Grantee Research Project Results
2006 Progress Report: Pollution-Enhanced Allergic Inflammation and Phase II Enzymes
EPA Grant Number: R831861C002Subproject: this is subproject number 002 , established and managed by the Center Director under grant R831861
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Southern California Children's Environmental Health Center
Center Director: McConnell, Rob Scot
Title: Pollution-Enhanced Allergic Inflammation and Phase II Enzymes
Investigators: Gilliland, Frank D. , Diaz-Sanchez, David
Institution: University of Southern California
EPA Project Officer: Callan, Richard
Project Period: November 1, 2003 through October 31, 2008 (Extended to October 31, 2010)
Project Period Covered by this Report: November 1, 2005 through October 31,2006
RFA: Centers for Children's Environmental Health and Disease Prevention Research (2003) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
There has been no change in the specific aims of this study, they are to study the role of Phase II enzymes in regulating responses to pollutants in: children's upper airways (Aim #1); the lower airways of healthy and asthmatic individuals (Aim #2) and in mechanistic animal and cellular models of allergic inflammation (Aim #3).
Progress Summary:
Aim #1: We will test the hypothesis that Phase II enzyme expression in the upper airways are induced by oxidant pollutants and differ between children and adults.
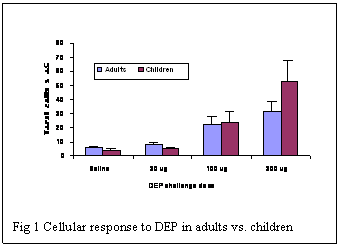
Last year we reported on phase II enzyme expression following nasal challenge with Diesel Exhaust Particles (DEP) in 10 adult subjects. In the last year we have expanded these studies and have completed similar challenges on an additional 10 adult subjects and 20 children. This was a single-blind randomized cross-over exposure study to test the expression of these enzymes in response to nasal challenge with four different DEP doses - 0 (control), 30, 100 or 300 g. Subjects performed nasal lavage sampling immediately before and 24 hours after each DEP challenge. DEP was administered by nasal spray into one nare via an atomizer. Each subject was randomized to one of 24 possible sequences of 4 DEP exposures. A four week wash-out period was observed between each DEP exposure. In adults, DEP enhanced production of IL-8, TNF- and GM-CSF in a dose-dependent fashion. Higher DEP challenge dose concentrations were associated with increased levels of chemokines as measured by ELISA. (see Fig1) In adults, DEP enhanced production of IL-8, TNF- and GM-CSF in a dose-dependent fashion. Higher DEP challenge dose concentrations were associated with increased levels of chemokines as measured by ELISA.
Fig 1. Cellular response to DEP in adults vs. children
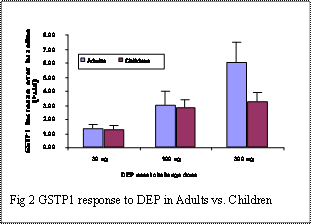
Fig 2. GSTP1 response to DEP in adults vs. children
The degree of cellular infiltration correlated with the dose of nasally administered DEP challenge. Higher concentrations of DEP elicited a larger number of total cells recovered from nasal lavage fluid obtained 24 hours after DEP exposure. As we have previously reported, we have developed real-time quantitative PCR (RT-PCR) to measure gene expression of sentinel Phase II enzymes (GSTM1, GSTP1, NQO1 and HO-1) from cells recovered from nasal washes. RNA was extracted from cells recovered from nasal lavages performed 24 hours after DEP challenge. In adults, DEP induced expression of GSTP1 in a dose-dependent fashion. With increasing concentrations of DEP challenge, there were higher levels of GSTP1 relative gene expression (Fig 2). In these adults, cellular infiltration was inversely correlated with GSTP1 enzyme gene expression. Increased levels of GSTP1 were associated with lower cell count numbers. In children, the degree of cellular infiltration also correlated with dose of nasally administered DEP challenge. Higher concentrations of DEP elicited a larger number of total cells recovered from nasal lavage fluid obtained 24 hours after DEP exposure. However, as compared to adults, this effect in children was more robust at the DEP challenge of 300 μg (see Fig 1). In these children, DEP also induced expression of GSTP1. At DEP concentrations of 30 and 100 μg, there were higher levels of GSTP1 relative gene expression. However, GSTP1 relative gene expression for the DEP nasal challenge of 300 μg was decreased in children as compared to adults. Similar results are seen with the other 3 Phase II enzymes measured.
Aim #2: We will test the hypothesis that Phase II enzyme expression in the lower airways are induced by oxidant pollutants and differ between asthmatic and non-asthmatic subjects.
In the past year we have exposed 10 subjects (5 asthmatic and 5 non-asthmatic) to diesel exhaust to study the effect of Phase II expression on lower airway responses. To date we have thus performed exposures on 10 healthy and 15 asthmatic subjects. We have refined our exposure system and can reproducibly produce a diesel particulate exposure level that is within 8% of the target level every time. These diesel particles are chemically and physically identical to diesel particles encountered in ambient air. Diesel exhaust resulted in increased expression of all four, phase II enzymes tested in cells recovered from sputum induced 24 hours after exposure. This increase was observed in both healthy and asthmatic subjects. There was wide heterogeneity in responses to diesel exhaust between subjects. While no enhanced inflammation was observed in any of the healthy subjects, four out of the fifteen asthmatic subjects demonstrated a robust inflammatory response to diesel exhaust but not filtered air. Phase II enzyme expression was measured in cells recovered from induced sputum performed 24 h after exposure. For all subjects measured to date we could observe an increase in phase II enzyme expression. Again there was wide heterogeneity in responses between subjects. This heterogeneity makes meaningful statistical analysis between asthmatic and healthy subjects premature.
Aim #3: We will determine the role of Phase II enzymes in regulating the adjuvant effects of particulate pollutants.
We have previously reported that treatment with sulforaphane (a phase II enzyme inducer) can prevent the production of pro-inflammatory cytokines in respiratory epithelial cells in vitro and IgE production from PBMCs. We have now explored this mechanism. We confirmed that DEPs and its extracts (DEPX) can act directly on B lymphocytes and showed that DEPX could enhance the IgH germline-transcription both in a B cell line and PBMC. DEPX increased NQO1 mRNA gene expression in both peripheral blood lymphocytes and the B lymphocyte cell line DG75 in a dose-dependent manner. NQO1 protein induction by DEPX in DG75 cells was confirmed by Western blot. A reporter gene assay showed that DEP could induce activity of the anti-oxidant response element (ARE) located in the NQO1 gene promoter. Induction of both NQO1 mRNA and protein expression could be blocked by co-culture with an anti-oxidant and partly repressed by inhibitors of PI3-K and p38 mitogen-activated protein kinase, but not by inhibitors of mitogen-activated protein kinase/extracellular signal-regulated kinase kinase (Mek/ERK) or protein kinase C (PKC).
The principal findings of our results is the discovery that children have enhanced inflammatory responses to the model pollutant DEP and that this seems to be related to their reduced capacity to make a cytoprotective Phase II enzymes response. Our in vitro data supports the idea that DEP suggest that phase II enzyme expression through activation of the ARE. Our studies illuminate why there may be increased susceptibility of certain vulnerable individuals and populations (such as children) to oxidant pollutants and suggest that increasing the body’s Phase II responses either by therapeutic or dietary means may counteract this effect.
Future Activities:
In the next year we intend to continue recruitment of adults and children for Aims #1 and #2 and use our in vitro system to examine the role of individual Phase II enzymes.
Journal Articles on this Report : 5 Displayed | Download in RIS Format
| Other subproject views: | All 138 publications | 90 publications in selected types | All 89 journal articles |
|---|---|---|---|
| Other center views: | All 202 publications | 132 publications in selected types | All 131 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Diaz-Sanchez D, Riedl M. Diesel effects on human health: a question of stress? American Journal of Physiology—Lung Cellular and Molecular Physiology 2005;289(5):L722-L723. |
R831861 (2004) R831861 (2005) R831861 (2006) R831861 (Final) R831861C001 (Final) R831861C002 (2006) R831861C002 (Final) R831861C003 (Final) R826708 (2000) R826708 (2001) R826708 (2002) R826708 (Final) |
Exit Exit Exit |
|
|
Diaz-Sanchez D, Rumold R, Gong Jr. H. Challenge with environmental tobacco smoke exacerbates allergic airway disease in human beings. Journal of Allergy and Clinical Immunology 2006;118(2):441-446. |
R831861 (2005) R831861 (2006) R831861 (Final) R831861C001 (Final) R831861C002 (2006) R831861C002 (2007) R831861C002 (Final) R831861C003 (Final) |
Exit Exit Exit |
|
|
Gilliland FD, Li YF, Gong Jr. H, Diaz-Sanchez D. Glutathione s-transferases M1 and P1 prevent aggravation of allergic responses by secondhand smoke. American Journal of Respiratory and Critical Care Medicine 2006;174(12):1335-1341. |
R831861 (2006) R831861 (Final) R831861C001 (Final) R831861C002 (2006) R831861C002 (2007) R831861C002 (Final) R831861C003 (2007) R831861C003 (Final) |
Exit Exit Exit |
|
|
Ritz SA, Wan J, Diaz-Sanchez D. Sulforaphane-stimulated phase II enzyme induction inhibits cytokine production by airway epithelial cells stimulated with diesel extract. American Journal of Physiology-Lung Cellular and Molecular Physiology 2007;292(1):L33-L39. |
R831861 (2006) R831861 (Final) R831861C001 (Final) R831861C002 (2006) R831861C002 (2007) R831861C002 (Final) R831861C003 (Final) |
Exit Exit Exit |
|
|
Wan J, Diaz-Sanchez D. Phase II enzymes induction blocks the enhanced IgE production in B cells by diesel exhaust particles. Journal of Immunology 2006;177(5):3477-3483. |
R831861 (2005) R831861 (2006) R831861 (Final) R831861C001 (Final) R831861C002 (2006) R831861C002 (2007) R831861C002 (Final) R831861C003 (Final) R826708 (2000) R826708 (2001) R826708 (2002) R826708 (Final) |
Exit Exit |
Supplemental Keywords:
RFA, Health, Scientific Discipline, ENVIRONMENTAL MANAGEMENT, HUMAN HEALTH, Health Risk Assessment, Children's Health, Biochemistry, Health Effects, Risk Assessment, childhood respiratory disease, Human Health Risk Assessment, allergen, asthma, susceptibility, animal model, community-based intervention, airway disease, children's environmental health, outreach and educationProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R831861 Southern California Children's Environmental Health Center Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R831861C001 Urban Air Pollution and Persistent Early Life Asthma
R831861C002 Pollution-Enhanced Allergic Inflammation and Phase II Enzymes
R831861C003 Air Pollution, Exhaled Breath Markers, and Asthma in Susceptible Children
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2009
- 2008
- 2007 Progress Report
- 2005 Progress Report
- 2004 Progress Report
- Original Abstract
89 journal articles for this subproject
Main Center: R831861
202 publications for this center
131 journal articles for this center