Grantee Research Project Results
2009 Progress Report: Epidemiological Studies on Extra Pulmonary Effects of Fresh and Aged Urban Aerosols from Different Sources
EPA Grant Number: R832415C002Subproject: this is subproject number 002 , established and managed by the Center Director under grant R832415
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Rochester PM Center
Center Director: Oberdörster, Günter
Title: Epidemiological Studies on Extra Pulmonary Effects of Fresh and Aged Urban Aerosols from Different Sources
Investigators: Peters, Annette , Zareba, Wojciech , Utell, Mark J. , Henneberger, Alexandra , Wichmann, Heinz-Erich , Stoelzel, M , Rückerl, Regina , Phipps, Richard , Breitner, Susanne
Current Investigators: Peters, Annette , Utell, Mark J. , Zareba, Wojciech , Phipps, Richard , Wichmann, Heinz-Erich , Henneberger, Alexandra , Breitner, Susanne , Stoelzel, M , Rückerl, Regina
Institution: GSF-National Research Center for Environment and Health , University of Rochester
EPA Project Officer: Chung, Serena
Project Period: October 1, 2005 through September 30, 2010 (Extended to September 30, 2012)
Project Period Covered by this Report: July 1, 2008 through June 30,2009
RFA: Particulate Matter Research Centers (2004) RFA Text | Recipients Lists
Research Category: Human Health , Air
Objective:
The objective of the epidemiological study is to examine the effect of fine and ultrafine particles on systemic responses, endothelial and cardiac function. The study was conducted in Augsburg, Germany, between March 19, 2007, and December 17, 2008.
The specific aims of the study are to:
Aim #1: Determine the effect of ambient fine and ultrafine particles on an acute phase reaction in the blood of subjects with type 2 diabetes mellitus (T2DM), impaired glucose tolerance (IGT), and potential genetic susceptibility (gen. susc.).
Aim #2: Determine the effect of ambient fine and ultrafine particles on pro-thrombotic states of the blood in the above subject panels.
Aim #3: Determine the effect of ambient fine and ultrafine particles on endothelial dysfunction as a key element of coronary vulnerability in a subset of the above subject panels.
Aim #4: Determine the effect of ultrafine particles on cardiac function as characterized by ECG measures of autonomic function and repolarization in a subset of the above subject panels.
The objectives have not been altered during this reporting period.
Progress Summary:
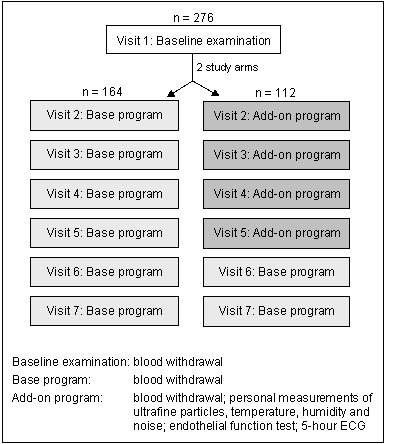
Figure 1 shows an overview of the study design. We examined 276 participants consisting of 1) 84 participants with T2DM, 2) 104 participants with IGT and 3) 88 participants with potential gen. susc. All individuals were invited to participate in seven repeated examinations, scheduled every 4-6 weeks on the same weekday and at the same time of the day. At each visit (base program) a blood withdrawal was conducted. Air pollution was measured at a central measurement site in Augsburg throughout the complete study period. In addition (add-on program), for a subgroup of 112 participants personal measurements of ultrafine particles using a portable condensation particle counter (CPC) as well as temperature, humidity and noise were conducted in up to four visits. Furthermore, endothelial function as a key element of coronary vulnerability as well as cardiac function characterized by ECG measures (including a resting ECG for 20 minutes) were assessed in this subset.
Figure 1. Study design.
Exclusions and drop outs, validity of examinations
Figure 2 shows the recruitment and progress status of the study. Nine participants were excluded after the first visit, five of them because of rheumatoid arthritis, three because of arthropathic psoriasis, polyarthritis and Crohn’s disease, respectively, and one individual turned out to be a regular smoker. Six persons dropped out of the study after the baseline examination. 276 participants were included into the study and 1799 visits were conducted. We considered only those participants for analysis who had at least either two blood withdrawals or one ECG-measurement or two endothelial function (ED) measurements. Therefore, we excluded one participant with two visits since he did not meet this criterion. Further 17 visits, which were part of the base program, were not considered due to invalid blood draws. In total, we included 275 participants and 1780 visits in the analysis. 163 of 275 subjects (59.3%) with 1051 valid visits (59.0%) exclusively participated in the base program.222 participants (80.7%) have completed at least 7 visits.
112 volunteers (40.7%), who participated in the add-on program, completed 729 valid visits (41.0%) including 385 visits with personal measurements and 344 visits only with blood withdrawal.
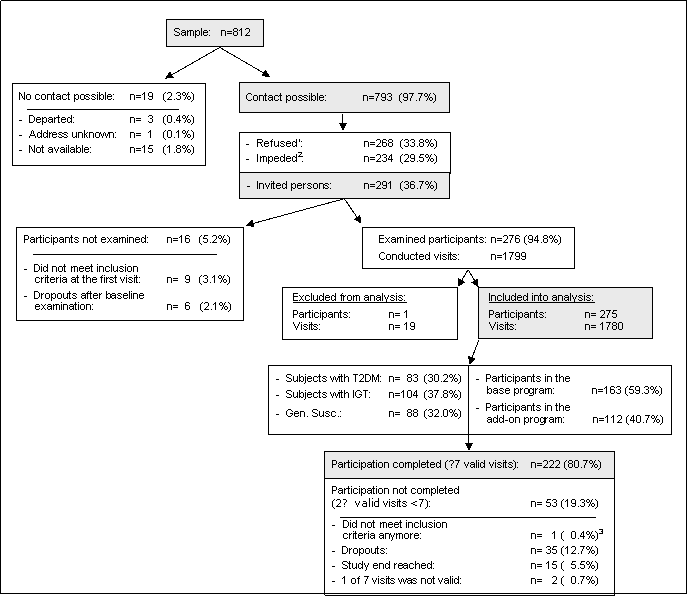
Figure 2. Flow diagram of recruitment and accomplishment.
1Refused: persons refused participation because of their negative attitude to research in general.
2Impeded: participation was not possible because of illness or having not enough time, but person had a positive attitude to research.
3One participant had a myocardial infarction between the fifth and sixth visit.
Table 1 shows the different sample sizes for the three outcomes (Blood, ECG, ED).
- Out of 1799 visits 23 blood draws (1.3%) could not be conducted due to an acute inflammatory disease of the participants. These 23 blood draws include the 17 excluded visits mentioned above. For the remaining six visits without blood withdrawal the ECG or ED measurement is available. One blood sample was missing.
- Out of 385 intended ECG measurements, three (0.8%) could not be conducted because no empty flash card was available at the time of the visit. In 16 of 382 conducted measurements (4.2%) the flash card remained empty or was unreadable. Incorrect data were recorded for three ECG measurements (0.8%) so that 363 of 382 ECG recordings (95.0%) were valid. In order to analyse A) the 5min or 1h segments of the ECG parameters at least one ECG recording has to be available per participant. For the analysis of B) the 20 min. resting ECG in supine position and the average of the complete long-term ECG, at least two ECG measurements are required per patient for the statistical analysis. Nine participants ended up with only one valid ECG recording each and thus, were excluded for part B) of the analysis.
- Out of 385 intended ED measurements, 371 were carried out (96.4%). Three ED measurements could not be conducted because of technical problems and five participants refused the measurement in eleven visits. Out of the 371 conducted measurements, nine ED recordings (2.4%) had to be excluded because of inadequate quality. Additionally, eight participants (2.2%) with only one ED measurement were excluded as at least two ED measurements per participant are required for data analysis. Thus, 354 of 371 ED measurements (95.4%) of 100 participants are available.
For 327 of 385 visits (84.9%), all three outcome measures were valid for analysis.
Table 1. Numbers of blood draws, ECG and ED measurements
| <> |
Not conducted |
|
||||
|
|
In total |
missing/ |
excluded (not valid for analysis) |
available |
||
| <>Blood |
|
23 |
1776 |
1 |
1a |
1774 |
|
ECG
|
<> |
3
|
382
|
19
|
|
|
|
resting ECG / overall mean |
9b
|
355
|
||||
| <>ED |
|
14 |
371 |
9 |
8c |
354 |
aFor one participant only 1 blood withdrawal was available. bFor 9 participants only 1 ECG-measurement was available. cFor 8 participants only 1 ED-measurement was available.
Participant characteristics
The baseline characteristics of the study population are shown in Table 2.
Table 2. Baseline characteristics of the study population
|
Characteristic |
All (N=275) |
T2DM |
IGT |
Gen. susc.
(N=88)
|
p-value for heterogeneity between panels |
|
Male (%) |
157 (57.1)
|
49 (59.0)
|
59 (56.7)
|
49 (55.7)
|
0.93c |
|
Age [yrs]a |
63.5
(32.8-82.3)
|
68.0 (50.0-80.2) |
66.1 (44.3-82.3) |
56.4 (32.8-79.1) |
<.0001d |
|
BMI [kg/m2]a |
29.3
(19.9-56.0)
|
31.6 b (22.4-43.3) |
29.6 (20.0-56.0) |
26.9 (19.9-46.7) |
<.0001d |
a mean (range); b for one participant the value for the bmi is missing; c chi-square test; d ANOVA
Genetic information
Table 3 shows the frequencies of selected SNPs (CRP rs1205; Fibrinogen rs1800790; GSTM1) within the study population and within the KORA-Survey 2000. The study population was recruited out of the KORA-Survey 2000 and the KORA F4 (Follow up of KORA-Survey 2000). Table 4 shows the frequencies of selected SNPs (CRP rs1205; Fibrinogen rs1800790; GSTM1) within the study population. Study subjects with potential genetic susceptibility were recruited to have GSTM1 00 and (rs1205 CC or rs 1800790 AA/AG).
Table 3. Frequencies of selected SNPs (CRP rs1205; Fibrinogen rs1800790; GSTM1) within the study population (Study pop) and within the KORA-Survey 2000 (KORA Survey).
|
rs1205 |
rs1800790 |
GSTM1 |
|||||||
|
n % |
Study pop
|
KORA Survey
|
n % |
Study pop
|
KORA Survey
|
n % |
Study pop
|
KORA Survey
|
|
|
CC
|
129
46.9
|
1698
43.6
|
AA
|
18
6.6
|
192
4.7
|
00
|
161
58.6
|
1755
42.5
|
|
|
CT
|
98
35.6
|
1757
45.1
|
AG
|
104
37.8
|
1323
32.1
|
01 or 11
|
92
33.5
|
1999
48.4
|
|
|
TT
|
26
9.5
|
442
11.3
|
GG
|
139
50.6
|
2356
57.1
|
|
|
|
|
|
Missing
|
22
8.0
|
230
5.6
|
Missing
|
14
5.1
|
256
6.2
|
Missing
|
22
8.0
|
373
9.0
|
|
|
Total
|
275
|
4127
|
Total
|
275
|
4127
|
Total
|
275
|
4127
|
|
Table 4. Frequencies of selected SNPs (CRP rs1205; Fibrinogen rs1800790; GSTM1) within the study population.
|
rs1205 |
|
Total |
|||||||||||||||||||||||
|
n |
T2DM
|
IGT |
Gen.susc. |
Future Activities:For the upcoming year, the following activities are planned:
Journal Articles on this Report : 2 Displayed | Download in RIS Format
Supplemental Keywords:Health, RFA, Scientific Discipline, Air, PHYSICAL ASPECTS, Health Risk Assessment, Physical Processes, Risk Assessments, particulate matter, Epidemiology, human exposure, long term exposure, aersol particles, atmospheric particles, exposure, PM, ambient particle health effects, atmospheric aerosol particles, atmospheric particulate matter, cardiovascular disease, human health risk, acute cardiovascular effectsProgress and Final Reports:Original AbstractMain Center Abstract and Reports:R832415 Rochester PM Center Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center). The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency. Project Research Results
18 journal articles for this subproject Main Center: R832415 191 publications for this center 144 journal articles for this center Site NavigationRelated InformationContact Us to ask a question, provide feedback, or report a problem.
Last updated April 28, 2023
| |||||||||||||||||||||