Grantee Research Project Results
Final Report: Improved Prediction of In-Cloud Biogenic SOA: Experiments and CMAQ Model Refinements
EPA Grant Number: R833751Title: Improved Prediction of In-Cloud Biogenic SOA: Experiments and CMAQ Model Refinements
Investigators: Turpin, Barbara , Seitzinger, Sybil
Institution: Rutgers
EPA Project Officer: Chung, Serena
Project Period: November 1, 2007 through August 31, 2010 (Extended to October 31, 2011)
Project Amount: $598,544
RFA: Sources and Atmospheric Formation of Organic Particulate Matter (2007) RFA Text | Recipients Lists
Research Category: Particulate Matter , Air Quality and Air Toxics , Air
Objective:
- Develop mechanistic/kinetic data needed to simulate in-cloud formation of secondary organic aerosol (SOA) in the presence of HNO3,
- Identify conditions for which predicted in-cloud SOA formed from isoprene decreases with reductions in interstitial concentrations of ·OH and HNO3 (atmospheric oxidants with anthropogenic precursors), and
- Incorporate an in-cloud SOA formation pathway into the Community Multiscale Air Quality (CMAQ) model and explore the magnitude of in-cloud SOA formation through a limited set of model simulations.
Summary/Accomplishments (Outputs/Outcomes):
Overview: It is now widely accepted that aqueous oxidation of water-soluble carbonyl compounds (e.g., glyoxal, methylglyoxal, glycolaldehyde, acetic acid) by OH radical in clouds and wet aerosols results in the formation of secondary organic aerosol (SOA; Ervens et al., ACP 2011). Reactions during cloud droplet evaporation (Loeffler et al., EST 2006; Ortiz- Montalvo, submitted), with inorganic constituents in wet aerosols (Noziere et al., JPC 2009; Yasmeen et al., ACP 2010; Perri et al., AE 2010), and with other oxidants (e.g., ozone; Grgic et al., PCCP 2010) also contribute. The potential importance of SOA formation through aqueous chemistry is all the more apparent when one realizes that liquid water is ubiquitous and abundant. In addition to liquid water in clouds, model estimates suggest that globally, the mass of aerosol water is two to three times the dry particle mass (Liao and Seinfeld, JGR 2005). Efforts are currently underway to add SOA formation through aqueous chemistry to several regional and global chemical transport models. While current modeling estimates have large uncertainties, SOA formed through aqueous chemistry appears to be comparable in magnitude to traditional SOA formed through gas-phase chemistry and vapor-pressure driven partitioning (Gong et al., Atmos. 2011; Fu et al., JGR 2008, AE 2009; Carlton et al., EST 2008). Aqueous SOA also largely explains the atmospheric burden of oxalate (Myriokefalitakis et al., ACP 2011). Based on the addition of this chemistry to a cloud parcel model, it appears that aqueous SOA formation from the biogenic hydrocarbon, isoprene, is enhanced at high NOx because the gas phase yields of water-soluble carbonyl compounds are higher at high NOx (Ervens et al., GRL 2008). This suggests that SOA formed from biogenic hydrocarbons through aqueous chemistry might be reduced through controls on anthropogenic emissions.
Anthropogenic atmospheric wet deposition of secondary organic nitrogen is implicated in the eutrophication of coastal estuaries (Seitzinger and Sanders, Mar. Ecol. Progr. Ser. 1997). Our analyses (Altieri et al., EST 2009; ACP 2009) identified nitrogen-containing organics in New Jersey rainwater samples. Smog chamber experiments suggest that water-soluble organic nitrogen compounds are formed through gas photochemistry at high NOx. We hypothesized that organic nitrogen also is formed from reactions in atmospheric waters. Formation of nitrogen-containing organics has now been identified in chamber experiments conducted with wet ammonium sulfate particles in the presence of glyoxal, demonstrating that nitrogen-containing organics can form in wet aerosols (Galloway et al., ACP 2009). Our experiments, reported below, suggest that organic nitrogen formation from glyoxal in the presence of nitric acid or ammonium sulfate is negligible at cloud-relevant concentrations.
Our most recent aqueous laboratory experiments with glyoxal/methylglyoxal and OH radical (plus control experiments) verify the formation of oxalate, other organic acids, and high molecular weight organic compounds (HMWCs), including oligomers from OH radical reactions (Altieri et al., AE 2008; Lim et al, ACP 2010; Lim et al., in preparation, 2012; Tan et al., EST 2009; Tan et al., AE 2010; Tan et al., ACP 2012). These are common constituents of atmospheric particles. For example, HMWCs account for roughly one-third of atmospheric organic aerosol mass and are not formed through atmospheric gas-phase chemistry (Zappoli, et al., 1999; Kiss et al., 2002; Lin et al., JAS 2010). SOA formation through aqueous photooxidation (e.g., cloud processing) was proposed by Blando and Turpin (AE 2000). It is the only identified process that can explain the atmospheric abundance/temporal dynamics of oxalate; it has different precursors than traditional SOA, and could be a substantial contributor to total SOA globally and regionally. We hypothesized that similar chemistry also occurs in aerosol water. In fact, our most recent experiments and modeling suggest that aqueous chemistry leads to the formation of oligomers/HMWCs in wet aerosols and organic acids/salts in clouds (Lim et al., ACP 2010). Recent atmospheric evidence for aqueous SOA formation is provided by ambient measurements showing an increase in SOA at relative humidities above 70% (Hennigan et al., GRL, 2008; ACP 2009; Sorooshian et al., GRL 2010; Ervens et al., ACP 2011).
One limitation of our previous work is that experiments were conducted at concentrations about 100 times greater than those found in clouds (and 1,000 times lower than concentrations in aerosol water). Our current STAR grant included support (50%) for the purchase of an ion chromatography (IC) system that has allowed us to conduct experiments at cloud-relevant (and higher) concentrations (Tan et al., EST 2009). Two additional analytical improvements have been made during the course of this project. We can now conduct experiments with real-time analysis by electrospray ionization (ESI) mass spectrometry (Perri et al., AE 2009). We also can analyze samples by ESI-MS after IC pre-separation (Tan et al., AE 2010). These techniques have improved our mechanistic understanding in substantial ways. For example, we now understand that, in the presence of OH radical, organic radical-radical reactions are the major source of HMWC/oligomers from both glyoxal and methylgloxal (Tan et al. ACP 2011; Lim et al, ACP 2010, ACP 2012 in preparation). Our understanding of the detailed radical chemistry has extended the range of concentrations for which we can make accurate predictions, and has enabled us to develop SOA yields for use in regional and global models (Lim et al., ACP 2010; Lim et al., ACPD 2012 in preparation). Finally, we have been collaborating with EPA scientists and others to facilitate incorporation of in-cloud SOA formation into CMAQ and other models.
Objective 1: With support from this grant and Rutgers University, we purchased a Dionex Ion Chromatography System (ICS-3000), which has allowed us to conduct aqueous photooxidation experiments at lower concentrations than previously possible. In year 1, we conducted experiments with glyoxal + ·OH (10-12 M) at 30 μM, 300 μM, and 3000 μM (Tan et al., EST 2009) (Figure 1). In year 2, we conducted the same suite of experiments with methylglyoxal (Tan et al., AE 2010) (Figure 2), and in year 3 with acetic acid (Tan, ACP 2011). A subset of these experiments has been repeated with the addition of cloud-relevant concentrations of H2SO4 (840 μM), HNO3 (840 μM), or (NH4)2SO4 (1.68 mM). In all cases, OH radical was formed from UV photolysis of H2O2. Therefore, control experiments were conducted in the absence of UV and separate control experiments in the absence of H2O2, to understand to what extent the observed products were the result of OH radical reactions. Our previous experiments were conducted only at 1000-3000 μM. The 30 μM experiments are representative of cloud concentrations. Concentrations of dissolved organics in aerosol water could be as high as 1-10 M. Acetic acid is a ubiquitous, water-soluble atmospheric constituent formed mostly through gas-phase chemistry and an intermediate aqueous-phase product of methylglyoxal plus OH radical. Acetic acid experiments were conducted to investigate the hypothesis that methylglyoxal oligomers are oligoesters formed through an acetic acid reaction pathway via condensation reactions (Altieri et al., AE 2008). Some experiments of each type were analyzed in real time by ESI-MS. Discrete samples were collected and analyzed for organic acids by Ion Chromatography (IC), and their mass spectra were obtained by ESI-MS with and without pre-separation by IC. Ultra-high resolution Fourier Transform Ion Cyclotron Resonance Electrospray Ionization Mass Spectroscopy (FTICR-MS) was conducted on selected samples. Total carbon in each sample was measured using a Total Organic Carbon Analyzer. Sulfate and nitrate were measured by IC. Dissolved oxygen and pH were measured at the beginning and end of each experiment. The chemistry in the reaction vessel also was modeled using our aqueous phase chemistry model (2005 Lim model). In this way, we could use the experiments to validate and refine our chemistry model. The rate constants in the model are reasonably well known, but in many cases the products had not previously been verified.
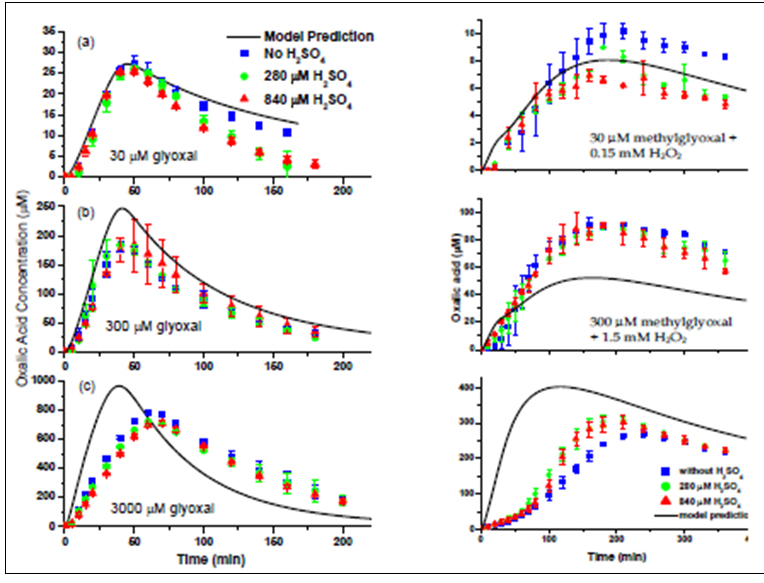
Figure 1. Oxalate time profiles from batch glyoxal ± H2SO4_ OH radical experiments (left) and methylglycol ± H2SO4 + OH radical
experiments (right) wiht model predictions. Solid lines are modeled concentrations, and data points are quantified oxalate concentrations
from IC andalysis H2SO4 concentration in µM is given in legend.
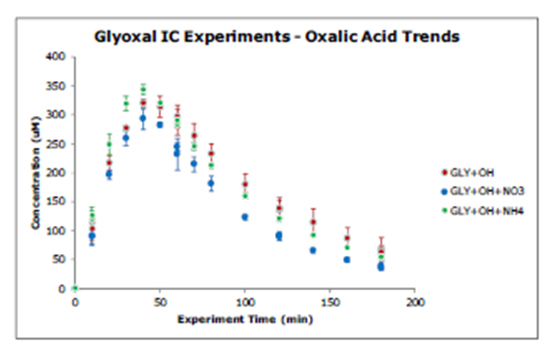
Figure 2. Oxalate time profiles from batch glyoxal + OH radical experiments ± 1.7 mM
HNO3 or 0.84 mM (NH4)2SO4.
The 2005 Lim model did a good job of capturing the magnitude and concentration dynamics of oxalic acid and total carbon for glyoxal, acetic acid and methylglyoxal experiments conducted at cloud-relevant concentrations in the presence and absence of inorganic constituents. This suggests that the Lim model is adequate for use in predicting glyoxal and methylglyoxal cloud chemistry leading to SOA formation. The presence of sulfate, nitrate and ammonium at cloud-relevant concentrations has little effect on this chemistry (Figures 1-3). SOA yields from the aqueous chemistry of these organics in clouds are provided by Lim et al (2012, in preparation).
By examining changes in the concentration dynamics with increasing concentrations, we are developing a better understanding of the chemistry in wet aerosols. As the initial concentrations of the organic precursor increased, the predictive capability of the original model degraded, and the measured organic acids accounted for a smaller and smaller portion of the total carbon in the reaction vessel. We found that increasing precursor concentrations resulted in samples with increasingly complex mass spectra and resulted in the formation of organic acids and oligomers with larger carbon numbers. Because higher carbon number products form, because they form only in the presence of OH radical, and because they only form at high organic concentrations, we hypothesized that these products form through radical-radical reactions.
To investigate this possibility, we expanded the glyoxal mechanism in the original Lim model to explicitly account for radical - radical reactions. Model performance for 30 μM experiments changed little; model performance was much improved for 3000 μM glyoxal experiments (Lim et al., ACP 2010). Specifically, the oxalic acid concentration dynamics were better captured, and the model captured the concentration dynamics of the most abundant quantified higher carbon number organic acid (tartaric acid). We ran this model for glyoxal concentrations from 10 μM to 10 M (OH radical concentrations of 10-12 M) and concluded that oxalate is the major SOA-forming product at cloud-relevant concentrations and oligomers are the major SOA products in wet aerosols. We expect OH radical chemistry will be the dominant daytime pathway for aqueous SOA formation from water-soluble aldehydes; our OH radical experiments and control experiments provide support for this.
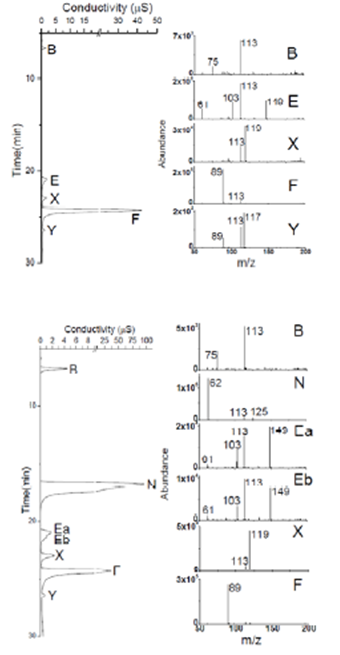
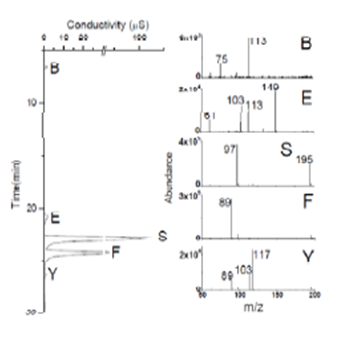
Figure 3. IC-ESI MS analysis of 40 min samples from (a)
glyoxal + OH radical experiments, (b) glyoxal + OH radical + HNO3
experiments, and (c) glyoxal + OH radical + (NH4)2SO4 experiments.
All experiments were conducted with 1 mM glyoxal and ~10-12 mM "
OH radical. With the exception of nitrate (peak N in figure 3b) and sulfate
(peak S in figure 3c), the mass spectra are quite similar, suggesting
that the addition of cloud-relavant concentratinos of (NH4)2SO4
or HNO3 has little effect on the in-cloud chemistry of glyoxal.
We previously proposed that oligomers forming from methylglyoxal and from pyruvic acid in the presence (but not absence) of OH radical were oligoesters formed via condensation reactions involving a hydroxy acid and one of several major organic acid products (Altieri et al., AE 2008). At that time, we proposed that hydroxy acids formed because succinic acid formed from the OH radical attack of acetic acid. While the formation of at least some succinic acid was confirmed by FT-ICR MS analysis of methylglyoxal experimental samples, analysis of acetic acid experiments by IC- ESI-MS showed that succinic acid was not a product of acetic acid plus OH radical (Tan et al., ACP 2011). A complex spectrum of high molecular weight products observed in methylglyoxal plus OH experiments and pyruvic acid plus OH experiments (but not controls) was not observed in acetic acid experiments conducted at identical concentrations (Tan et al., ACP 2011)(Figure 4).
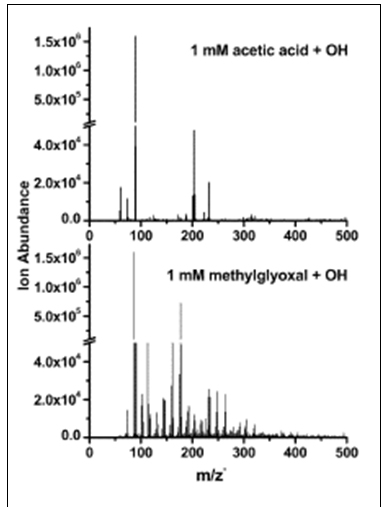
Figure 4. ESI mass spectra of 1 mM acetic acid + OH radical
experiment (120 min. reaction time) and 1 mM methylglyoxal + OH
radical experiment (120 min. reaction time).
These results, our new insights regarding the chemistry of glyoxal, and the formation of these products only in the presence of OH radical and not in mixtures containing a hydroxy acid (i.e., lactic acid), lead us to conclude that oligomers from methylglyoxal plus OH radical form through organic radical-radical reactions. This chemistry has now been elucidated and the original Lim model has been expanded to include organic radical-radical reactions and enable us to predict the behavior of glyoxal (Lim et al., 2010; Figure 5, 6) and methylglyoxal (Lim et al., 2012, in preparation; Figure 7,8) in the presence of OH radical in wet aerosols and clouds. We have used this chemistry to derive SOA yields for use in regional and global chemical transport models (Lim et al., 2012, in preparation). Predicted SOA molar yields are 77% (glyoxal) and 63% (methylglyoxal) for clouds and 40% (glyoxal and methylglyoxal) for wet aerosols (Lim et al., in preparation) (Figure 9). Oligomers are the major SOA products in wet aerosols, and small organic acids (oxalate, pyruvate) are the main SOA-forming products in clouds in the presence of OH radical. Acetic acid also is an aqueous SOA precursor, important because of its high atmospheric concentrations, but its OH radical oxidation does not lead to oligomer formation (Tan et al., ACP 2011). This chemistry, condensed versions of this chemistry, or these yields can be used in regional and global chemical transport models.
Sulfur-containing organic products were seen in the ultra high-resolution FT-ICR MS analyses of samples containing inorganic constituents but not in controls. These might be important products in wet aerosols. To investigate this possibility, we developed a model to investigate the radical chemistry of glycolaldehyde, OH radical and H2SO4. The model suggests that organosulfates could be a measureable product (as high as 10-30% of the total organic carbon) in wet acidic aerosols, but that they form in negligible quantities at concentrations found in clouds (less than 1% of organic carbon; Perri et al., AE 2010). Samples from 1 mM glyoxal experiments conducted in the presence of nitrogen (HNO3, (NH4)2SO4) have identical IC-ESI mass spectra to those conducted in the absence of nitrogen, and the concentrations of major products were not affected by the addition of nitrogen (Kirkland et al., in preparation). This suggests that the addition of cloud- relevant concentrations of nitrate and ammonium had a negligible effect on the formation of SOA-forming products. While others have observed the formation of nitrogen-containing organics in high concentration aldehyde experiments with ammonium sulfate in the absence of OH radical (Galloway et al., ACP 2009; Shapiro ACP 2009), we did not observe this in our OH radical experiments conducted at cloud-relevant and higher concentrations, even by ultra high-resolution FT-ICR MS.
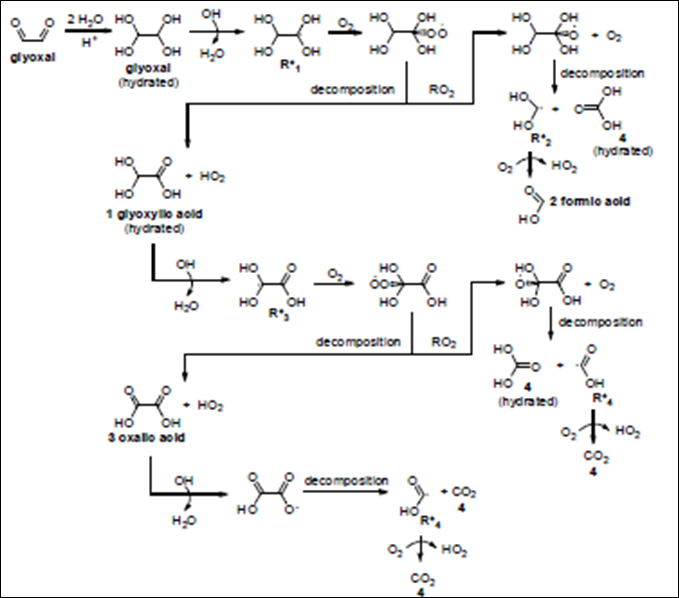
Figure 5. Glyoxal + OH radical. At low concentration, oxalate is the main product. At high concentration, organic
radical-radical reactions (e.g. R*1) become important, forming tartaric acid and other higher carbon number products,
as described in detail in Lim et al (2010). Using this mechanism, predictin of oxalic acid is much improved and
prediction of tartanic acid is possibel in the 3000 µM experiments.
radical-radical reactions (e.g. R*1) become important, forming tartaric acid and other higher carbon number products,
as described in detail in Lim et al (2010). Using this mechanism, predictin of oxalic acid is much improved and
prediction of tartanic acid is possibel in the 3000 µM experiments.
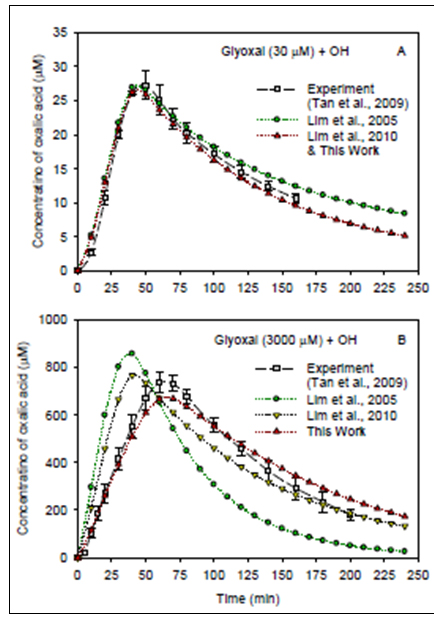
Figure 6. The real-time profiles of oxalic acid produced from the OH radical
reactions with glyoxal at initial concentrations of 30 µM(B). The oxalic acid
profiles include the experimental results (Tan et al., 2009) and previous/current
model simulations (Lim et al., 2005; Lim et al., 2010; Lim et al., 2012 in
preparation for this work).
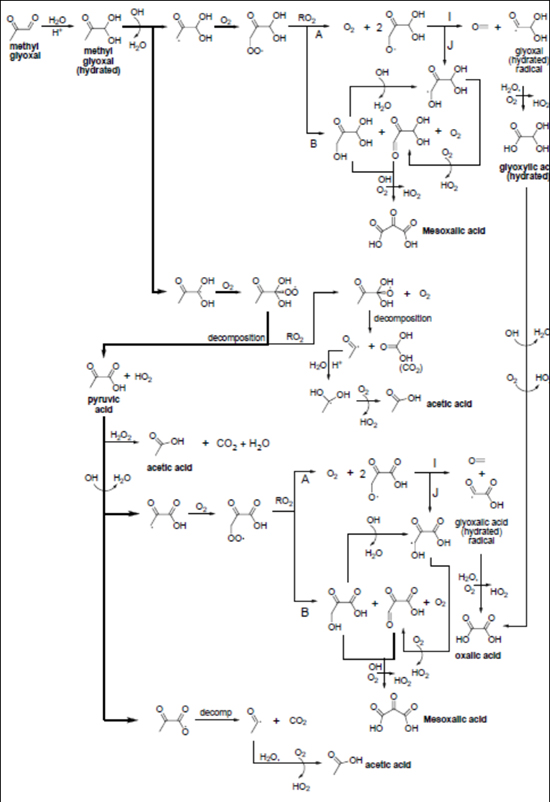
Figure 7. Aqueous methylglyoxal + OH radical reaction mechanism. Organic radical-radical
reactions become importan at high concentrations, allowing the formation of higher carbon number
products.
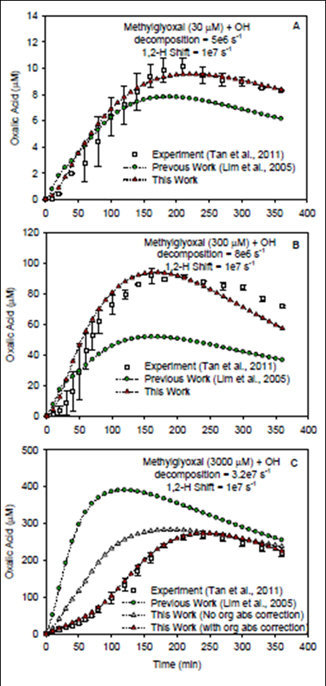
Figure 8. The real-time profiles of oxalic acid produced from
OH radical reactions of methylglyoxal at the concentrations
of 30 µM (A) and 300 µM (B) and 3000 µM (C). The oxalic
acid profiles include the experimental results (Tan et al., 2011)
, and previous/current model simulations (Lim et al., 2005;
Lim et al, 2012 in preparation - this work). In Fig. 8 (C), two
simulations were performed with/without organic (org) absorption
(abs) wiht an extinction coefficient (15000 cm-1M-1).
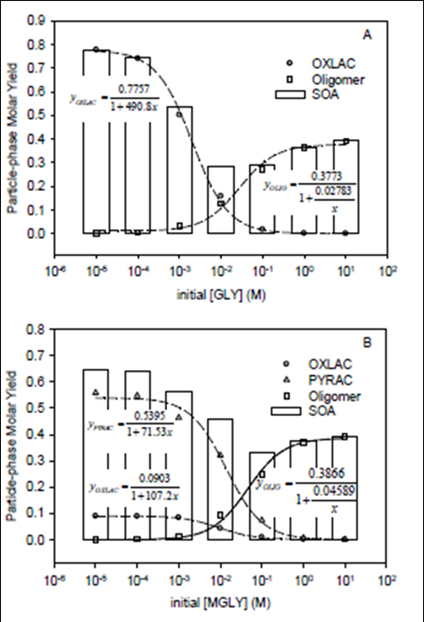
FIgure 9. (A) Simulated particle-phase molar yields of oxalic acid (YOXLAC),
Oligomers (YOLIG) and SOA (= YOXLAC + YOLIG) vs. initial concentration (X)
for aqueous-p[hase reactions of glyoxal and OH radical; and (B) for particle-
phase molar yields of oxalic acid (YOXALAC), pynruvic acid (YPYRAC) oligomers
(YOLIG) and SOA (= YOXLAC + YPYRAC + YOLIG) vs. initial concentration for (X)
for aqueous-phase reactions of methylglyoxal with OH radical. Concentrations
of 1-10 M provice insights into aldehyde chemistry in wet aerosols, where
concentrations of dissolvbed, water-soluable organics are quite high.
of 1-10 M provice insights into aldehyde chemistry in wet aerosols, where
concentrations of dissolvbed, water-soluable organics are quite high.
Objective 2: Based on the experiments, modeling and collaborative research made possible by this EPA STAR grant, we conclude that increased NOx results in higher yields of SOA from the aqueous chemistry of isoprene reaction products. Our experiments discussed above suggest that nitrate has little effect on the aqueous chemistry leading to SOA formation in clouds (Kirkland et al., in preparation). However, the yields of water soluble carbonyls from gas-phase isoprene chemistry are higher at high NOx (Ervens et al., GRL 2008). This suggests that more aqueous SOA will form from a biogenic hydrocarbon like isoprene in polluted conditions than in clean conditions. It should be noted also that the formation of aqueous SOA depends strongly on the atmospheric burden of liquid water in aerosols, fogs and clouds. This was elegantly demonstrated in the smog chamber experiments of Zhao et al. (AE 2011) and Kamens et al. (AE 2011).
Objective 3: Early in this grant, in-cloud production of SOA from glyoxal and methylglyoxal was added to the CMAQ model using a simple 4% molar yield based approach. The model was used to predict organic carbon (OC) concentrations measured on an airplane during the ICARTT experiment (Figure 10). The addition of cloud processing to CMAQ improved agreement between modeled OC and measured water-soluble organic carbon (WSOC) for all flights, most notably aloft. SOA formation through cloud processing was negligible in certain areas and substantial in others.
To further improve the treatment of cloud chemistry in CMAQ, a Rosenbrock solver (ROS3) has been incorporated into the full scale version of CMAQv4.7. The ROS3 version implements the same aqueous chemical mechanism as the base model, with the sole exception that gas-to-water partitioning is described kinetically, rather than assuming instantaneous thermodynamic equilibrium. Another version using the Kinetic Pre-Processor (KPP) is under development. The use of this solver enables easy calculation of the aqueous chemistry adjoint. Adjoint calculations are receptor based, and allow one to calculate the sensitivity of an output with respect to a large number of parameters. For example, we can compute the ozone concentration sensitivity with respect to changes in various grid species at earlier times (dO3/dNO2). The current solver (forward Euler within bisection) does not allow for an explicit adjoint calculation.
We are cooperating with several modelers (e.g., Kostas Tsigaridis, NASA-GISS; Mary Barth, WRF-Chem; Larry W. Horowitz, GFDL AM3; Annmarie Carlton, CMAQ) who are working toward adding SOA formation through aqueous chemistry into regional and global models, using the new knowledge gained through this research.
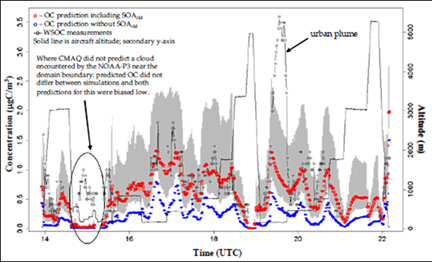
Figure 10. Results of CMAQ model wiht (red, uncertainties in grey) and
without (blue) SOA though cloud processing for Augusust 14th ICARTT flight.
Also shown is water-soluble organic carbon (open black circles_ and plane
altitude (black line). Inclusino of "aqueous" SOA improves agreement between
model and measurements.
altitude (black line). Inclusino of "aqueous" SOA improves agreement between
model and measurements.
Achievements With Respect to Stated Objectives: We met our stated objectives, which were focused around the OH radical chemistry in clouds and fogs. During the course of this research, it became clear that chemistry in wet aerosols can also yield secondary organic aerosol. As a result, we considerably expanded our research effort to understand how that chemistry differs from chemistry in clouds. We validated the OH radical oxidation pathways for glyoxal and methylglyoxal and used this chemistry to draw conclusions about SOA formation in clouds and wet aerosols.
Broader Impacts:
While there remain uncertainties regarding the magnitude of SOA formed through aqueous chemistry, considerable progress has been made toward understanding the aqueous chemistry underlying SOA formation and modeling it. This chemical understanding is quite important, given the ubiquitous presence of water in particles, clouds and fogs, which undoubtedly increases the SOA yields of some precursors and decreases the yields of others. Organic aerosols account for roughly half of fine particle mass globally, and a large fraction of this material is believed to be formed in the atmosphere because the ambient aerosol is much more oxidized than primary particulate emissions (Zhang et al., GRL 2007). Organics play an important role in new particle formation (Smith et al., 2008). Their properties and atmospheric behavior are greatly affected by their formation mechanism (Hallquist et al., 2009; Ervens et al., ACP 2011). This work improves the understanding of atmospheric chemistry, ultimately enabling improved prediction of atmospheric aerosol concentrations and composition from emissions. Such predictive models form the basis of air quality management plans.
Conclusions:
We hypothesized based on their atmospheric concentrations, Henry's Law constants and OH radical rate constants, that glyoxal, methylglyoxal, acetic acid and glycolaldehyde would be important precursors of aqueous SOA formation. We are now developing and testing a method to scrub water soluble gases from ambient air and conduct OH radical reaction experiments on this material. The main goal of this work is to determine what really are the most atmospherically important precursors of aqueous chemistry leading to SOA formation. We have succeeded collecting 120 mL aqueous samples containing 100-300 µM Carbon (i.e., water soluble gases) using 3 mist chambers in parallel. We have also succeeded in conducting glyoxal plus OH radical experiments in 10 3 mL cuvettes, enabling analysis of reaction products over 8 time points (2 in duplicate). Shortly, we will conduct OH radical reaction experiments with aqueous solutions of water-soluble gases scrubbed from New Jersey air. In the mean time, we have conducted OH radical experiments on filtered rainwater samples. We found that precursors were predominantly aldehydes, alcohols and/or organic peroxides. Products were predominantly organic acids. Many precursors and products were identified based on the change in their unit mass ESI-MS ion abundance (Figure 11). The oxidation of glyoxal, glycolaldehyde and methylglyoxal and formation of their identified products (i.e., pyruvic, glyoxylic, oxalic acids) is suggested by these experiments (Figure 12).
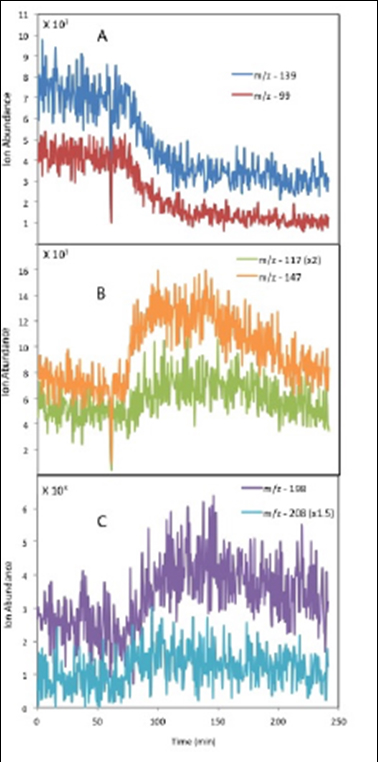
Figure 11. Examples of precursors (A) and products (B, C) seen in
the OH radical oxidation of filtered rainwater.
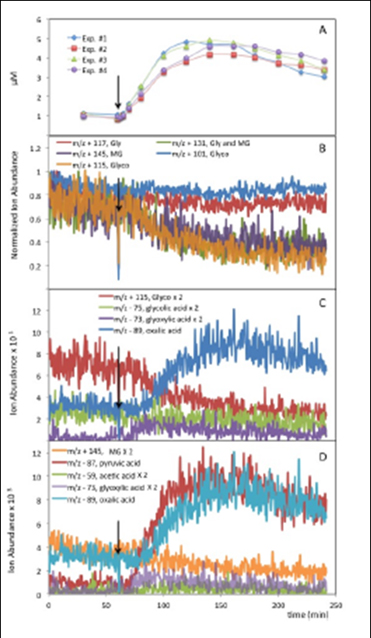
Figure 12. OH radical oxidations of filtered rainwater. (A) Oxalate
by IC for the four replicate experiments, (B) real-time ESI-MS
positive mode analysis of ions found in glyoxal (Gly), methylglyoxal
(MG) and glycolaldehyde (Glyco) standards, (C) ions expected to be
associated with the OH radical oxidation of glycoladehyde, and (D)
ions expected to be associated with the OH radical oxidation of
methylglyoxal. Figures C and D show positive mode (aldehudes) and
negative mode (acids) analysis. Teh black arrow shows when H2O2
and the UV were added and therefore OH radical reactions can begin.
{Evidence for methylgloxal oxidation is quite strong in the experiment
conducted with this rainwater sample.
References:
Altieri, K., Turpin, B. J., Seitzinger, S. P. (2009) The composition of dissolved organic nitrogen in continental precipitation investigated by ultra-high resolution FT-ICR mass spectrometry, Environ. Sci. Technol., 43:6950-6955.
Altieri, K. E., Turpin, B. J., Seitzinger, S. P. (2009) Oligomers, organosulfates, and nitrooxy organosulfates in rainwater identified by ultra-high resolution electrospray ionization FT-ICR mass spectrometry, Atmos. Chem. Phys., 9:2533-2542.
Altieri, K. E., Seitzinger, S. P., Carlton, A. G., Turpin, B. J., Klein, G. C., Marshall, A. G. (2008) Oligomers formed through in-cloud methylglyoxal reactions: Chemical composition, properties, and mechanisms investigated by ultra-high resolution FT-ICR Mass Spectrometry, Atmos. Environ., 42:1476-1490.
Blando, J. D. and Turpin, B. J. (2000) Secondary Organic Aerosol Formation in Cloud and Fog Droplets: A Literature Evaluation of Plausibility, Atmos. Environ. 34:1623-1632.
Carlton, A. G., Turpin, B. J., Altieri, K., Seitzinger, S., Mathur, R., Roselle, S. (2008) CMAQ model performance enhanced when in-cloud secondary organic aerosol is included: Comparisons of organic carbon predictions with measurements, Environ. Sci. Technol., 42:8798-8802.
Ervens, B, Carlton, AG, Turpin, BJ, Altieri, KE, Kreidenweis, SM, Feingold, G (2008) Secondary organic aerosol yields from cloud-processing of isoprene oxidation products, Geophys. Res. Lett. 35: L02816, doi:10.1029/2007GL031828.
Ervens, B., Turpin, B. J., and Weber, R. J. (2011) Secondary organic aerosol formation in cloud droplets and aqueous particles (aqSOA): A review of laboratory, field and model studies, Atmos. Chem. Phys., 11, 11069-11102.
Fu, T., Jacob, D. J., Wittrock, F., Burrows, J. P., Vrekoussis, M., and Henze, D. K. (2008) Global budgets of atmospheric glyoxal, methylglyoxal, and implications for formation of secondary organic aerosol, J. Geophys. Res., 113, D15303, doi:10.1029/2007JD009505.
Fu, T., Jacob, D. J., and Heald, C. L. (2009) Aqueous-phase reactive uptake of dicarbonyls as a source of organic aerosol over eastern North America, Atmos. Environ., 43, 1814-1822.
Galloway, M. M., Chhabra, P. S., Chan, A. W. H., Surratt, J. D., Flagan, R. C., Seinfeld, J. H., and Keutsch, F. N. (2009) Glyoxal uptake on ammonium sulphate seed aerosol: reaction products and reversibility of uptake under dark and irradiated conditions, Atmos. Chem. Phys., 9, 3331-3345, doi:10.5194/acp-9-3331-2009.
Gong, W., Stroud, C. and Zhang, L. (2011) Cloud processing of gases and aerosols in air quality modeling, Atmosphere 2, 567-616; doi:10.3390/atmos2040567.
Grgic, I., Nieto-Gligorovski, L. I., Net, S., Temime-Roussel, B., Gligorovski, S., and Wortham, H. (2010) Light induced multiphase chemistry of gas-phase ozone on aqueous pyruvic and oxalic acids, Phys. Chem. Chem. Phys., 12, 698-707.
Hallquist, M., Wenger, J. C., Baltensperger, U., Rudich, Y., Simpson, D., Claeys, M., Dommen, J., Donahue, N. M., George, C., Goldstein, A. H., Hamilton, J. F., Herrmann, H., Hoffmann, T., Iinuma, Y., Jang, M., Jenkin, M. E., Jimenez, J. L., Kiendler-Scharr, A., Maenhaut, W., McFiggans, G., Mentel, T. F., Monod, A., Prevot, A. S. H., Seinfeld, J. H., Surratt, J. D., Szmigielski, R., and Wildt, J. (2009) The formation, properties and impact of secondary organic aerosol: current and emerging issues, Atmos. Chem. Phys., 9, 5155-5236.
Hennigan, C. J., Bergin, M. H., Dibb, J. E., and Weber, R. J. (2008) Enhanced secondary organic aerosol formation due to water uptake by fine particles, Geophys. Res. Lett., 35, L18801, doi:10.1029/2008GL035046.
Hennigan, C. J., Bergin, M. H., Russell, A. G., Nenes, A., and Weber, R. J. (2009) Gas/particle partitioning of water-soluble organic aerosol in Atlanta, Atmos. Chem. Phys., 9, 3613-3628, doi:10.5194/acp-9-3613-2009.
Kamens, R. M., Zhang, H., Chen, E. H., Zhou, Y., Parikh, H. M., Wilson, R. L., Galloway, K. E. and Rosen, E. P. (2011) Secondary organic aerosol formation from toluene in an atmospheric hydrocarbon mixture: Water and particle seed effects, Atmos. Environ. 45, 2324-2334.
Kirkland, J.R., Lim, Y. B., Tan, Y., Altieri, K.E., Seitzinger, S.P. and Turpin,* B.J. (2012) Aqueous glyoxal photooxidation in the presence of inorganic nitrogen, in preparation.
Kiss, G., Tombacz, E., Varga, B., Alsberg, T., and Persson, L. (2003) Estimation of the average molecular weight of humic-like substances isolated from fine atmospheric aerosol, Atmos. Environ. 37, 3783-3794.
Liao, H., and Seinfeld, J. H. (2005) Global impacts of gas-phase chemistry aerosol interactions on direct radiative forcing by anthropogenic aerosols and ozone, J. Geophys. Res., 110, D18208, doi:10.1029/2005JD005907.
Lim, Y. B., Tan, Y., Perri, M. J., Seitzinger, S. P., Turpin, B. J. (2010) Aqueous chemistry and its role in secondary organic aerosol (SOA) formation, Atmos. Chem. Phys, 10, 10521-10539, doi:10.5194/acp-10-10521-2010.
Lim, Y. B., Tan, Y., Seitzinger, S. P, Turpin, B. J. (2012) Secondary organic aerosol formation via aqueous chemistry of glyoxal and methylglyoxal: Chemical insights, kinetic model studies and yields, in preparation for Atmos. Chem. Phys.
Lin, P., Huang, X.-F., He, L.-Y., and Zhen Yu, J. (2010) Abundance and size distribution of HULIS in ambient aerosols at a rural site in South China, J. Aerosol Sci., 41, 74-87.
Loeffler, K. W., Koehler, C. A., Paul, N. M., and de Haan, D. O. (2006). Oligomer Formation in Evaporating Aqueous Glyoxal and Methyl Glyoxal Solutions. Environ. Sci. Technol. 40:6318-6323.
Myriokefalitakis, S., Tsigaridis, K., Mihalopoulos, N., Sciare, J., Nenes, A., Kawamura, K., Segers, A., and Kanakidou, M. (2011) In-cloud oxalate formation in the global troposphere: A 3-d modeling study, Atmos. Chem. Phys., 11, 5761-5782, 10.5194/acp-11-5761-2011.
Noziere, B., Dziedzic, P., and Cordova, A. (2009) Products and kinetics of the liquid-phase of glyoxal catalyzed by ammonium ions (NH+4), J. Phys. Chem. A., 113, 231-237.
Ortiz-Montalvo, D. L., Lim, Y. B., Perri, M. J., Seitzinger, S. P., Turpin, B. J. (2012) Volatility and yield of glycolaldehyde SOA formed through aqueous photochemistry and droplet evaporation, Aerosol Sci. Technol., submitted.
Perri, M., Lim, Y. B., Seitzinger, S. P., and Turpin, B. J. (2010) Organosulfates from glycoladehyde in aqueous aerosols and clouds: Laboratory studies, Atmos. Environ., 44, 2658-2664.
Perri, M., Seitzinger, S. P., Turpin, B. J. (2009) Secondary organic aerosol production from aqueous photooxidation of glycolaldehyde: Laboratory experiments, Atmos. Environ., 43:1487-1497.
Seitzinger, S. P. and Sanders, R. W. (1997) Contribution of dissolved organic nitrogen from rivers to estuarine eutrophication, Marine Ecology Progress Series, 159, 1-12.
Shapiro, E. L., Szprengiel, J., Sareen, N., Jen, C. N., Giordano, M. R., and McNeill, V. F. (2009) Light-absorbing secondary organic material formed by glyoxal in aqueous aerosol mimics, Atmos. Chem. Phys., 9, 22892300, doi:10.5194/acp-9-2289-2009.
Sorooshian, A., Murphy, S. M., Hersey, S., Bahreini, R., Jonsson, H., Flagan, R. C., and Seinfeld, J. H. (2010) Constraining the contribution of organic acids and AMS m/z 44 to the organic aerosol budget: On the importance of meteorology, aerosol hygroscopicity, and region, Geophys. Res. Lett., 37, L21807, doi:10.1029/2010gl044951.
Smith, J. N., Dunn, M. J., VanReken, T. M., Iida, K., Stolzenburg, M. R., McMurry, P. H., and Huey, L. G. (2008) Chemical composition of atmospheric nanoparticles formed from nucleation in Tecamac, Mexico: Evidence for an important role for organic species in nanoparticle growth, Geophys. Res. Lett., 35, L04808.
Tan, Y., Carlton, A. G., Seitzinger, S. P., and Turpin, B. J. (2010) SOA from methylglyoxal in clouds and wet aerosols: Measurement and prediction of key products, Atmos. Environ., 44, 5218-5226, doi:10.1016/j.atmosenv.2010.08.045.
Tan, Y., Perri, M. J., Seitzinger, S. P., Turpin, B. J. (2009) Effects of precursor concentration and acidic sulfate in aqueous glyoxal OH radical oxidation and implications for secondary organic aerosol, Environ. Sci. Technol., 43, 8105-8112, doi: 10.1021/es901742f.
Tan, Y., Lim, Y. B., Altieri, K. E., Seitzinger, S., Turpin, B. J.* (2012) Mechanisms leading to oligomers and SOA through aqueous photooxidation: Insights from OH radical oxidation of acetic acid and methylglyoxal, Atmos. Phys. Chem., 12, 801-813.
Yasmeen, F., Sauret, N., Gal, J.-F., Maria, P.-C., Massi, L., Maenhaut, W., and Claeys, M. (2010) Characterization of oligomers from methylglyoxal under dark conditions: a pathway to produce secondary organic aerosol through cloud processing during nighttime, Atmos. Chem. Phys., 10, 3803-3812.
Zappoli, S., Andracchio, A., Fuzzi, S., Facchini, M. C., Gelencse r, A., Kiss, G., Krivacsy, Z., Molnar, A., Meszaros, E., Hansson, H. C., Rosman, K., and Zebuhr, Y. (1999) Inorganic, organic and macro- molecular components of fine aerosol in different areas of Europe in relation to their water solubility, Atmos. Environ., 33, 2733-2743.
Zhang, Q., Jimenez, J. L., Canagaratna, M. R., Allan, J. D., Coe, H., Ulbrich, I., Alfarra, M. R., Takami, A., Middlebrook, A. M., Sun, Y. L., Dzepina, K., Dunlea, E., Docherty, K., DeCarlo, P. F., Salcedo, D., Onasch, T., Jayne, J. T., Miyoshi, T., Shimono, A., Hatakeyama, S., Takegawa, N., Kondo, Y., Schneider, J., Drewnick, F., Weimer, S., Demerjian, K., Williams, P., Bower, K., Bahreini, R., Cottrell, L., Griffin, R. J., Rautiainen, J., and Worsnop, D. R. (2007) Ubiquity and dominance of oxygenated species in organic aerosols in anthropogenically influenced northern hemisphere mid-latitudes, Geophys. Res. Lett., 34, L13801, doi:10.1029/2007GL029979.
Zhou, Y., Zhang, H., Parikh, H. M., Chen, E. H., Rattanavaraha, W., Rosen, E. P., Wang, W., Kamens, R. M. (2011) Secondary organic aerosol formation from xylenes and mixtures of toluene and xylenes in an atmospheric urban hydrocarbon mixture: Water and particle seed effects (II), Atmos. Environ. 45, 3882-3890.
Journal Articles on this Report : 15 Displayed | Download in RIS Format
| Other project views: | All 46 publications | 16 publications in selected types | All 16 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Altieri KE, Seitzinger SP, Carlton AG, Turpin BJ, Klein GC, Marshall AG. Oligomers formed through in-cloud methylglyoxal reactions: chemical composition, properties, and mechanisms investigated by ultra-high resolution FT-ICR mass spectrometry. Atmospheric Environment 2008;42(7):1476-1490. |
R833751 (2008) R833751 (2009) R833751 (2010) R833751 (Final) R831073 (Final) |
Exit Exit Exit |
|
|
Altieri KE, Turpin BJ, Seitzinger SP. Oligomers, organosulfates, and nitrooxy organosulfates in rainwater identified by ultra-high resolution electrospray ionization FT-ICR mass spectrometry. Atmospheric Chemistry and Physics 2009;9(7):2533-2542. |
R833751 (2009) R833751 (Final) |
Exit Exit |
|
|
Altieri KE, Turpin BJ, Seitzinger SP. Composition of dissolved organic nitrogen in continental precipitation investigated by ultra-high resolution FT-ICR mass spectrometry. Environmental Science & Technology 2009;43(18):6950-6955. |
R833751 (2009) R833751 (2010) R833751 (Final) |
Exit Exit Exit |
|
|
Carlton AG, Turpin BJ, Altieri KE, Seitzinger SP, Mathur R, Roselle SJ, Weber RJ. CMAQ model performance enhanced when in-cloud secondary organic aerosol is included:comparisons of organic carbon predictions with measurements. Environmental Science & Technology 2008;42(23):8798-8802. |
R833751 (2008) R833751 (2009) R833751 (2010) R833751 (Final) R831073 (Final) |
Exit Exit Exit |
|
|
Ervens B, Carlton AG, Turpin BJ, Altieri KE, Kreidenweis SM, Feingold G. Secondary organic aerosol yields from cloud-processing of isoprene oxidation products. Geophysical Research Letters 2008;35(2):L02816 (5 pp.). |
R833751 (2008) R833751 (2009) R833751 (2010) R833751 (Final) R831073 (Final) |
Exit Exit |
|
|
Ervens B, Turpin BJ, Weber RJ. Secondary organic aerosol formation in cloud droplets and aqueous particles (aqSOA):a review of laboratory, field and model studies. Atmospheric Chemistry and Physics 2011;11(21):11069-11102. |
R833751 (Final) |
Exit Exit |
|
|
Kirkland JR, Lim YB, Tan Y, Altieri KE, Turpin BJ. Glyoxal secondary organic aerosol chemistry: effects of dilute nitrate and ammonium and support for organic radical-radical oligomer formation. Environmental Chemistry 2013;10(3):158-166. |
R833751 (Final) |
Exit Exit Exit |
|
|
Lim YB, Tan Y, Perri MJ, Seitzinger SP, Turpin BJ. Aqueous chemistry and its role in secondary organic aerosol (SOA) formation. Atmospheric Chemistry and Physics 2010;10(21):10521-10539. |
R833751 (2010) R833751 (Final) |
Exit Exit |
|
|
Lim YB, Tan Y, Turpin BJ. Chemical insights, explicit chemistry, and yields of secondary organic aerosol from OH radical oxidation of methylglyoxal and glyoxal in the aqueous phase. Atmospheric Chemistry and Physics 2013;13(17):8651-8667. |
R833751 (Final) |
Exit Exit |
|
|
Ortiz-Montalvo DL, Lim YB, Perri MJ, Seitzinger SP, Turpin BJ. Volatility and yield of glycolaldehyde SOA formed through aqueous photochemistry and droplet evaporation. Aerosol Science and Technology 2012;46(9):1002-1014. |
R833751 (Final) |
Exit Exit Exit |
|
|
Ortiz-Montalvo DL, Häkkinen SA, Schwier AN, Lim YB, McNeill VF, Turpin BJ. Ammonium addition (and aerosol pH) has a dramatic impact on the volatility and yield of glyoxal secondary organic aerosol. Environmental Science & Technology 2014;48(1):255-262. |
R833751 (Final) |
Exit Exit Exit |
|
|
Ortiz-Montalvo DL, Schwier AN, Lim YB, McNeill VF, Turpin BJ. Volatility of methylglyoxal cloud SOA formed through OH radical oxidation and droplet evaporation. Atmospheric Environment 2016;130:145-152. |
R833751 (Final) |
Exit Exit Exit |
|
|
Tan Y, Perri MJ, Seitzinger SP, Turpin BJ. Effects of precursor concentration and acidic sulfate in aqueous glyoxal–OH radical oxidation and implications for secondary organic aerosol. Environmental Science & Technology 2009;43(21):8105-8112. |
R833751 (2009) R833751 (2010) R833751 (Final) |
Exit Exit Exit |
|
|
Tan Y, Carlton AG, Seitzinger SP, Turpin BJ. SOA from methylglyoxal in clouds and wet aerosols: measurement and prediction of key products. Atmospheric Environment 2010;44(39):5218-5226. |
R833751 (2010) R833751 (Final) |
Exit Exit Exit |
|
|
Tan Y, Lim YB, Altieri KE, Seitzinger SP, Turpin BJ. Mechanisms leading to oligomers and SOA through aqueous photooxidation: insights from OH radical oxidation of acetic acid and methylglyoxal. Atmospheric Chemistry and Physics 2012;12(2):801-813. |
R833751 (Final) |
Exit Exit |
Supplemental Keywords:
SOA, secondary organic aerosol, PM2.5, cloud processing, isoprene, ambient airProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
