Grantee Research Project Results
2009 Progress Report: Aptamer Capture and Optical Interferometric Detection of Cyanobacteria
EPA Grant Number: R833839Title: Aptamer Capture and Optical Interferometric Detection of Cyanobacteria
Investigators: Campbell, Daniel P , Ellington, Andy , Xu, Jie
Institution: Georgia Institute of Technology , The University of Texas at Austin
EPA Project Officer: Aja, Hayley
Project Period: June 1, 2008 through April 30, 2011 (Extended to January 31, 2013)
Project Period Covered by this Report: June 6, 2009 through June 5,2010
Project Amount: $600,000
RFA: Development and Evaluation of Innovative Approaches for the Quantitative Assessment of Pathogens and Cyanobacteria and Their Toxins in Drinking Water (2007) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
The objective of this program is to take the optical interferometric sensor that has been under development at the Georgia tech Research Institute for the past two decades and configured it to detect and monitor a series of cyanobacterial toxins. After initial testing with known antibodies for a series of cyanobacterial toxins, the detection scheme will convert over to aptamers to provide a more rugged reusable sensor for field deployment.
Progress Summary:
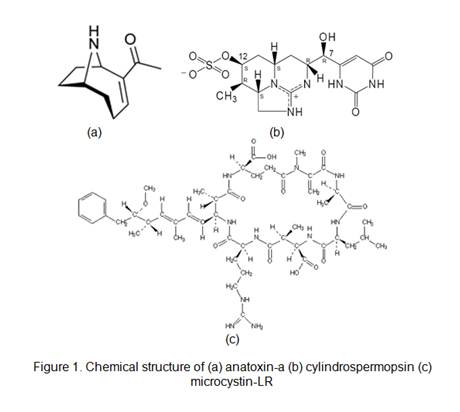
Cyanobacterial toxins have been recognized as a potential hazard in drinking water throughout the world. These bacteria are ubiquitous growing wherever the conditions are favorable that that includes almost any standing water one can imagine such as a freshwater pond, a swampy area, a dog's water bowl and even one's family toilet. Cyanobacterial toxins have been categorized into three groups based on their chemical structures. The toxins with highest priority are the neurotoxins anatoxin-a, cylindrospermopsin and most frequently found microcystin-LR. Their chemical structures of the more common toxins are shown in Figure 1.
The optical interferometric sensor provides a rapid, field deployable and inexpensive device that can be adapted for detecting of cyanobacterial toxins. The GTRI interferometric optical sensor has been shown to be capable of detecting a wide variety of chemical and biological species. The sensor is responds quickly, with high sensitivity and specificity, and provides a direct measurement with no additional steps or consumable reagents. Some of the advantages of this sensor system for environmental applications are its low initial and per-sample cost, the ability to detect multiple analytes simultaneously, and the speed and sensitivity of detection. Systems can be designed for mobile, on-site field analysis with instant results or in-situ monitoring with data logging and communication to a monitoring base.
In this project, the GTRI sensor will be first evaluated for detecting the cyanobacterial toxins using known antibodies as the recognition element that will bind with the toxins. In this detection scheme, the antibodies are covalently linked to the optical sensors surface. When the toxin is present it will bind to the recognition element causing a change in the refractive index. This refractive index change is detected by the waveguide interferometer and is converted after calibration to a concentration of the compound or species. Examples of this detection scheme employing this optical interferometer have included the detection of biotin with an avidin as the recognition element, and the detection of ricin A-chain a protein toxin employing an aptamer detection element among countless other examples. After evaluating the sensors ability to detect the cyanobacterial toxins using known commercially available antibodies, the sensors recognition element will be changed over to a nucleic acid based aptamers as the recognition element. The aptamer provide a much more durable, reusable and reproducible recognition element than that of antibodies.
The development of aptamers for these specific toxins will constitute a major element of this program. The development of the aptamers is headed up by Dr. Andy Ellington at the University of Texas who has spent a large portion of his career in developing aptamers for a wide variety of chemical and biochemical species. The other half of the program is head up by Dr. Dan Campbell at the Georgia Tech Research Institute who has worked on developing the optical interferometric sensor for a wide array chemical species including organic and inorganic compounds (aromatic and chlorinated compounds, ammonia, chlorine, TNT), proteins and cofactors (avidin, biotin, ricin, IgG, IgE, DNA), real-time monitoring of nucleic acid hybridization, and whole organisms detection (including Salmonella, Campylobacter and Anthrax spores). Current levels of detection are in the sub-parts-per -billion range for TCE and TNT, the femtomolar range for proteins and 1000 cfu/ml for whole organisms. The interferometric sensor provides a direct, fast measurement (in seconds to minutes) without the need for additional and time-consuming washing or incubation steps.
INTERFEROMETRIC SENSOR AND WAVEGUIDES
At the heart of the interferometric sensor is a planar waveguide. Planar waveguides have evanescent fields sensitive to index of refraction changes in the volume immediately above (up to 5000 Å) the surface (Figure 2).
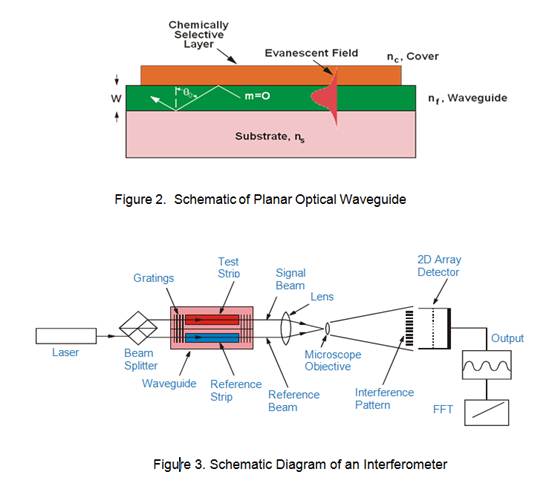
Placing a chemoadsorptive or recognition element within this evanescent region provides the basis for the sensor response. To measure this response, a reference beam, traveling adjacent to the sensing beam, is optically combined with the sensing beam creating an interference pattern of alternating dark and light fringes that is imaged by the detector. Chemical or physical interactions (such as the toxin binding to an antibody) change the index of refraction causing the propagating light speed, or phase, to change in a direction opposite to that of the index change. To measure this change an interferometer, shown schematically in Figure 3, is assembled.
The sensing and reference beams that are combined to create an interference pattern which is imaged on a two-dimensional array detector. When chemical or physical interactions occurs on the waveguide surface the interference pattern shifts, producing a sinusoidal output. A single mathematical algorithm converts this sinusoidal output into total phase shift that is related to the amount of material detected by the interferometer. With detectable sensitivities on the order of 0.01 radians, index changes less than 10-6 can be measured.
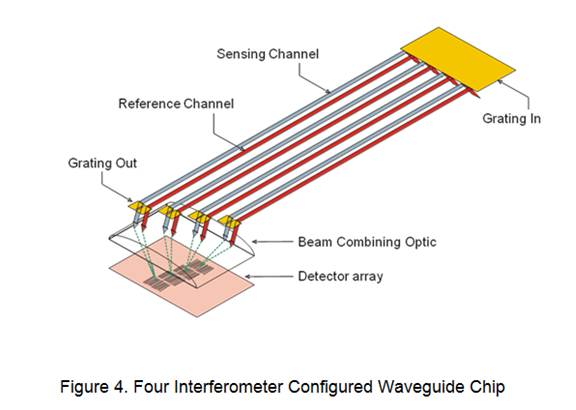
Currently the integrated optic waveguide platform consists of one, four or eight interferometers or sensors on a 1x2-cm glass substrate; a four interferometer chip is shown in Figure 4. A different receptor film can be deposited on each channel allowing for multiple analyte sensing. The current software converts the optical response to total phase change which in turn corresponds to the concentration of the analyte present.
A great deal of progress has been made in the sensor during the first year of this program. The sensor platform and the box which houses it is an integral component of every sensor program currently being worked on at GTRI. The advantage of this overlaying of programs means that no one program is burdened by the development of the sensor system itself but is shared by all the programs. At the onset of this program the sensor platform and box were a bit cumbersome and expensive. The initial sensors optical bench had all the optical components mounted on a 6 x 4 inch plate of aluminum. There were many adjustments that needed to be made with this configuration in order to align the optical beams from the laser to the input gratings, and out to the detector. An earlier version of the waveguide chip was also very complicated. However, a few years the optical chip was reinvented to make its fabrication and function a lot simpler. The fabrication of the chip is done using standard semiconductor microfabrication steps. The earlier version required over a dozen of these steps and the yield was quite low. The new sensor chip provided a great leap forward requiring only a few steps with no very complicated or precise alignment steps. The design allowed multiple interferometers to be fabricated on each chip. The new optical design of the chip allowed for the elimination of several of the optical components and adjustments required on the optical platform. Fabrication on a large scale is now possible.
The initial sensor system platform and boxes were also very complicate in design and cost approximately $15K to provide each unit. Since this program began, a great step in simplification of the optical platform was accomplished, shown in Figure 5. The device shown in Figure 5 contains the laser, waveguide and detector. A couple of simple adjustments bring the sensor into overall alignment and the one adjustment knob is used to translate the optical input beam so as to strike the front of the input grating. This adjustment is designed for any slight variation in dicing of the chip or offset of the grating that make occur in fabrication. Whereas the earlier optical platform and all the pieces that held all the optical components were machined out of aluminum, this new design was made entirely out of Delrin, an inexpensive plastic. The plastic components of which there are seven with only the base piece having any complicated machining can be manufactured using rapid prototyping/photofabrication techniques which permits the sensor pieces to be manufactured overnight with a AutoCAD drawing and for a couple of hundred dollars. This monumental leap in the sensor design allows one to envision thousands of the sensors in almost any application one can imagine, not just for the toxins but for any chemical or biochemical sensing application and even for the individual to wear in situations where exposure to chemicals or biological toxins or bacteria/viruses are likely or possible.

An array of sensor chips has been fabricated for the program. They include several single interferometer chips, a series of four interferometer chips for looking at cross-reactivity of the receptor groups and the toxins and a few eight interferometer chips for wide component testing with some interferometer for replication. A number of the sensor heads have been fabricated. Since the price of fabricating the sensor heads has become inexpensive, additional sensor will be fabricated as backups. There are enough sensors to run experiments in series.
New software has been written and debugged for use with the single and multiple interferometer chips.
TOXINS, ANTIBODIES AND APTAMERS:
During this year, a search was made for antibodies for the three toxins of interest. A number of commercial sources have been located for the antibodies. In addition, commercial sources for the cyanobacterial toxins have been found. Since antibodies have varying structures and affinities for the toxins, a series of commercially obtained antibodies will be screened in this next year for their binding to the toxins. The best antibodies will be used as a comparison to the aptamers synthesized in the Ellington Lab. Aptamers identified can be synthesized using standard PCR techniques. This is one of the beauties of aptamers is that once a structure is identified then large amounts of the material can be made in which all the copies are molecularly identical. As opposed to antibodies in which the structures vary with the source host, i.e. rat, mouse, goat, rabbit, and therefore, have varying affinities, the aptamers all have the same affinities for the target due to their identical structure. Granted there may be different aptamers that complex with each toxin the highest binding structures will be the ones to pass on to the sensor testing.
Binding protocol for attaching either the antibodies or the aptamers to the silicon dioxide surface of the waveguide has already been determined. The antibodies are covalently linked either by reductive amination of an aldehyde on the waveguide surface to an aldehyde to the free amino of a lysine group on the antibody, or through a disulfide linkage between cysteine and a mercaptan group on a small pendant group coming off a functionalized waveguide. Aptamers are attached using a biotin-avidin linkage where the aptamer is biotin labeled and the waveguide surface is functionalized with avidin using the similar linkage used for the antibodies.
A subcontract between Georgia Tech and the University of Texas took some time to accomplish but is now in place and work on the aptamers has begun.
SUMMARY
Waveguides interferometers have been made and tested for optical throughput and sensitivity to refractive index changes. Waveguides with single interferometers, four and eight interferometers have been fabricated. All of these waveguides are constructed on a 16 x 32 mm piece of glass. Numerous sensor heads have been constructed. These sensor heads are constructed to hold any of the waveguide chips made. Flow cells also have been made to contain the samples of solutions to be tested. New software has been written and debugged for use with the single and multiple interferometer chips. A couple of sensor heads have been mounted in boxes which contain pumps to move solution samples over the waveguides.
Commercial sources for the toxins and the antibodies have been identified. Materials have not been purchased as of yet. Next on the program agenda is to purchase the toxins and antibodies, functionalize the waveguides and develop an evaluation protocol for each of the toxins. The aptamer subcontract with the University of Texas has been put in place.
Supplemental Keywords:
RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Water, Health Risk Assessment, Environmental Chemistry, Drinking Water, Environmental Engineering, Environmental Monitoring, microbial contamination, gene microarray assay, early warning, microbial risk assessment, monitoring, cyanobacteria, drinking water system, drinking water contaminants, aquatic organisms, other - risk assessmentProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
