Grantee Research Project Results
2010 Progress Report: Assessment of Microbial Pathogens in Drinking Water using Molecular Methods Coupled with Solid Phase Cytometry
EPA Grant Number: R833830Title: Assessment of Microbial Pathogens in Drinking Water using Molecular Methods Coupled with Solid Phase Cytometry
Investigators: Pyle, Barry H , Ford, Timothy E.
Current Investigators: Pyle, Barry H , Camper, Anne
Institution: Montana State University - Bozeman , Little Big Horn College
EPA Project Officer: Aja, Hayley
Project Period: March 1, 2008 through February 28, 2011 (Extended to February 28, 2013)
Project Period Covered by this Report: June 10, 2010 through June 9,2011
Project Amount: $599,996
RFA: Development and Evaluation of Innovative Approaches for the Quantitative Assessment of Pathogens and Cyanobacteria and Their Toxins in Drinking Water (2007) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
To develop and evaluate innovative approaches for quantitative assessment of pathogens in drinking water sources, using fluorescent in situ hybridization (FISH) coupled with the Solid Phase Laser Cytometer (ScanRDI) and other molecular methods.
Target organisms: Escherichia coli O157:H7, Helicobacter pylori, Legionella pneumophila, Mycobacterium avium, Aeromonas hydrophila, Giardia lamblia, Cryptosporidium parvum.
Progress Summary:
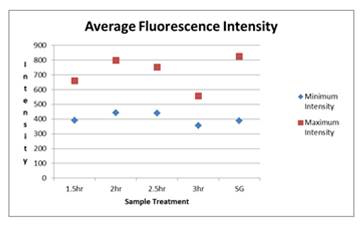
Figure 1. Average Flourenscence Intensity of E. coli cells detected
by epiflourescence microscopy after hybridization with EUB388-
HRP at 20 ng/µl for 1.5-3.0 h. Labeling with SYBR Green (SG)
was included as a control
| Table 1 | |
| No Preincubation | 0.48 |
| Chloramphenicol 30 min | 0.60 |
| NA 30 min | 0.62 |
| NA 60 min | 0.70 |
| NA 120 min | 0.68 |
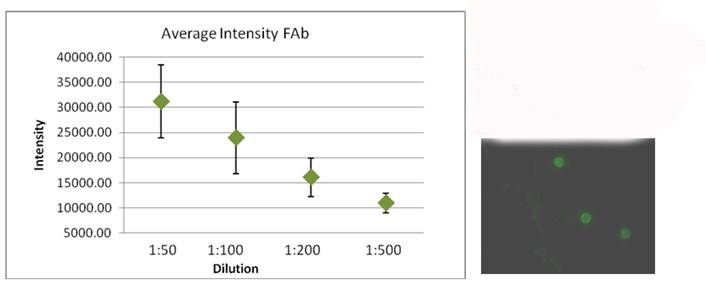
Figure 2. Comparing the flourescent intensity of Cryptosporidium oocysts labled with flourescent antibody (LifeSpan).
The image on the right shows Oocysts labled with antibody diliuted 1:100.
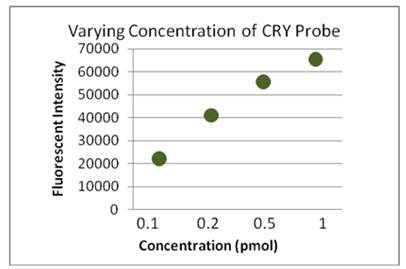
Figure 3. Flurescent intensity data Cryptosporidium oocysts hybridized
with the CRY-HRP probe at concentratins from 0.1 to 1.0 pmol for
2.0 h at 48 oC.
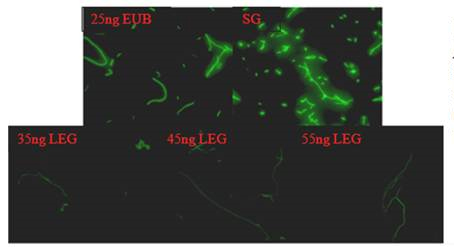
Figure 5. Hubridization of L. pneumophila with 25ng of EUB-HRP probe (EUB),
35, 45 and 55 ng of LEGPNE1-HRP probe (LEG), and labeling with SYBR
Green (SG).
Future Activities:
Further work is being done to optimize FISH for use with Cryptosporidium, including TSA-CY3 labeling and labeling oocysts with both FAb and FISH at once. Simultaneous detection of E. coli and Cryptosporidium as well as other target organisms using FISH techniques and the ScanRDI will be investigated. The optimization for L. pneumophila cells will be adapted for the membrane filter method for use with the ScanRDI and for other target bacterial pathogens. Procedures for Giardia will be added in the near future. Sampling and analysis of water from the Crow Indian Reservation, applying the procedures that have been developed, will start this summer (2011).
References:
1. Amann, R. I., B. Zarda, D. A. Stahl, and K.-H. Schleifer. 1992. Identification of Individual Prokaryotic Cells by Using Enzyme-Labeled, rRNA-Targeted Oligonucleotide Probes. Applied and Environmental Microbiology 58:3007-3011.
2. Baudart, J., J. Coallier, P. Laurent, and M. Prévost. 2002. Rapid and Sensitive Enumeration of Viable Diluted Cells of Members of the Family Enterobacteriaceae in Freshwater and Drinking Water. Applied & Environmental Microbiology 68:5057.
3. Broadaway, S. C., S. A. Barton, and B. H. Pyle. 2003. Rapid staining and enumeration of small numbers of total bacteria in water by solid-phase laser cytometry. Applied and Environmental Microbiology 69:4272-4273.
4. Deere, D., G. Vesey, M. Milner, K. Williams, N. Ashbolt, and D. Veal. 1998. Rapid method for fluorescent in situ ribosomal RNA labelling of Cryptosporidium parvum. Journal of Applied Microbiology 85:807-818.
5. Moreno, Y., M. A. Ferrus, J. L. Alonso, A. Jimenez, and J. Hernandez. 2003. Use of fluorescent in situ hybridization to evidence the presence of Helicobacter pylori in water. Water Research 37:2251-2256.
6. Moter, A., and U. B. Gobel. 2000. Fluorescence in situ hybridization (FISH) for direct visualization of microorganisms. Journal of Microbiological Methods 41:85-112.
7. Ouverney, C. C., and J. A. Fuhrman. 1997. Increase in fluorescence intensity of 16S rRNA in situ hybridization in natural samples treated with chloramphenicol. Applied and Environmental Microbiology 63:2735-2740.
8. Pernthaler, J., F. O. Glockner, W. Schonhuber, and R. Amann. 2001. Fluorescence in situ hybridization (FISH) with rRNA-targeted oligonucleotide probes. Methods in Microbiology, Vol 30 30:207-226.
9. Vesey, G., N. Ashbolt, E. J. Fricker, D. Deere, K. L. Williams, D. A. Veal, and M. Dorsch. 1998. The use of a ribosomal RNA targeted oligonucleotide probe for fluorescent labelling of viable Cryptosporidium parvum oocysts. Journal of Applied Microbiology 85:429-440.
Journal Articles:
No journal articles submitted with this report: View all 6 publications for this projectSupplemental Keywords:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
