Grantee Research Project Results
2011 Progress Report: Aptamer Capture and Optical Interferometric Detection of Cyanobacteria
EPA Grant Number: R833839Title: Aptamer Capture and Optical Interferometric Detection of Cyanobacteria
Investigators: Campbell, Daniel P , Xu, Jie , Ellington, Andy
Current Investigators: Campbell, Daniel P , Ellington, Andy , Xu, Jie
Institution: Georgia Institute of Technology , The University of Texas at Austin
EPA Project Officer: Aja, Hayley
Project Period: June 1, 2008 through April 30, 2011 (Extended to January 31, 2013)
Project Period Covered by this Report: June 1, 2011 through May 31,2012
Project Amount: $600,000
RFA: Development and Evaluation of Innovative Approaches for the Quantitative Assessment of Pathogens and Cyanobacteria and Their Toxins in Drinking Water (2007) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
Develop a sensor capable of detecting cyanobacterial toxins using aptamers as capture receptor.
Progress Summary:
The program was to develop aptamers, which are synthetic antibodies for cyanobacterial toxins, and bind them onto the Georgia Tech Research Institute’s planar waveguide interferometric sensor. The aptamer response is to be compared to the response obtained from natural antibodies. Of the three cyanobacterial toxins – anatoxin-a, cylindrospermopsin and microcystin-LR, shown in Figure 1 – the microcystin toxin, its antibody and aptamer are being investigated.
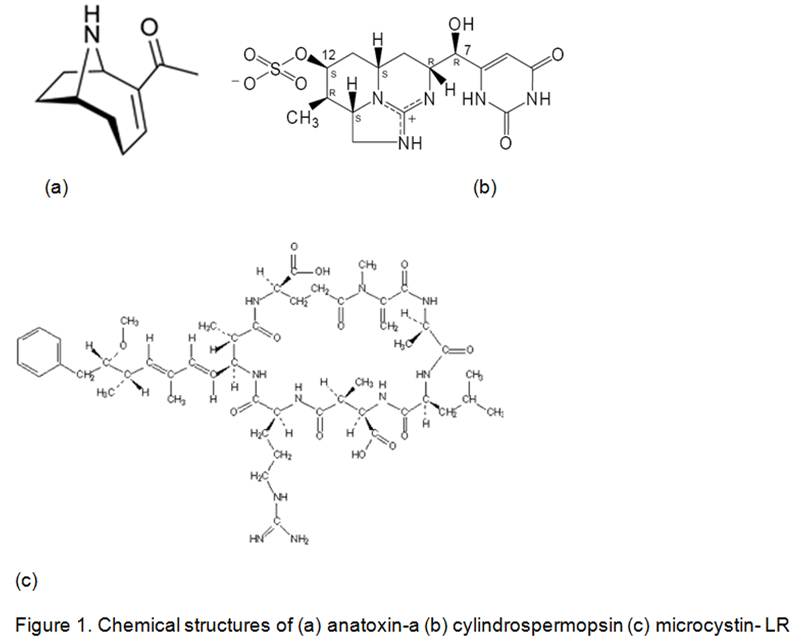
Aptamers and Antibodies
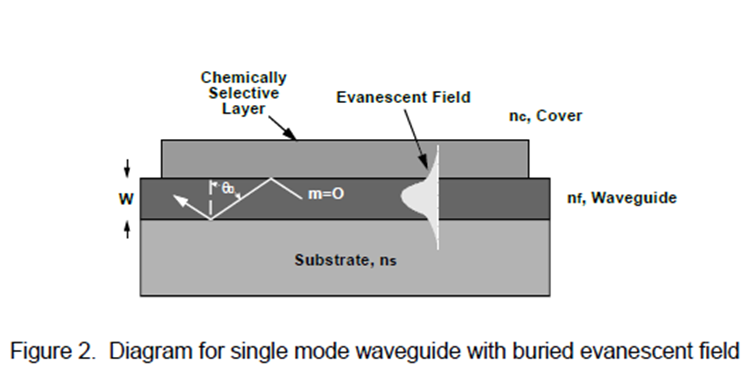
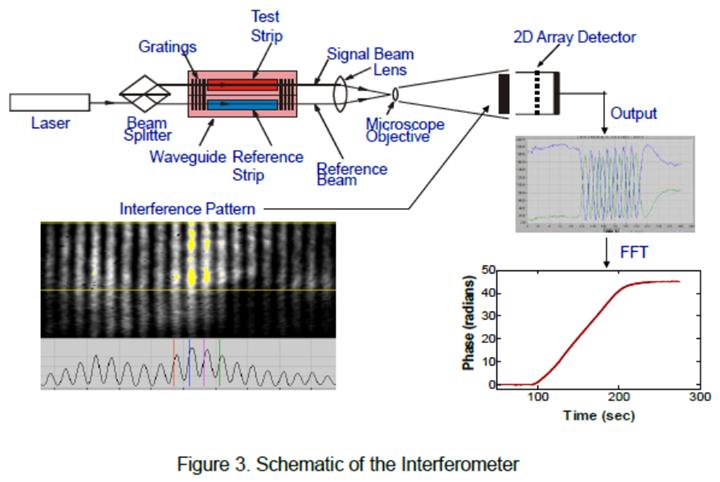
Future Activities:
The future activities are to: biotinylate the aptamers, bind the aptamers to the waveguide surface, and test with microcystin.
Supplemental Keywords:
Interferometric sensor, microcystin, waveguide, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Water, Health Risk Assessment, Environmental Chemistry, Drinking Water, Environmental Engineering, Environmental Monitoring, microbial contamination, gene microarray assay, early warning, microbial risk assessment, monitoring, aquatic organisms, drinking water system, drinking water contaminants, other - risk assessment, cyanobacteriaProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
