fair. The 2014 Green Kids Health Fair provided information about environmental health, gave children and adults a place to be physically active while promoting the messaging of the HHHC campaign. The event brought together community organizations to provide information to residents, including lead testing by Microecologies, lead education and resources from WE ACT, Corbin Hill Farms, and the NYC Department of Emergency Management. Other highlights included Zumba, yoga for kids, and physical activity games.
Aim 3
COTC staff continued meeting with the Center’s Pediatric Health Specialist (Dr. Oberfield) and Administrative Core staff to discuss strategies for presenting scientific research findings at Grand Rounds, community health events, local policy forums, and trainings of local leaders on environmental health risks.
A Grand Rounds on prenatal exposures to endocrine disruptors took place at the Columbia University Medical Center Pediatrics Department on October 15, 2010. Drs. Perera, Oberfield, and Rundle presented. COTC staff helped prepare the presentation and designed a tip sheet for physicians and their patients, which was distributed at the event. Core staff scheduled investigator testimony and presentations to a variety of audiences including community groups and local non-profits. Over the grant period, the Center Director and investigators presented findings to staff of the New York City Council (at their invitation), Washington Heights Community Board 12, EPA regional children’s health staff, and participated with WE ACT in a webinar hosted by the National Center for Children In Poverty. Center investigators also presented at a town hall meeting organized by WE ACT and the NIEHS Northern Manhattan Center for Environmental Health in April 2010.
During 2011, Center investigators made 19 presentations to scientific or clinical groups during the year, including presentations by Drs. Perera, Miller, Ho, Whyatt, Rauh, Horton, Oberfield, Rundle, Herbstman, Champagne, Donahue, Iyer, Chillrud and Kinney.
In 2012, COTC staff and Ms. Peggy Shepard presented "A Partnership between CCCEH and WE ACT for Community Outreach and Engagement" at the 2012 Children's Environmental Health Symposium. The COTC Program Coordinator, Brennan Rhodes, also presented "Identifying Perceptions, Attitudes and Health Behaviors around Bisphenol-A Through Community-Based Focus Groups" at the 2012 Partnerships for Environmental Public Health Conference. Center investigators made 33 presentations to scientific or clinical groups during the year, including presentations by Drs. Perera, Miller, Whyatt, Rauh, Evans, Rundle, Herbstman, Perzanowski, Hoepner, Andrews, Orjuela, Just, and Iyer.
Throughout the 2012-2013 grant year, Center staff translated our findings to scientific, public health, and clinical groups. WE ACT’s Peggy Shepard discussed the partnership with the Center in her presentation “Working Against the Clock: Protecting Children’s Health, Safety and their Futures” at the 2012 APHA Annual Meeting. Center investigators, including Drs. Perera, Miller, Rauh, Rundle, and Herbstman, made 21 presentations on our research during the year at grand rounds, online forums, and conferences.
In 2014, Center staff translated our findings to scientific, public health, and clinical groups. The successful partnership between the Center and WE ACT has been highlighted by WE ACT’s Peggy Shepard at the 2014 EPA/NIEHS Children’s Centers Webinar Series as well as COTC co-director Dr. David Evans and WE ACT’s Ogonnaya Dotson-Newman at a conference, "Bridging the Expertise of Communities and Academia", hosted by the Irving Institute of Clinical and Translational Research. Center investigators, including Drs. Perera, Miller, Rauh, Rundle, and Herbstman, made over 40 presentations on our research during the year at grand rounds, online forums and conferences throughout 2014.
In 2015, Center staff made several presentations to public health, scientific, and clinical groups. Dr. Perera spoke on the fetal window of susceptibility in an NIEHS Distinguished Lecture. In September 2014, Alyssa Creighton attended the “PEPH Annual Meeting: Communication Research in Environmental Health Sciences – Environmental Health Literacy” in Research Park Triangle, NC, and presented a poster entitled “Streamlining Environmental Health Messaging: Lessons Learned from the Healthy Home, Healthy Child Educational Campaign.” Center investigators, including Drs. Perera, Miller, Rauh, Rundle and Herbstman made more than 12 presentations on our research during the year at grand rounds, online forums, and conferences.
Stakeholder Briefing Series
Each year, the Center and WE ACT host a community stakeholder briefing, inviting community leaders, elected officials, and policymakers to engage in a dialogue about the Center’s research findings.
On October 25, 2011, at the first stakeholder briefing breakfast, Dr. Frederica Perera discussed health risks associated with prenatal and early exposure to diesel exhaust, pesticides, indoor air allergens, and endocrine disrupting chemicals. WE ACT’s Executive Director Peggy Shepard talked about the current state of policy reform regarding exposures common in our urban communities. More than 30 stakeholders engaged in an active discussion on how to translate Center findings to encourage advocacy and prevention. Attendees included representatives from the Department of Health and Mental Hygiene, the Environmental Defense Fund, the Environmental Protection Agency, and other health and community service organizations.
The following year, on October 2, 2012, the second stakeholder briefing focused on endocrine disrupting chemicals. Dr. Frederica Perera discussed neurodevelopmental health risks associated with bisphenol A (BPA); Dr. Andrew Rundle spoke about his work linking obesity with polycyclic aromatic hydrocarbons (PAH); and WE ACT’s Executive Director, Peggy Shepard, presented on the current state of policy reform regarding endocrine disrupting exposures at the local, state, and national levels. Attendance doubled at the second stakeholder briefing, with more than 60 people attending the event, including representatives from the NYC Department of Health; the New York State Office of the Attorney General; the Environmental Protection Agency; the Center for Health, Environment & Justice; and other health and community organizations.
The third stakeholder briefing took place on October 23, 2013. The briefing focused on clean air and included presentations from Center Director Dr. Frederica Perera, Columbia University professor Dr. Diana Hernandez, and WE ACT federal policy analyst Dr. Jalonne White Newsome. There were 65 attendees from a variety of organizations, such as the NYC Department of Health and Mental Hygiene, Environmental Defense Fund, and Northern Manhattan Improvement Corporation. The event was open to community leaders, elected officials, and policymakers to engage in dialogue about our research findings and to discuss how these organizations can be more involved with the Center and WE ACT. As in past years, the event received very positive feedback from the participants.
Aim 4
In 2010, working in collaboration with the Department of External Affairs at the Mailman School of Public Health and CCCEH investigators, COTC staff assisted in the development and dissemination of five press releases. COTC helped draft a press release about findings from the parallel Polish study that confirmed earlier CCCEH findings with respect to prenatal PAH exposure and children’s IQ at age 5. COTC staff also organized and facilitated interviews with several news organizations, including a visit to the Center by a reporter from the New Yorker Magazine. Finally, COTC staff developed a listserv of individuals and organizations interested in receiving updates on the Center’s scientific findings.
During 2011, in collaboration with the Department of External Affairs at Mailman School of Public Health and CCCEH investigators, COTC staff assisted in the development and dissemination of press releases about Center-related findings. This includes the publication from Dr. Virginia Rauh entitled Prenatal Exposure to Common Insecticide Linked to Decreases in Cognitive Functioning at Age Seven, which was cited in more than 250 articles online. COTC staff also organized and facilitated interviews with several news organizations, including a Time magazine cover story on October 4, 2010, which highlighted the work of CCCEH and Director Dr. Frederica Perera. Other media coverage includes CNN, NPR, ABC World News Report, Today Show, and others.
In 2012, working with the Mailman School of Public Health, COTC staff helped develop and disseminate press releases about Center findings. Dr. Virginia Rauh’s paper about Prenatal Exposure to Insecticide Chlorpyrifos Linked to Alterations in Brain Structure and Cognition was featured in more than 90 online media outlets, including Scientific American, CNN, FOX News, and ABC News. COTC staff also organized and facilitated interviews with news organizations, including the Wall Street Journal cover story on November 8, 2011, which highlighted the work of CCCEH and Director Dr. Frederica Perera. Other media coverage includes a CUMC documentary about air pollution and health, and stories in Business Week, US News and World Report, CNN, NPR, and others. COTC staff continued distributing “Center Updates” on new findings to our listserv of more than 900 subscribers, created a Facebook page and Twitter account to provide children's environmental health resources, and assisted with the Center’s website redesign process.
In 2013 the Center’s findings from 2012 linking BPA exposure and asthma were featured in more than 50 online media outlets, including TIME, CNN, and Scientific American. COTC staff helped to coordinate on-air interviews, including spots on CBS This Morning highlighting Dr. Herbstman and a NY1 interview with Dr. Robin Whyatt about BPA. Other media coverage included a highlight in the Mailman School of Public Health magazine about the Center’s air pollution research and stories in NY Times, PBS, Chicago Tribune, Bloomberg, Huffington Post, and others. Center findings were distributed through “Center Updates” to the Center listserv of more than 1,200 subscribers, posts on our Facebook and Twitter accounts, and highlights on the Center’s website.
In 2014, COTC staff continued to work closely with the Mailman School of Public Health in the development and dissemination of press releases about Center findings. The Center’s recent findings were featured in more than 60 online media outlets, including TIME, Bloomberg News, Scientific American, the Guardian, and On Earth Magazine. Center findings also were distributed through “Center Updates” to the Center’s listserv of more than 1,200 subscribers, posts on the Center’s Facebook and Twitter accounts, and highlights on the Center’s website.
In 2015, the Center’s findings on prenatal exposure to PAH and
Conclusions:
During this funding period the Center has had many accomplishments from both our research and community outreach efforts. In terms of research on the relationship between early exposures to the endocrine disrupting chemicals, polycyclic aromatic hydrocarbons (PAH), and bisphenol A (BPA), and the outcomes of obesity and neurodevelopment in our cohort, we reported that prenatal PAH exposure was significantly associated with an increased risk of obesity at ages 5 and 7 (Rundle, et al., 2012) and a decrease in IQ at age 5 (Perera, et al., 2009). We also reported a link between prenatal BPA exposure and sexually dimorphic differences in epigenetic disruption in the brain that possibly underlie enduring effects of BPA on brain function and behavior (Kundakovic, et al., 2012). These findings, among others, add to the growing body of evidence that PAH and BPA exposures during the sensitive fetal period can have long lasting impacts on health and development. This research has been designed to be both rigorous and policy relevant, which is a direct reflection of our Center’s mission to creating a world in which every child has a healthy start.
The Community Outreach and Translation of the work of our Center is critical in disseminating our findings to the community, clinicians and policymakers. Some of our outreach highlights include the tip sheets created for our Healthy Home Healthy Child campaign covering the topics of green cleaning, stress management, and healthy eating, among others. The Center’s work has received press coverage in Time Magazine, The Wall Street Journal, and The New York Times. We also renovated our website over the grant period with a newer, more inviting design so that anyone with internet access can access our findings, tip sheets, and updates.
The Data Management Core (DMC) has developed an integrated approach to data and project management. Research data are entered into the relational database system, which automatically passes information to a project management system that is used by research assistants to anticipate and schedule upcoming visits, identify missing data, and manage contact information. The data management system has a unique feature that allows for the generation of analytic-ready statistical system files, including variable and value labeling information and missing value indicators. This system significantly enhances productivity by minimizing the latency between data collection, manuscript development, and publication of important research findings.The research is beneficial to children’s health by revealing the adverse impacts of prenatal exposure to PAH and BPA on children’s health and increasing public awareness of the problem of pollution and chemical contamination. An increased awareness of these impacts can motivate policymakers to take protective action and can inform pregnant women of measures to reduce their exposures to these chemicals and prevent negative outcomes for their children.
The Community Outreach and Translation Core’s (COTC) main methods of dissemination of research results are press releases, social media platforms, community briefings, an educational campaign (Healthy Home, Healthy Child Campaign), and presentations at conferences. All are relatively low cost and have been widely used in the environmental education field for several years.
Journal Articles: 60 Displayed | Download in RIS Format
| Other center views: | All 104 publications | 62 publications in selected types | All 60 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Albert DA, Begg MD, Andrews HF, Williams SZ, Ward A, Conicella ML, Rauh V, Thompson JL, Papapanou PN. An examination of periodontal treatment, dental care, and pregnancy outcomes in an insured population in the United States. American Journal of Public Health 2011;101(1):151-156. |
R834509 (Final) |
Exit |
|
|
Buckley JP, Engel SM, Braun JM, Whyatt RM, Daniels JL, Mendez MA, Richardson DB, Xu Y, Calafat AM, Wolff MS, Lanphear BP, Herring AH, Rundle AG. Prenatal phthalate exposures and body mass index among 4- to 7-year-old children: a pooled analysis. Epidemiology 2016;27(3):449-458. |
R834509 (Final) R834509C001 (Final) |
Exit |
|
|
Choi H, Wang L, Lin X, Spengler JD, Perera FP. Fetal window of vulnerability to airborne polycyclic aromatic hydrocarbons on proportional intrauterine growth restriction. PLoS One 2012;7(4):e35464 (11 pp.). |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) R827027 (2002) R832141 (Final) |
Exit Exit Exit |
|
|
Choi H, Perera FP. Sources of greater fetal vulnerability to airborne polycyclic aromatic hydrocarbons among African Americans. Journal of Epidemiology and Community Health 2012;66(2):121-126. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) R827027 (2002) R832141 (Final) |
Exit Exit |
|
|
Durham T, Guo J, Cowell W, Riley K, Wang S, Tang D, Perera F, Herbstman J. Prenatal PM2.5 Exposure in Relation to Maternal and Newborn Telomere Length at Delivery. Toxics 23;10(1):13. |
R834509 (Final) R827027 (2002) R832141 (Final) R836154 (Final) |
Exit Exit |
|
|
Genkinger JM, Stigter L, Jedrychowski W, Huang T-J, Wang S, Roen EL, Majewska R, Kieltyka A, Mroz E, Perera FP. Prenatal polycyclic aromatic hydrocarbon (PAH) exposure, antioxidant levels and behavioral development of children ages 6-9. Environmental Research 2015;140:136-144. |
R834509 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Herbstman JB, Tang D, Zhu D, Qu L, Sjodin A, Li Z, Camann D, Perera FP. Prenatal exposure to polycyclic aromatic hydrocarbons, benzo[a]pyrene-DNA adducts, and genomic DNA methylation in cord blood. Environmental Health Perspectives 2012;120(5):733-738. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (2012) R834509C002 (Final) |
|
|
|
Herbstman JB, Wang S, Perera FP, Lederman SA, Vishnevetsky J, Rundle AG, Hoepner LA, Qu L, Tang D. Predictors and consequences of global DNA methylation in cord blood and at three years. PLoS One 2013;8(9):e72824 (10 pp.). |
R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit |
|
|
Hoepner LA, Whyatt RM, Just AC, Calafat AM, Perera FP, Rundle AG. Urinary concentrations of bisphenol A in an urban minority birth cohort in New York City, prenatal through age 7 years. Environmental Research 2013;122:38-44. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C001 (2012) R834509C001 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Hoepner LA, Whyatt RM, Widen EM, Hassoun A, Oberfield SE, Mueller NT, Diaz D, Calafat AM, Perera FP, Rundle AG. Bisphenol A and adiposity in an inner-city birth cohort. Environmental Health Perspectives 2016;124(10):1644-1650. |
R834509 (Final) R836154 (2017) |
|
|
|
Iyer S, Perera F, Zhang B, Chanock S, Wang S, Tang D. Significant interactions between maternal PAH exposure and haplotypes in candidate genes on B[a]P-DNA adducts in a NYC cohort of non-smoking African-American and Dominican mothers and newborns. Carcinogenesis 2014;35(1):69-75. |
R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Jung KH, Bernabe K, Moors K, Yan B, Chillrud SN, Whyatt R, Camann D, Kinney PL, Perera FP, Miller RL. Effects of floor level and building type on residential levels of outdoor and indoor polycyclic aromatic hydrocarbons, black carbon, and particulate matter in New York City. Atmosphere 2011;2(2):96-109. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Jung KH, Perzanowski M, Rundle A, Moors K, Yan B, Chillrud SN, Whyatt R, Camann D, Perera FP, Miller RL. Polycyclic aromatic hydrocarbon exposure, obesity and childhood asthma in an urban cohort. Environmental Research 2014;128:35-41. |
R834509 (2014) |
Exit Exit Exit |
|
|
Jung KH, Liu B, Lovinsky-Desir S, Yan B, Camann D, Sjodin A, Li Z, Perera F, Kinney P, Chillrud S, Miller RL. Time trends of polycyclic aromatic hydrocarbon exposure in New York City from 2001 to 2012: assessed by repeat air and urine samples. Environmental Research 2014;131:95-103. |
R834509 (2014) R834509 (Final) R834509C002 (Final) R832096 (Final) |
Exit Exit Exit |
|
|
Jung K, Goowin K, Perzanowski M, Chillrud S, Perera F, Miller R, Lovinsky-Desir S. Personal Exposure to Black Carbon at School and Levels of Fractional Exhaled Nitric Oxide in New York City. Environmental Health Prespectives 2021;129(9). |
R834509 (Final) R832141 (Final) |
|
|
|
Kundakovic M, Champagne FA. Epigenetic perspective on the developmental effects of bisphenol A. Brain, Behavior, and Immunity 2011;25(6):1084-1093. |
R834509 (2011) R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit Exit Exit |
|
|
Kundakovic M, Gudsnuk K, Franks B, Madrid J, Miller RL, Perera FA. Champagne FA. Sex-specific epigenetic disruption and behavioral changes following low-dose in utero bisphenol A exposure. Proceedings of the National Academy of Sciences of the United States of America 2013;110(24):9956-9961. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C003 (Final) |
Exit Exit Exit |
|
|
Kundakovic M, Gudsnuk K, Herbstman JB, Tang D, Perera FP, Champagne FA. DNA methylation of BDNF as a biomarker of early-life adversity. Proceedings of the National Academy of Sciences of the United States of America 2015;112(22):6807-6813. |
R834509 (2014) R834509 (Final) R834509C003 (Final) |
Exit Exit Exit |
|
|
Lovasi GS, Quinn JW, Rauh VA, Perera FP, Andrews HF, Garfinkel R, Hoepner L, Whyatt R, Rundle A. Chlorpyrifos exposure and urban residential environment characteristics as determinants of early childhood neurodevelopment. American Journal of Public Health 2011;101(1):63-70. |
R834509 (2011) R834509 (Final) R827027 (2002) R832141 (Final) |
Exit Exit |
|
|
Lovasi GS, O’Neil-Dunne JPM, Lu JWT, Sheehan D, Perzanowski MS, MacFaden SW, King KL, Matte T, Miller RL, Hoepner LA, Perera FP, Rundle A. Urban tree canopy and asthma, wheeze, rhinitis, and allergic sensitization to tree pollen in a New York City birth cohort. Environmental Health Perspectives 2013;121(4):494-500. |
R834509 (Final) R827027 (2002) R832141 (Final) |
|
|
|
Lovasi GS, Eldred-Skemp N, Quinn JW, Chang HW, Rauh VA, Rundle A, Orjuela MA, Perera FP. Neighborhood social context and individual polycyclic aromatic hydrocarbon exposures associated with child cognitive test scores. Journal of Child and Family Studies 2014;23(5):785-799. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit |
|
|
Lovinsky-Desir S, Miller RL. Epigenetics, asthma, and allergic diseases:a review of latest advancements. Current Allergy and Asthma Reports 2012;12(3):211-220. |
R834509 (2012) R834509 (2013) R834509 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit |
|
|
Maresca MM, Hoepner LA, Hassoun A, Oberfield SE, Mooney SJ, Calafat AM, Ramirez J, Freyer G, Perera FP, Whyatt RM, Rundle AG. Prenatal exposure to phthalates and childhood body size in an urban cohort. Environmental Health Perspectives 2015 June 12 [Epub ahead of print], doi:10.1289/ehp.1408750. |
R834509 (2013) R834509C001 (Final) R836154 (2017) |
|
|
|
Maresca MM, Hoepner LA, Hassoun A, Oberfield SE, Mooney SJ, Calafat AM, Ramirez J, Freyer G, Perera FP, Whyatt RM, Rundle AG. Prenatal exposure to phthalates and childhood body size in an urban cohort. Environmental Health Perspectives 2016;124(4):514-520. |
R834509 (Final) |
|
|
|
Margolis AE, Herbstman JB, Davis KS, Thomas VK, Tang D, Wang Y, Wang S, Perera FP, Peterson BS, Rauh VA. Longitudinal effects of prenatal exposure to air pollutants on self-regulatory capacities and social competence. Journal of Child Psychology and Psychiatry 2016;57(7):851-860. |
R834509 (Final) R836154 (2017) |
Exit Exit |
|
|
Miller RL, Garfinkel R, Lendor C, Hoepner L, Li Z, Romanoff L, Sjodin A, Needham L, Perera FP, Whyatt RM. Polycyclic aromatic hydrocarbon metabolite levels and pediatric allergy and asthma in an inner-city cohort. Pediatric Allergy and Immunology 2010;21(2 Pt 1):260-267. |
R834509 (2011) R834509 (Final) R832096 (Final) R832141 (2007) R832141 (Final) |
Exit |
|
|
Mueller NT, Whyatt R, Hoepner L, Oberfield S, Dominguez-Bello MG, Widen EM, Hassoun A, Perera F, Rundle A. Prenatal exposure to antibiotics, cesarean section and risk of childhood obesity. International Journal of Obesity 2015;39(4):665-670. |
R834509 (2014) R834509 (Final) R834509C001 (Final) |
Exit |
|
|
Nobel KG, Fifer WP, Rauh VA, Nomura Y, Andrews HF. Academic achievement varies with gestational age among children born at term. Pediatrics 2012;130(2):e257-e264. |
R834509 (Final) |
Exit Exit |
|
|
Orjuela MA, Liu X, Warburton D, Siebert AL, Cujar C, Tang D, Jobanputra V, Perera FP. Prenatal PAH exposure is associated with chromosome-specific aberrations in cord blood. Mutation Research 2010;703(2):108-114. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Orjuela MA, Liu X, Miller RL, Warburton D, Tang D, Jobanputra V, Hoepner L, Suen IH, Diaz-Carreno S, Li Z, Sjodin A, Perera FP. Urinary naphthol metabolites and chromosomal aberrations in 5-year-old children. Cancer Epidemiology, Biomarkers & Prevention 2012;21(7):1191-1202. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) R832096 (Final) |
Exit Exit Exit |
|
|
Patel MM, Hoepner L, Garfinkel R, Chillrud S, Reyes A, Quinn JW, Perera F, Miller RL. Ambient metals, elemental carbon, and wheeze and cough in New York City children through 24 months of age. American Journal of Respiratory and Critical Care Medicine 2009;180(11):1107-1113. |
R834509 (2011) R834509 (Final) R827027 (2002) R832141 (2007) R832141 (Final) |
Exit Exit Exit |
|
|
Perera FP, Li Z, Whyatt R, Hoepner L, Wang S, Camann D, Rauh V. Prenatal airborne polycyclic aromatic hydrocarbon exposure and child IQ at age 5 years. Pediatrics 2009;124(2):e195-e202. |
R834509 (2014) R834509 (Final) R834509C002 (Final) R832141 (Final) |
Exit |
|
|
Perera FP, Wang S, Vishnevetsky J, Zhang B, Cole KJ, Tang D, Rauh V, Phillips DH. Polycyclic aromatic hydrocarbons-aromatic DNA adducts in cord blood and behavior scores in New York City children. Environmental Health Perspectives 2011;119(8):1176-1181. |
R834509 (2011) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) |
|
|
|
Perera FP, Tang D, Wang S, Vishnevetsky J, Zhang B, Diaz D, Camann D, Rauh V. Prenatal polycyclic aromatic hydrocarbon (PAH) exposure and child behavior at age 6-7 years. Environmental Health Perspectives 2012;120(6):921-926. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (2012) R834509C002 (Final) |
|
|
|
Perera FP, Chang HW, Tang D, Roen EL, Herbstman J, Margolis A, Huang TJ, Miller RL, Wang S, Rauh V. Early-life exposure to polycyclic aromatic hydrocarbons and ADHD behavior problems. PLoS One 2014;9(11):e111670 (9 pp.). |
R834509 (2014) R834509 (Final) R834509C002 (Final) R832096 (Final) |
Exit Exit |
|
|
Perera F, Herbstman J. Prenatal environmental exposures, epigenetics, and disease. Reproductive Toxicology 2011;31(3):363-373. |
R834509 (2011) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Perera F, Vishnevetsky J, Herbstman JB, Calafat AM, Xiong W, Rauh V, Wang S. Prenatal bisphenol A exposure and child behavior in an inner-city cohort. Environmental Health Perspectives 2012;120(8):1190-1194. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (2012) R834509C002 (Final) |
|
|
|
Perera F, Weiland K, Neidell M, Wang S. Prenatal exposure to airborne polycyclic aromatic hydrocarbons and IQ:estimated benefit of pollution reduction. Journal of Public Health Policy 2014;35(3):327-336. |
R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit |
|
|
Perera F, Phillips DH, Wang Y, Roen E, Herbstman J, Rauh V, Wang S, Tang D. Prenatal exposure to polycyclic aromatic hydrocarbons/aromatics, BDNF and child development. Environmental Research 2015;142:602-608. |
R834509 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Peterson BS, Rauh VA, Bansal R, Hao X, Toth Z, Nati G, Walsh K, Miller RL, Arias F, Semanek D, Perera F. Effects of prenatal exposure to air pollutants (polycyclic aromatic hydrocarbons) on the development of brain white matter, cognition, and behavior in later childhood. JAMA Psychiatry 2015;72(6):531-540. |
R834509 (Final) R834509C002 (Final) R832096 (Final) |
Exit |
|
|
Rauh VA, Horton MK, Miller RL, Whyatt RM, Perera F. Neonatology and the environment:impact of early exposure to airborne environmental toxicants on infant and child neurodevelopment. Neoreviews 2010;11(7):363-369. |
R834509 (2011) R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit Exit |
|
|
Roen EL, Wang Y, Calafat AM, Wang S, Margolis A, Herbstman J, Hoepner LA, Rauh V, Perera FP. Bisphenol A exposure and behavioral problems among inner city children at 7-9 years of age. Environmental Research 2015;142:739-745. |
R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit Exit |
|
|
Rundle AG, Gallagher D, Herbstman JB, Goldsmith J, Holmes D, Hassoun A, Oberfield S, Miller RL, Andrews H, Widen EM, Hoepner LA. Prenatal exposure to airborne polycyclic aromatic hydrocarbons and childhood growth trajectories from age 5-14 years. Environmental research 2019;177:108595. |
R834509 (Final) R827027 (2002) R832141 (Final) |
Exit Exit |
|
|
Rundle A, Hoepner L, Hassoun A, Oberfield S, Freyer G, Holmes D, Reyes M, Quinn J, Camann D, Perera F, Whyatt R. Association of childhood obesity with maternal exposure to ambient air polycyclic aromatic hydrocarbons during pregnancy. American Journal of Epidemiology 2012;175(11):1163-1172. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C001 (2012) R834509C001 (Final) |
Exit Exit Exit |
|
|
Rundle A, Rauh VA, Quinn J, Lovasi G, Transande L, Susser E, Andrews HF. Use of community-level data in the National Children’s Study to establish the representativeness of segment selection in the Queens Vanguard Site. International Journal of Health Geographics 2012;11:18 (11 pp.). |
R834509 (Final) |
Exit Exit |
|
|
Tang WY, Levin L, Talaska G, Cheung YY, Herbstman J, Tang D, Miller RL, Perera F, Ho SM. Maternal exposure to polycyclic aromatic hydrocarbons and 5'-CpG methylation of interferon-γ in cord white blood cells. Environmental Health Perspectives 2012;120(8):1195-1200. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (2012) R834509C002 (Final) |
|
|
|
Vishnevetsky J, Tang D, Chang HW, Roen EL, Wang Y, Rauh V, Wang S, Miller RL, Herbstman J, Perera FP. Combined effects of prenatal polycyclic aromatic hydrocarbons and material hardship on child IQ. Neurotoxicology and Teratology 2015;49:74-80. |
R834509 (2014) R834509 (Final) R834509C002 (Final) R832096 (Final) |
Exit Exit Exit |
|
|
Wang S, Chanock S, Tang D, Li Z, Edwards S, Jedrychowski W, Perera FP. Effect of gene-environment interactions on mental development in African American, Dominican, and Caucasian mothers and newborns. Annals of Human Genetics 2010;74(1):46-56. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) R832141 (2007) R832141 (Final) |
Exit Exit Exit |
|
|
Wang S, Yu Z, Miller RL, Tang D, Perera FP. Methods for detecting interactions between imprinted genes and environmental exposures using birth cohort designs with mother-offspring pairs. Human Heredity 2011;71(3):196-208. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit |
|
|
Wang S, Xiong W, Ma W, Chanock S, Jedrychowski W, Wu R, Perera FP. Gene-environment interactions on growth trajectories. Genetic Epidemiology 2012;36(3):206-213. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (2012) R834509C002 (Final) |
Exit Exit |
|
|
Wang Y, Perera F, Guo J, Riley K, Durham T, Ross Z, Ananth C, Baccarelli A, Wang S, Herbstman J. A methodological pipeline to generate an epigenetic marker of prenatal exposure to air pollution indicators. Epigenetics 2021;1-9. |
R834509 (Final) R827027 (2002) R832141 (Final) |
Exit |
|
|
Weiland K, Neidell M, Rauh V, Perera F. Cost of developmental delay from prenatal exposure to airborne polycyclic aromatic hydrocarbons. Journal of Health Care for the Poor and Underserved 2011;22(1):320-329. |
R834509 (2011) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) |
Exit Exit |
|
|
Widen EM, Whyatt RM, Hoepner LA, Ramirez-Carvey J, Oberfield SE, Hassoun A, Perera FP, Gallagher D, Rundle AG. Excessive gestational weight gain is associated with long-term body fat and weight retention at 7 y postpartum in African American and Dominican mothers with underweight, normal, and overweight prepregnancy BMI. American Journal of Clinical Nutrition 2015;102(6):1460-1467. |
R834509 (Final) R834509C001 (Final) |
Exit Exit |
|
|
Widen EM, Whyatt RM, Hoepner LA, Mueller NT, Ramirez-Carvey J, Oberfield SE, Hassoun A, Perera FP, Gallagher D, Rundle AG. Gestational weight gain and obesity, adiposity and body size in African-American and Dominican children in the Bronx and Northern Manhattan. Maternal and Child Nutrition 2016;12(4):918-928. |
R834509 (Final) |
Exit Exit |
|
|
Widen E, Burns N, Daniels M, Backlund G, Rickman R, Foster S, Nichols A, Hoepner L, Kinsey E, Ramireaz-Carvey J. Gestational weight change and childhood body composition trajectories from pregnancy to early adolescence. Obesity 10;. |
R834509 (Final) R827027 (2002) R832141 (Final) |
Exit Exit |
|
|
Widen E, Nichols A, Kahn L, Factor-Livak P, Insel B, Hoepner L, Dube S, Rauh V, Perera F, Rundel A. Prepregnancy obesity is associated with cognitive outcomes in boys in a low-income, multiethnic birth cohort. BMC Pediatrics 2019;19(1):507. |
R834509 (Final) R827027 (2002) |
Exit Exit |
|
|
Witherspoon NO, Trousdale K, Bearer CF, Miller RL. The public health and policy implications of epigenetics and pediatric health research. Environmental Health Perspectives 2012;120(10):a380-a381. |
R834509 (2012) R834509 (2013) R834509C003 (2012) |
|
|
|
Yan Z, Zhang H, Maher C, Arteaga-Solis E, Champagne FA, Wu L, McDonald JD, Yan B, Schwartz GJ, Miller RL. Prenatal polycyclic aromatic hydrocarbon, adiposity, peroxisome proliferator-activated receptor (PPAR) γ methylation in offspring, grand-offspring mice. PLoS One 2014;9(10):e110706 (15 pp.). |
R834509 (2014) R834509 (Final) R834509C003 (Final) |
Exit Exit |
|
|
Zeinomar N, Grant-Alfieri A, Burke K, de Hoz M, Tehranifar P, Walker D, Morton T, Shepard P, Herbstman J, Miller R, Pera F, Terry M. Cancer Risk Reduction Through Education of Adolescents:Development of a Tailored Cancer Risk-Reduction Educational Tool. Journal of Cancer Education 2021;. |
R834509 (Final) R827027 (2002) R832141 (Final) |
Exit Exit |
|
|
Widen EM, Whyatt RM, Hoepner LA, Mueller NT, Ramirez‐Carvey J, Oberfield SE, Hassoun A, Perera FP, Gallagher D, Rundle AG. Gestational weight gain and obesity, adiposity and body size in African-American and Dominican children in the Bronx and Northern Manhattan. Maternal & Child Nutrition 2016;12(4):918-928. |
R834509C001 (Final) R836154 (2017) |
Exit |
Supplemental Keywords:
environmental health, epidemiology, molecular epidemiology, PAH, BPA, prenatal, obesity, metabolic syndrome, BMI, neurodevelopment, children, New York City, NYC, community modeling, mice, community outreach, integrated pest management, IPM, indoor air qualityProgress and Final Reports:
Original Abstract Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834509C001 The Role of Endocrine Disruptors in Childhood Obesity
R834509C002 The Role of Endocrine Disruptors in Neurodevelopmental Disorders
R834509C003 The Mechanisms of Endocrine Disruptors in Laboratory Mice
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- 2014 Progress Report
- 2013 Progress Report
- 2012 Progress Report
- 2011 Progress Report
- 2010
- Original Abstract
60 journal articles for this center