Grantee Research Project Results
2012 Progress Report: Risk-Based Decision Making for Assessing Potential Impacts of Geologic CO2 Sequestration on Drinking-Water Sources
EPA Grant Number: R834387Title: Risk-Based Decision Making for Assessing Potential Impacts of Geologic CO2 Sequestration on Drinking-Water Sources
Investigators: McCray, John , Sitchler, Alexis , Kaszuba, John , Maxwell, Reed , Marcon, Virginia
Current Investigators: McCray, John , Sitchler, Alexis , Maxwell, Reed
Institution: Colorado School of Mines , University of Wyoming
Current Institution: Colorado School of Mines
EPA Project Officer: Aja, Hayley
Project Period: February 1, 2010 through January 31, 2013 (Extended to January 31, 2014)
Project Period Covered by this Report: February 1, 2012 through January 31,2013
Project Amount: $899,987
RFA: Integrated Design, Modeling, and Monitoring of Geologic Sequestration of Anthropogenic Carbon Dioxide to Safeguard Sources of Drinking Water (2009) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
The total risk associated with CO2 leakage from deep geologic formations can be broken into two categories: 1) the risk of leakage through the caprock and 2) the risk of decreased aquifer water quality upon introduction of CO2. Methods used in caprock assessment for oil and gas reservoirs currently are being adapted for sequestration purposes by other researchers. The focus of this research is to develop a methodology to quantify the susceptibility of an aquifer to contamination from CO2 leakage, and to assess the risk of decreased aquifer quality. Research to rigorously quantify risk of aquifer contamination due to CO2 leakage from deeper sequestration targets has not been previously published.
Progress Summary:
Literature Review
Current potential CO2 sequestration sites
A review of current potential CO2 sequestration sites was performed in previous reporting periods.
Current research on leakage of CO2 from sequestration sites
The study on impacts of brine leakage on shallow aquifer quality was accepted for publication in the journal Ground Water.Geochemical modeling and experiments
Geochemical Modeling and Experiments
Intermediate-scale experiments on carbonate rocks
Laboratory experiments of clay-rich carbonate rocks under elevated CO2 partial-pressures were performed to assess the relative impact of carbonate dissolution and metal desorption on metal release from carbonate aquifers impacted by CO2. The rock samples were reacted in acrylic beakers as the partial pressure of CO2 was increased in stages, from 0 (pure N2) through 1% (99% N2), 10% (90% N2) and 100% CO2. Aqueous samples were drawn from the beakers at increasing time intervals, and pH was measured in situ.
Clay minerals appeared to influence the metal concentration in solution more through metal adsorption after release from carbonate dissolution than as a source through desorption. Only Mn exceeded the MCL, by a factor of 13 in one sample and by a factor of 1.6 in another. Metal release in the clay-rich limestones was mostly limited to weakly sorbed metals (Na, Ba and Sr) via ion-exchange reactions. Availability of Ca controlled ion exchange of Sr and Ba at low pCO2, but the overall supply of Sr and Ba in sorption sites affected the release of these elements at high pCO2.
Depressurization of the reactors at the end of the experiment, and degassing of CO2, resulted in rapid decrease of metal concentrations in solution, likely due to uptake of the metals by mineral phases. Concentrations of Na and Mg did not decrease during depressurization, probably due to preferential sorption of large amounts of Ca. Dissolution of carbonate material was diminished in the presence of clays. Calcite and dolomite were not as far from equilibrium throughout the duration of the experiment compared to previous experimental rounds, due to buffering of pH through proton uptake by clays, and due to release of Ca via desorption. As a result, adsorption-desorption processes were more important in controlling metal release than dissolution of carbonate minerals
Results from the metal analysis of the aqueous samples showed that minor and trace metals were released into solution during the experiment, sometimes in accordance with calcium release, and sometimes not. Stabilization of pH, due to dissolution of carbonate minerals, did not necessarily correspond to stabilization of the aqueous concentrations of these metals. Minor and trace elements that were found to be released into solution include lead, arsenic, nickel, cobalt, uranium and chromium. Some elements were detected in certain beakers, and not in others. Among the elements that were detected consistently, the release pattern of these metals into solution was overall similar among different rock samples, but the degree of increase in solution varied between rocks. Most elements did not exceed their EPA-mandated MCL for drinking water under our experimental constraints. In specific cases, however, arsenic, chromium and nickel did exceed the EPA-mandated MCLs (State of California-mandated MCL for nickel), under 1 bar of CO2.
Supporting analyses included high-resolution energy-dispersive spectroscopy (EDS), to identify and quantify the distribution of minerals present in the rock samples, and X-ray diffraction (XRD) analysis of the whole rocks and clay fraction.
Intermediate scale experiments on silicate rocks
The same experimental set up described in the previous section was used to react silicate rocks from the Mesa Verde Group at partial pressures of CO2 of 0.01 bar (background) and 1 bar (small CO2 leak). The samples were characterized using sequential extraction, X-ray diffraction, scanning electron microscopy, and optical microscopy prior to and after performing the experiments. Results from these batch experiments provided an estimate of the magnitude and the rate at which metals are released from sandstones in response to an elevated partial pressure of CO 2 . Results imply that under shallow aquifer conditions (<25ºC and 1 bar of pressure) a small CO2 leak could mobilize potentially toxic constituents well above background concentrations, but the primary maximum contaminant levels would never be exceeded, at least over the short term.
Sequential extractions revealed that major and trace elements released during the batch experiments (e.g., Ca, Mg, Sr, Ba, Cd, U, Pb) were bound predominantly to the carbonate-phase within the sandstones. Other metals, namely Fe and Mn, were extracted with the reduction of iron and manganese oxides. However, it is plausible that they are actually associated with a less soluble carbonate mineral, like dolomite, that may require a longer digestion in the acetic acid-sodium acetate buffer solution.
Geochemical modeling demonstrated that dissolution of calcite and dolomite, even though volumetrically insignificant in the sandstones, could be the primary process buffering pH in the batch experiments. When the carbonate minerals in the model were modified to include some common impurities, their dissolution also could explain the increase in Sr, Fe and Mn concentrations. Other metals, like Ba, Cd, and U, which were in the carbonate-bound fraction of the sandstones, and showed a similar release pattern in the batch experiment, could likely be modeled as impurities too.
Geochemical modeling
The numerical reactive transport study, detailed in previous reporting period reports, was completed in 2012, submitted and accepted for publication in Advances in Water Resources.
High T and P Laboratory Geochemical Experiments
Injection of supercritical CO2 into a saline reservoir decreases pH 1 to 2 units. The decrease in pH can lead to mineral dissolution, which may release harmful metals such as arsenic (As), lead (Pb), and barium (Ba) from the reservoir and/or caprock. Small discontinuities in the caprock along faults, fractures, old petroleum wells, or horizontal migration inclusions could allow the reservoir brine and/or CO2 to seep out of the saline aquifer and into an overlying potable aquifer impacting the shallower zones. In order for CCS to become viable, an understanding of how leakage may affect overlying potable aquifers is imperative. Hydrothermal experiments were performed to investigate the release of harmful metals into solution as a result of CO2 injection into a saline reservoir. These experiments focus on reservoir-caprock interface and on the deep reservoir.
The experimental methods used in this project are based on techniques developed in previous work. The experiments reacted rock and brine saturated with constituent minerals at 160ºC and 25MPa, followed by injection of super-critical CO2 (scCO2). The temperature was selected to accelerate kinetics without changing in-situ water-rock reactions, an approach used in similar studies. Table 1 outlines experimental conditions and parameters for both experiments.
Table 1: Experimental conditions and parameters for hydrothermal experiments
| Experiment |
Brine-Reservoir Caprock Interface + scCO2† |
Brine-Deep Reservoir + scCO2† |
| Initial pH | 6.6 ± 0.1 | 6.7 ± 0.1 |
| Temperature (°C) | 160.4 ± 6.5 | 160.0 ± 0.4 |
| Pressure (MPa), Pre-scCO2 Injection | 24.6 ± 1.3 | 25.4 ± 0.4 |
| Pressure (MPa), Post-scCO2 Injection | 38.4 ± 5.4 | 37.8 ± 1.0 |
| Initial Water:Rock Ratio | 21.5 | 21.5 |
| Mineral Proportions (Cc:Do:Py:Sh*) | 31:31:2:0:36 | 47:47:5:0:0 |
| Water-Rock Reaction Time (hours) | 673 | 676 |
| Water-Rock-scCO2 Reaction Time (hours) | 1157 | 815 |
| Surface Area of Unreacted Powders (m2/g) | 3.2914 ± 0.1910 | 0.5394 ± 0.0180 |
| Surface Area of Reacted Powders (m2/g) | 1.5442 ± 0.0203 | 0.2106 ± 0.0116 |
* Gothic shale core samples from the Greater Aneth Field were used in experiments
† Parallel experiments without CO2 were performed; results not shown here
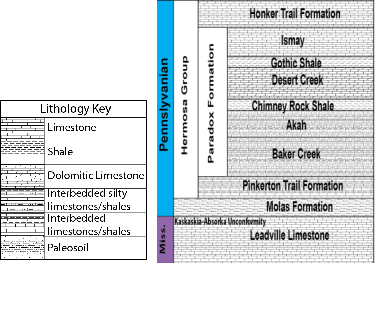
The experiments replicate the Desert Creek Limestone reservoir and Gothic Shale caprock in the Paradox Basin in southeastern Utah (Fig. 1). The Pennsylvanian Desert Creek Limestone, an evaporite carbon sequence, is the main producing zone within the Greater Aneth Field. This formation currently is a combined enhanced oil recovery and sequestration pilot site for the Southwest Regional Partnership on Sequestration. The organic rich Gothic Shale is the sealing unit above the Desert Creek Limestone. The overlying shallow aquifer in the region is the Navajo Sandstone. Experiments emulate injection of CO2 at two different zones within a sequestration reservoir; near the top of the reservoir, where water-rock interactions encompass both reservoir and caprock, and deep within the reservoir, away from the shale caprock.
Figure 1. Stratigraphic column of the producing zones in the Aneth Field within the Paradox Basin, Utah. This project focuses on the Desert Creek limestone and Gothic Shale caprock
Equilibrium modelling is done using Geochemist Work Bench (GWB) to calculate the initial brine compositions and interpret the experimental results. The initial brine composition is calculated to be in equilibrium or as close as possible to the rocks so that there is little to no reaction between rocks and brine prior to scCO2injection.
In both experiments, major ion concentrations remain relatively constant with a few exceptions. In the experiment replicating the caprock-reservoir boundary, an increase in SiO2 and Fe is seen with injection of scCO2. As the experiment continues, the concentration of both dissolved species decreases. These data are consistent with SEM and modeling results, which suggest initial dissolution of minerals followed by precipitation of clays, Fe-sulfides, and Fe-carbonates (Fig. 2). The experiment replicating injection deeper into the reservoir does not exhibit a large change in Fe. Sulfate concentrations increase after scCO2 injection, and subsequently decrease with time. Aqueous chemistry, SEM and modeling results indicate initial dissolution followed by precipitation of anhydrite, pyrite, and various metal sulfides.
Ba, Cu, Pb, and Zn concentrations increase with injection in both experiments. Concentrations subsequently decrease to approximately steady state values 120-330 hours after injection of CO2. In experiments that emulate the caprock-reservoir boundary, final Fe (700 ppb), an element of secondary concern for the U.S. Environmental Protection Agency (EPA), and Pb (50 ppb) concentrations exceed EPA limits, whereas Ba (140 ppb), Cu (48 ppb), and Zn (~450-500 ppb) concentrations remain well below EPA limits. In experiments that simulate deeper reservoir conditions, away from the caprock boundary, final Fe (3500 ppb), Pb (17 ppb) and Ba (155 ppb) concentrations exceed values seen at the caprock-reservoir boundary; whereas Zn (50 ppb) is far less in the deeper reservoir than at the caprock-reservoir boundary. Arsenic values in both experiments exceed the EPA limit. Unfortunately, reliable determination of As concentrations in waters with high ionic strength such as used in this study (I = 3.3 m) is an analytical issue and currently under evaluation.
The experimental results suggest that if brines leak from a storage reservoir and mix with a potable aquifer, Ba, Cu, and Zn will not be contaminants of concern. Lead, Fe and As (still under consideration) initially exceed the EPA threshold and may require careful attention in a sequestration scenario. The caprock plays an active role as a source of metals, although subsequent precipitation may remove metals from solution. However, experimentally observed trends of decreasing trace metal concentrations suggest that these metals could become less of a concern during the life of a carbon repository.
Problems Encountered and Solutions Implemented:
A graduate student started working on the project in June 2010. That student left the University of Wyoming in December 2010. Experimental work was delayed until a new student began working on the project in June 2011. With the no-cost extension in place, this student has the needed time to complete her work.
Risk Modelings
Basin-scale hydrologic modeling
The parallel integrated hydrologic model ParFlow was used to simulate basin-scale hydrology and the streamline modeling approach developed as a part of this project was used to evaluate the role of coupled physical and geochemical heterogeneity in plume development.
Streamline modeling
In the interest of investigating the role of coupled physical and geochemical heterogeneity in plume development, simulations of three-dimensional heterogeneous transport systems with non-linear kinetic reactions were performed for 100 realizations each. Three geochemical conditions were simulated over ensembles of 100 physically heterogeneous realizations, 1) spatially homogeneous reactive mineral surface area, 2) positively correlated reactive mineral surface area to permeability, and 3) negatively correlated reactive mineral surface area to permeability. Large-scale reaction rates were calculated at a sequence of transverse planes to quantify changes in plume evolution due to non-linear kinetics and solute residence time (t). Here, reactive conditions were a product of a hypothetical CO2 intrusion from a carbon capture utilization and storage (CCUS) operation into groundwater, which results in aqueous dis-equilibrium with aquifer mineralogy. In order to illustrate the complicated reactive process, a conceptual model of the reaction path is shown that relates residence time to reach chemical equilibrium (teq) to the advected distance traveled at which equilibrium is achieved (Leq). Results first show that the spatially heterogeneous distribution of geochemical reactive surface areas will affect both effective reaction rate (Krxn,eff) and Pb concentrations before equilibrium is met. Furthermore, the ensemble Krxn,eff and Pb concentrations are the same as equilibrium is achieved for all three geochemical conditions simulated. Finally, we demonstrate that positively and negatively correlating reactive surface area to hydraulic conductivity will result in varying teqand Leq.
Future Activities:
High Temperature-Pressure Laboratory Experiments
Final data interpretation for the reservoir-caprock and deep reservoir experiments will be completed.
Additional hydrothermal experiments were performed to react idealized injection reservoir (dolomite), caprock (illite), and caprock-reservoir in water at 160°C and 25MPa for ~55 days; 15 to reach steady state before CO2 is injected and 40 to monitor changes due to CO2. In all three experiments, preliminary data analysis has been completed. Our analysis suggests that Ca, Fe, Mg, Mn and SO4 increase with injection, but slowly decline through termination of the experiments. This trend suggests initial dissolution followed by re-precipitation of carbonates, which can be seen in initial modeling and SEM results. Trace metals in these experiments do not exceed the U.S. EPA's primary maximum contaminate limit (MCL), but geochemical patterns denote valuable information for metal release, co-precipitation, and sorption of metals in a sequestration scenario. Experiments replicating an idealized carbonate injection reservoir show metals more readily mobilized than in the caprock and caprock-reservoir experiments. Once the caprock is included in the system, water chemistry, SEM and modeling results suggest increased sorption and co-precipitation of the potentially harmful metals. Final data interpretation for these idealized reservoir-caprock and deep reservoir experiments will be completed.
Aquifer Experiments
Final data collection and analysis (including laser ablation ICP-MS, QEMSCAN, and additional geochemical modeling) and manuscript submission for batch experiments with carbonate rocks and silicate rocks will be completed by August 2013. Experimental design for intermediate-scale experiments is complete, tanks will be built by May 2013, and experiments will be completed by September 2013.
Risk Assessment
Final data analysis and journal manuscript submission of combined physical and geochemical heterogeneous models will be completed by June 2013.
Journal Articles on this Report : 8 Displayed | Download in RIS Format
| Other project views: | All 70 publications | 14 publications in selected types | All 14 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Atchley AL, Maxwell RM, Navarre-Sitchler AK. Human health risk assessment of CO2 leakage into overlying aquifers using a stochastic, geochemical reactive transport approach. Environmental Science & Technology 2013;47(11):5954-5962. |
R834387 (2012) R834387 (Final) |
Exit Exit |
|
|
Atchley AL, Maxwell RM, Navarre-Sitchler AK. Using streamlines to simulate stochastic reactive transport in heterogeneous aquifers: kinetic metal release and transport in CO2 impacted drinking water aquifers. Advances in Water Resources 2013;52:93-106. |
R834387 (2012) R834387 (Final) |
Exit Exit Exit |
|
|
Bearup LA, Navarre-Sitchler AK, Maxwell RM, McCray JE. Kinetic metal release from competing processes in aquifers. Environmental Science & Technology 2012;46(12):6539-6547. |
R834387 (2011) R834387 (2012) R834387 (Final) |
Exit |
|
|
Navarre-Sitchler AK, Maxwell RM, Siirila ER, Hammond GE, Lichtner PC. Elucidating geochemical response of shallow heterogeneous aquifers to CO2 leakage using high-performance computing: implications for monitoring of CO2 sequestration. Advances in Water Resources 2013;53:45-55. |
R834387 (2011) R834387 (2012) |
Exit Exit Exit |
|
|
Siirila ER, Navarre-Sitchler AK, Maxwell RM, McCray JE. A quantative methodology to assess the risks to human health from CO2 leakage into groundwater. Advances in Water Resources 2012;36:146-164. |
R834387 (2011) R834387 (2012) R834387 (Final) |
Exit Exit Exit |
|
|
Wunsch A, Navarre-Sitchler AK, Moore J, Ricko A, McCray JE. Metal release from dolomites at high partial-pressures of CO2. Applied Geochemistry 2013;38:33-47. |
R834387 (2012) R834387 (Final) |
Exit Exit Exit |
|
|
Wunsch A, Navarre-Sitchler AK, McCray JE. Geochemical implications of brine leakage into freshwater aquifers. Groundwater 2013;51(6):855-865. |
R834387 (2012) R834387 (Final) |
Exit |
|
|
Wunsch A, Navarre-Sitchler AK, Moore J, McCray JE. Metal release from limestones at high partial-pressures of CO2. Chemical Geology 2014;363:40-55. |
R834387 (2012) R834387 (Final) |
Exit Exit |
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.