Grantee Research Project Results
Final Report: The Mechanisms of Endocrine Disruptors in Laboratory Mice
EPA Grant Number: R834509C003Subproject: this is subproject number 003 , established and managed by the Center Director under grant R834509
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Water Environment and Reuse Foundation's National Center for Resource Recovery and Nutrient Management
Center Director: Olabode, Lola
Title: The Mechanisms of Endocrine Disruptors in Laboratory Mice
Investigators: Champagne, Frances , Miller, Rachel L.
Institution: Columbia University in the City of New York
EPA Project Officer: Callan, Richard
Project Period: September 24, 2009 through September 23, 2014 (Extended to September 23, 2015)
RFA: Children's Environmental Health and Disease Prevention Research Centers (with NIEHS) (2009) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
The specific aims of the project were to:
- Examine the consequence of prenatal oral bisphenol A (BPA) exposure on neurobehavioral, obesity and immune dysfunction in Balb/c mice by determining whether prenatal BPA exposure is associated with abnormal brain cytoarchitecture, impaired social, anxiety-like and cognitive performance, greater adult body weight, body fat composition and organ fat and immune dysfunction in the adult offspring or grand-offspring.
- Examine the consequence of prenatal oral BPA exposure for tissue-specific molecular modifications in mice by determining prenatal BPA exposure-induced changes in DNA methylation in genes sensitive to endocrine disruption and immune dysregulation in the brain (hippocampus, hypothalamus, cortex), adipocytes and blood of the prenatally BPA exposed offspring and grand-offspring at gestation day 19 and adulthood (PND60). Determine corresponding changes in gene expression, neurobehavioral, obesity and immunologic outcomes, and whether changes in DNA methylation and gene expression of genes critical for adipogenesis persist in blood of F1 male offspring from PND 28 to adulthood.
- Examine the consequence of prenatal inhaled polycyclic aromatic hydrocarbon (PAH) exposure at current levels determined to exist in New York City's Northern Manhattan/South Bronx on neurodevelopment and obesity in Balb/c mice by determining whether prenatal PAH exposure is associated with abnormal brain cytoarchitecture, impaired anxiety-like and cognitive performance, greater body weight through weaning to adulthood, body fat composition and organ fat content in adult offspring and grand offspring.
- Examine the consequence of prenatal inhaled PAH exposure for tissue specific molecular modifications (DNA methylation) in mice of gene sensitive to endocrine disruption and immune dysregulation in the brain, adipocytes and blood of the prenatally exposed offspring and grand-offspring at gestation day 19 and PND60. Determine corresponding changes in gene expression, neurobehavioral, obesity and immunologic outcomes, and whether changes in DNA methylation and gene expression of genes critical for adipogenesis persist in blood of F1 male offspring from PND 28 to adulthood.
Summary/Accomplishments (Outputs/Outcomes):
Aims 1 & 2
Studies were conducted by personnel trained in rodent phenotyping and behavioral neuroscience (Kathryn Gudsnuk) and in molecular epigenetics (Dr. Maria Kundakovic). The oral daily administration of BPA in mice was an effective strategy in achieving dose-dependent variation in both behavioral and molecular outcomes in mice and is viewed as a more optimal strategy for daily gestational exposure than more stress-inducing procedures such as gavage and injection. Urine levels confirmed the dose-dependent increases in BPA exposure, corresponding to a vehicle, 2 µg/kg, 20 µg/kg, and 200 µg/kg dosing regime. Initial analyses focused on behavioral and brain gene expression changes with follow-up analyses of DNA methylation in identified target genes. Due to the inclusion of multiple doses and both male and female offspring, our analyses were able to reveal a dose-dependent sex by BPA interaction on hypothalamic expression of estrogen receptor isoforms (alpha and beta). This same pattern of effect was observed in the expression of DNA methyltransferases. The curvilinear dosage effects were evident in the hypothalamus and cortex but less evident in other brain structures, such as the hippocampus. Epigenetic changes in the estrogen receptor alpha (Esr1) gene promoter revealed that BPA-induced changes in DNA methylation in the brain varied by sex and by brain region. Behavioral effects of prenatal BPA included increased social investigation and reduced aggression at the low dose exposure, with increased aggression at the 200 µg/kg dose in both males and females. This work was published in Proceedings of the National Academy of Sciences in 2013 (PNAS 2013;110(24):9956-9961). This paper also included findings that there are BPA-induced changes in the maternal behavior of treated dams, and our analyses have confirmed that these postnatal influences may impact specific behavioral and epigenetic outcomes. We currently are working with human developmental psychologists and environmental health researchers to determine whether these BPA-induced effects on mother-infant interactions are relevant in human studies.
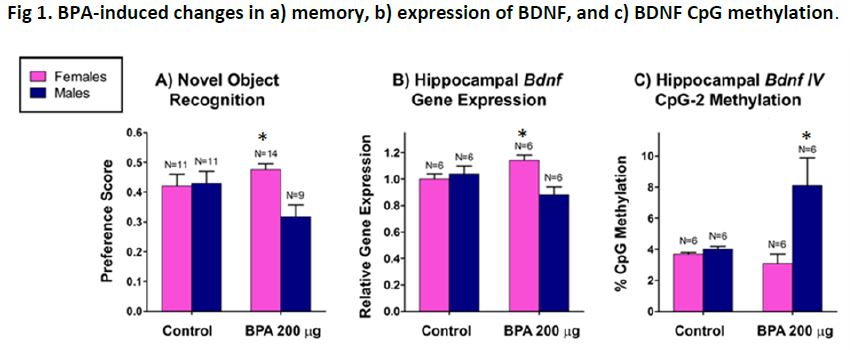
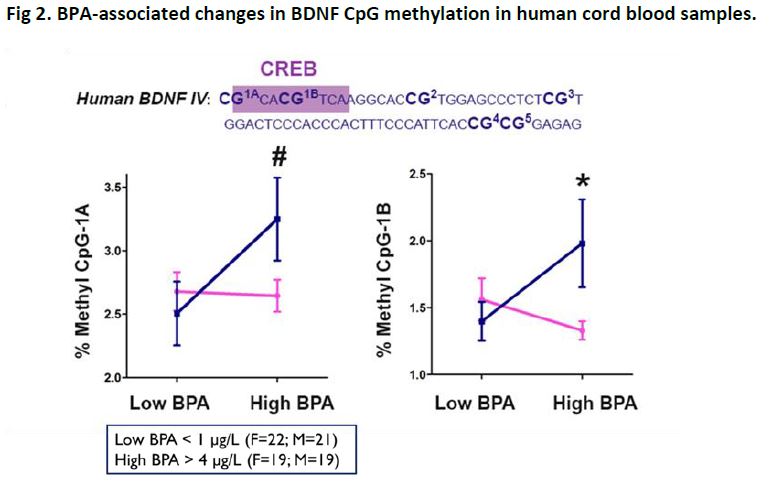
Consistent with human studies of prenatal BPA-induced effects, on some measures we observe more severe behavioral impairments in male offspring in mice. Working memory was found to be impaired in male offspring that had received 200 µg/kg BPA, with no impairments observed in female offspring. Consistent with this behavioral finding, we determined reductions in hippocampal expression of brain derived neurotrophic factor (BDNF)—a critical mechanism regulating working memory—in male offspring. Epigenetic analyses revealed that in the hippocampus, male offspring exposed to prenatal BPA had increased DNA methylation of the BDNF gene promoter (Figure 1). These epigenetic changes also were observed in blood samples—indicating that peripheral DNA can be used to predict epigenetic changes in the brain. Analyses of human cord blood samples from the human cohort revealed that the BPA-associated epigenetic effects observed in mice was likewise observed in humans (Figure 2), suggesting a possible biomarker of exposure and neurobehavioral outcomes. This work was recently published in Proceedings of the National Academy of Sciences (PNAS 2014;112(22):6807-6813.).
Analyses of growth trajectories indicated significant weight gains in males exposed to 2 µg/kg BPA (compared to vehicle)—an effect evident at weaning that persisted until adulthood. However, we did not observe BPA-induced changes in body fat weights. We currently are exploring the expression of genes within the placenta that regulate growth and neurodevelopment to determine how these are impacted by prenatal BPA. These analyses may lead to additional studies in which human placental samples can be analyzed.
Aims 3 & 4
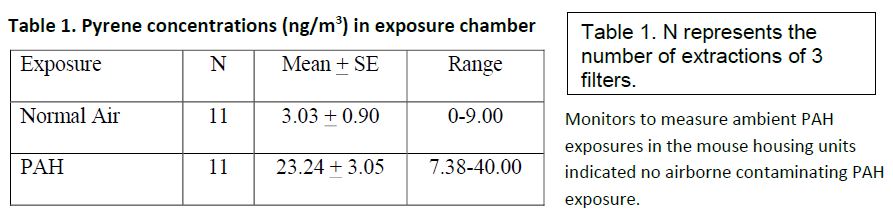
The components of the PAH aerosol have been published by us previously (Chu, 2012). Measured levels of the particulate fraction of PAH collected from the filters in the PAH mouse exposure chamber suggested good exposure of mice during experimental, but not during negative control, delivery conditions (Table 1).
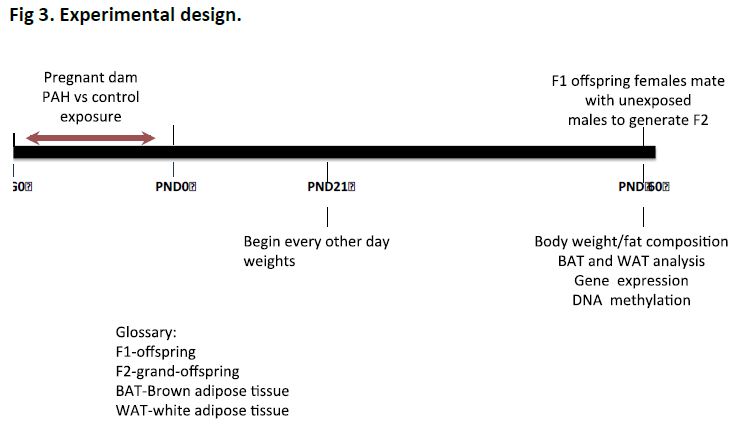
Female BALB/c mice shortly after mating were exposed to PAH versus nebulized negative control solution alone in the PAH-chamber unit 5 hours a day, 5 days a week, over several rounds of exposure as shown below (Fig 3).
Mice were weighed every other day. In addition, epigenetic and clinical (i.e., open field, light dark box) neurological outcomes were assessed at PND 60.
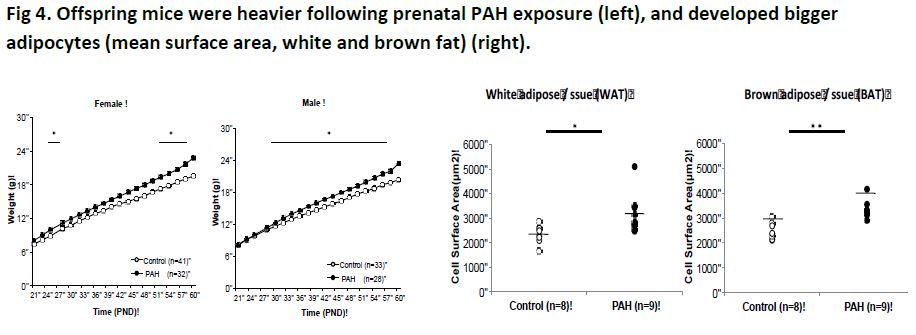
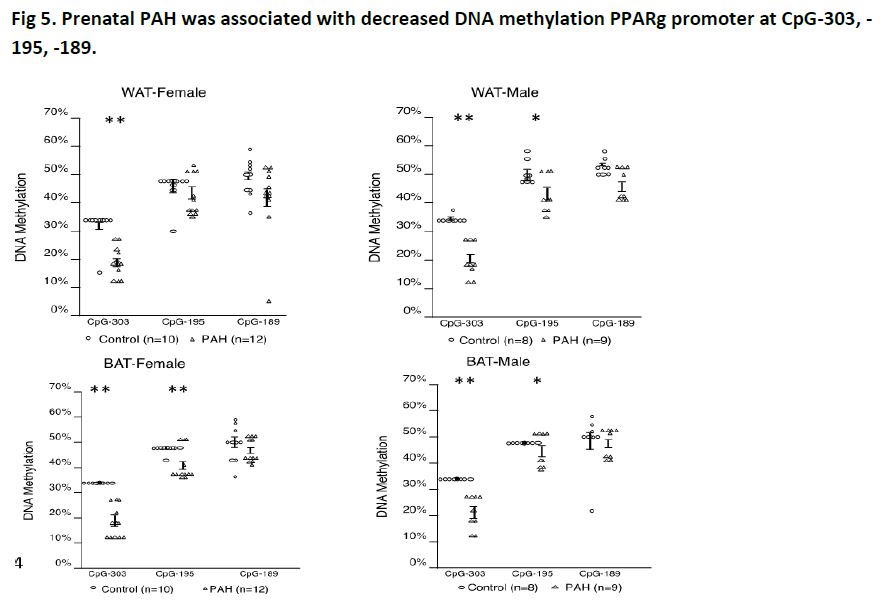
We found that offspring of dams exposed to greater PAH during gestation had increased weight (Fig 4, left), fat mass (Fig 4, right), as well as higher gene expression of PPARg, C/EBPa, Cox2, FAS and adiponectin and lower DNA methylation of PPARg (Fig 5).
Of note, PPARg promoter adipose tissue methylation and expression negatively correlated in offspring, consistent with methylation induced gene silencing. Moreover, the average methylation level of three CpG sites in PPARg promoter correlated negatively with body weight and body mass index at PND60 (r = -0.43 and -0.62, 95% CI -0.68, -0.17 and 95% CI -0.78, -0.38, p = 0.003 and p=0.00003, respectively, n = 38) and cell surface area at PND60 (r = -0.61, 95% CI-0.84, -0.18, p = 0.01, n = 17). Similar differences in phenotype, DNA methylation and gene expression extended through the grand-offspring mice.
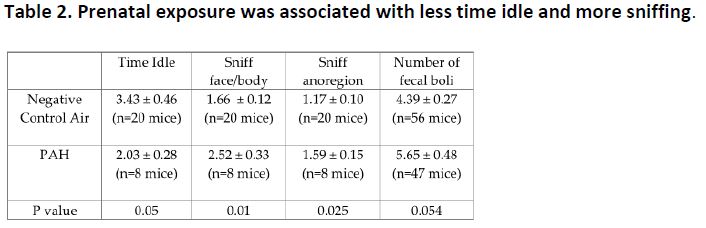
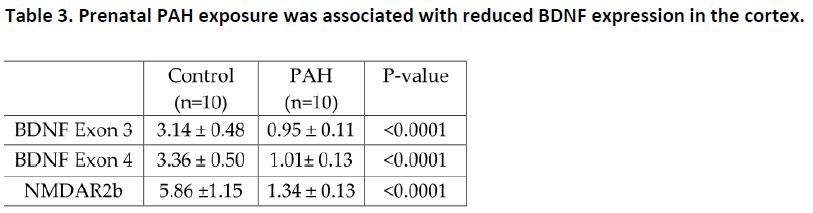
Further, prenatal PAH exposure was associated with more activity, and anxiety-like behaviors (Table 2), and decreased relative expression of BDNF and NRDAR2b in the mouse cerebral cortex (Table 3). High methylation of BDNF at exon 4 was found as well.
Our findings suggest that prenatal exposure to BPA or PAH can have a lasting impact on tissue-specific gene expression patterns and that this effect is associated with epigenetic changes within the regulatory regions of target genes. We believe that these molecular alterations mediate the behavioral, neurodevelopmental and metabolic effects associated with BPA or PAH exposure. In the case of BPA, we have determined that there are non-linear dosage effects of BPA on outcomes—an important challenge to the “increased dose–increased disruption” assumption in toxicology and environmental health research. We also have determined that social experiences (mother-infant interactions) may moderate or mediate the effects of BPA. This finding has implications for future studies in humans and intervention strategies that focus on the interplay between toxin exposure and the social environment. In addition, we have determined that human epigenetic biomarkers can be used to detect the impact of BPA and PAH exposure. Future studies can use these epigenetic biomarkers to determine whether long-term neurobehavioral outcomes associated with BPA and PAH exposure in humans is predicted by this epigenetic change.
Journal Articles on this Report : 8 Displayed | Download in RIS Format
| Other subproject views: | All 21 publications | 9 publications in selected types | All 7 journal articles |
|---|---|---|---|
| Other center views: | All 104 publications | 62 publications in selected types | All 60 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Lovinsky-Desir S, Miller RL. Environmental epigenetics, asthma, and allergy: our environment's molecular footprints. In: Sahu SC, ed. Toxicology and Epigenetics. Chichester, UK: John Wiley & Sons, Ltd., 2012, Chapter 9, pp. 191-203. |
R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit |
|
|
Miller RL. Asthma epigenetics: emergence of a new paradigm? In: Michels KB, ed. Epigenetic Epidemiology. New York: Springer Publishing, 2012, Chapter 16, pp. 307-320. |
R834509 (2011) R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit |
|
|
Kundakovic M, Champagne FA. Epigenetic perspective on the developmental effects of bisphenol A. Brain, Behavior, and Immunity 2011;25(6):1084-1093. |
R834509 (2011) R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit Exit Exit |
|
|
Kundakovic M, Gudsnuk K, Franks B, Madrid J, Miller RL, Perera FA. Champagne FA. Sex-specific epigenetic disruption and behavioral changes following low-dose in utero bisphenol A exposure. Proceedings of the National Academy of Sciences of the United States of America 2013;110(24):9956-9961. |
R834509 (2013) R834509 (2014) R834509 (Final) R834509C003 (Final) |
Exit Exit Exit |
|
|
Kundakovic M, Gudsnuk K, Herbstman JB, Tang D, Perera FP, Champagne FA. DNA methylation of BDNF as a biomarker of early-life adversity. Proceedings of the National Academy of Sciences of the United States of America 2015;112(22):6807-6813. |
R834509 (2014) R834509 (Final) R834509C003 (Final) |
Exit Exit Exit |
|
|
Lovinsky-Desir S, Miller RL. Epigenetics, asthma, and allergic diseases:a review of latest advancements. Current Allergy and Asthma Reports 2012;12(3):211-220. |
R834509 (2012) R834509 (2013) R834509 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit |
|
|
Rauh VA, Horton MK, Miller RL, Whyatt RM, Perera F. Neonatology and the environment:impact of early exposure to airborne environmental toxicants on infant and child neurodevelopment. Neoreviews 2010;11(7):363-369. |
R834509 (2011) R834509 (2012) R834509 (2013) R834509 (2014) R834509 (Final) R834509C002 (Final) R834509C003 (2012) R834509C003 (Final) |
Exit Exit |
|
|
Yan Z, Zhang H, Maher C, Arteaga-Solis E, Champagne FA, Wu L, McDonald JD, Yan B, Schwartz GJ, Miller RL. Prenatal polycyclic aromatic hydrocarbon, adiposity, peroxisome proliferator-activated receptor (PPAR) γ methylation in offspring, grand-offspring mice. PLoS One 2014;9(10):e110706 (15 pp.). |
R834509 (2014) R834509 (Final) R834509C003 (Final) |
Exit Exit |
Supplemental Keywords:
PAH, BPA, mice, obesity, neurodevelopment;, RFA, Health, Scientific Discipline, INTERNATIONAL COOPERATION, ENVIRONMENTAL MANAGEMENT, POLLUTANTS/TOXICS, Chemicals, Biochemistry, Children's Health, Environmental Policy, Biology, Risk Assessment, air toxics, developmental neurotoxicity, air pollution, childhood obesity, endocrine disruptors, assessment of exposure, children's vulnerablity, children's environmental health, growth & developmentRelevant Websites:
Columbia Center for Children’s Environmental Health ExitProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R834509 Water Environment and Reuse Foundation's National Center for Resource Recovery and Nutrient Management Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834509C001 The Role of Endocrine Disruptors in Childhood Obesity
R834509C002 The Role of Endocrine Disruptors in Neurodevelopmental Disorders
R834509C003 The Mechanisms of Endocrine Disruptors in Laboratory Mice
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
7 journal articles for this subproject
Main Center: R834509
104 publications for this center
60 journal articles for this center