Grantee Research Project Results
Final Report: A Rapid In Vivo System for Determining Toxicity of Manufactured Nanomaterials
EPA Grant Number: R833320Title: A Rapid In Vivo System for Determining Toxicity of Manufactured Nanomaterials
Investigators: Tanguay, Robyn L. , Harper, Stacey
Institution: Oregon State University
EPA Project Officer: Hahn, Intaek
Project Period: September 1, 2006 through August 30, 2009
Project Amount: $400,000
RFA: Exploratory Research: Nanotechnology Research Grants Investigating Environmental and Human Health Effects of Manufactured Nanomaterials: a Joint Research Solicitation-EPA, NSF, NIOSH, NIEHS (2006) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
Rapid growth of the nanotechnology industry will result in increased humans and the environment exposures to nanomaterials. It is clear that there is a need to develop rapid, relevant and efficient testing strategies to assess these emerging materials of concern. It is expected that the biological activity of nanomaterials will depend on inherent physicochemical properties not routinely considered in toxicity studies (e.g. particle size and size distribution, agglomeration status, interactions with environmental and biological moieties). Here we used a powerful approach that utilizes a dynamic whole animal assay to reveal whether a nanomaterial poses hazard. It is anticipated that the biological response will be dependent on toxicokinetic properties so we propose that those parameters be determined following a “hit” in our in vivo assay. A toxic “hit” in our assay would warrant further testing in other models.
Our hypothesis is that inherent properties of some engineered nanomaterials make them toxic. To test this hypothesis we specifically proposed to: 1) further develop our zebrafish toxicity assay to define in vivo responses to nanomaterials, 2) define structural properties of nanomaterials that lead to adverse biological consequences, and 3) develop a Nanomaterials Effects Database to integrate nanotoxicity data.
We originally proposed a three-tier approach that exploits the advantages of the embryonic zebrafish model to assess the toxicity of nanomaterials. Tier 1: Rapid screening experiments will be conducted to assess the toxicity of a wide range of structurally well-characterized nanomaterials commercially available or produced by researchers of the Oregon Nanoscience and Microtechnologies Institute (ONAMI). Nanomaterials found to elicit significant adverse effects will proceed to Tier 2 testing. Tier 2: Potential cellular targets and modes of action will be defined in vivo using a suite of transgenic fluorescent zebrafish and indicators of cellular oxidative state. Nanomaterials will be grouped according to structural indices and effects. Representative nanomaterials from each group will be selected for Tier 3 testing. Tier 3: Global gene expression profiles will be used to define the genomic responses to nanomaterials. Data from these studies will be used to define structure-activity relationships using a Nanomaterials Effects Database we have created to collate, organize and analyze data on nanomaterial effects across species and exposure scenarios.
Summary/Accomplishments (Outputs/Outcomes):
Objective 1: To further develop our zebrafish toxicity assay to define in vivo responses to nanomaterials. Toxicity of engineered nanomaterials will be determined by investigating the biological consequences of embryonic exposure in zebrafish.
The embryonic toxicity assay has been refined to be a model experimental platform for testing in vivo biological responses to nanomaterials. Our tier testing approach has helped define the biological activity and toxic potential of nanomaterials. The screening level (tier 1) has been streamlined to assess 16 animals per a set of standard concentrations (0.4, 2, 10, 50 µg/mL) of nanoparticles, with two sets of controls (positive: to ensure animals are sensitive to stressors, and a negative control: to ensure the animals were of good quality). Automated liquid handling has also been implemented into the screening process to improve efficiency and accuracy of nanoparticle delivery.
In addition to assessing the overt toxicity of nanoparticles, we have included assessments to evaluate behavioral responses following exposures. This assessment measures motor activity levels induced by a change in lighting. Prior to evaluation, embryos are acclimated to the light for 20 minutes, after which the lights are turned off (dark period) for 10 minutes, and then on for five minutes (light period). The raw output data files are then processed using a perl script that we have written to average total pixel changes per second for the dark period. We have scaled this assay to measure up to 500 animals simultaneously.
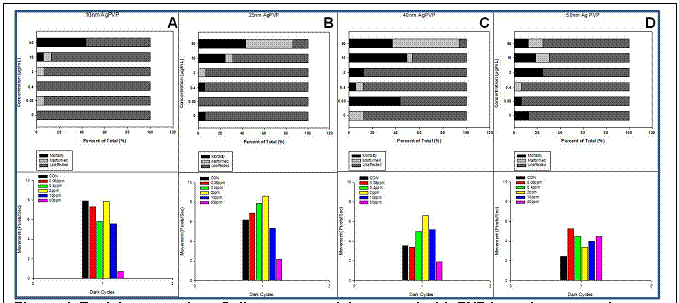
At present, we have evaluated over 130 ‘types’ of nanomaterials through this newly refined screening level (See Appendix for detailed list). These nanomaterials include a fifteen member gold library, copper and cobolt coated nanoparticles, a series of dendrimers, single-walled and multi-walled carbon nanotubes, 11 commercially available silver nanoparticles, PbS semiconductor nanoparticles, various zinc oxide, carboxylated and sulfonated nanocellulose nanotubes, a series of titanium oxide nanotubes, and various surface coated silver, carbon based fullerenes C60 and titanium dioxide nanoparticles. Figure 1 is a representative of the data generated from a size series of silver nanoparticles coated in polyvinylpyrrolidone (PVP).
Figure 1. Toxicity screening of silver nanoparticles coated wiht PVP in various core size. Embryos were dechorionated
and exposed to five concentrations of 10nm (a), 25nm (b), 40nm (c), and 50 nm (d) Ag-PHP at 6 hours postfertilization (HPF).
At 120 hpf, exposed embryos are evaluated for mortality and motbidity, in addition to assessing behavioral abnormalities.
Objective 2: Define structural properties of nanomaterials that lead to adverse biological consequences. Groups of congeneric structures (i.e. gold and dendrimers) will be tested, in addition to a wide range of other nanomaterial types (i.e. polymeric, semi-conductive and carbon-based). Data obtained will be used to develop generalizations about how size, surface chemistry and exposure concentrations relate to biological responses.
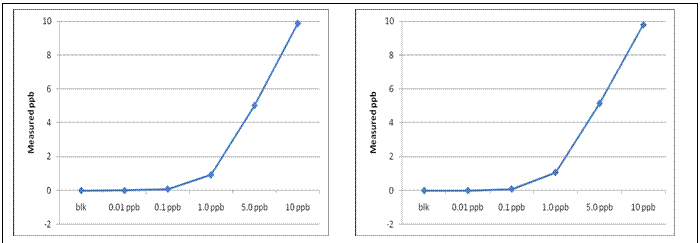
Nanomaterials that induced adverse biological responses in the screening level (tier 1) were further investigated to identify potential cellular targets and mode of action (tier 2). We first recognized the need to measure nanomaterial uptake in zebrafish. Previously, we utilized instrument neutron activation analysis (INAA); however, due to its inability to measure metals without gamma radioactive isotopes (such as lead), the requirement of a radiation center and the relatively high cost of the analysis, it made it difficult to be used as a routine instrumentation. As an alternative, we focused on determining the level of detection using Inductively Coupled Plasma – Mass Spectrometry (ICP-MS). As a proof of concept, we measured both gold and lead using ICP-MS. ICP-MS is able to detect these metals in the 10 ppt for gold and 20 ppt for lead (figure 2), both are with a 5% accuracy. This method has been applied to estimate total embryonic NP load following exposure to a series of metal-cored NPs.
Figure 2. (Left) Lead ICP-MS Calibration Curve. (Right) Gold ICP-Calibration Curve.
The zebrafish model can be effectively used to identify NPs that alter in vivo cellular death. To determine whether gold nanoparticles caused an increase in cellular death, embryos were exposed to test materials such as the AuNPs, then incubated with acridine orange for 40 minutes. Embryos are then immediately euthanized and mounted on a cover slide for 11 slice imaging of the caudal fin (Zeiss – Axiovert Z-stack). This approach is reproducible; however, very time-consuming taking up to 30 minutes to analyze a single animal. To overcome this limitation we developed and optimized a fluorescent plate reader-based assay. This plate-based approach makes the assay scalable to rapid throughput experiments where 96 animals can be analyzed simultaneously in less than 20 minutes. With proof of concept in place, we have now implemented this assay into tier 2.
Using a series of nanomaterials that differ in one aspect (surface functionalization); we are able to test the effects of isolated changes on biological activity. For example, lead sulfide (PbS) nanoparticles conjugated with monothiol (PbS-MT) produced significant adverse responses while the dithiol conjugated to PbS (PbS-DT) was less toxic (Truong et al., 2010). These differential responses were not directly attributed to the ligands (data not shown here). We compared the responses of the two nanoparticles (PbS-MT and PbS-DT) to lead nitrate (PbNO3) to identify if the differential responses could be attributed the lead core. The effects observed by the PbS NPs were consistent with the morbidity induced by free lead. To determine if the reduced toxicity of the PbS-DT could be explained by reduced PbS-DT uptake relative to the PbS-MT, tissue lead concentrations were quantified using ICP-M. Embryos were exposed to a broad range of concentrations of PbS-DT, PbS-MT and PbNO3. Samples were collected at two time points (12 and 24 hpf). In general, lead (regardless of form) was quantitatively detected in animals exposed to PbS-DT at both developmental time points. It is noteworthy that at each exposure concentration, the lead levels were consistently higher in the PbS-DT exposed groups than in the PbS-MT exposed groups. Therefore, since appreciable levels of lead are detected in the in PbS-DT exposed embryos, the lack of material uptake cannot explain the reduced toxicity of the PbS-DTS.
With the knowledge that at 24 hpf, more lead is internalized with PbS-DT exposed embryos compared to PbNO3, and PbS-MT, cellular death frequency was quantified using a plate based method where the fluorescence detected for each PbS NP was compared to water soluble lead (PbNO3) and a ratio was calculated to determine the cellular death frequency induced by the nanoparticle. Both thiol capped lead sulfide nanoparticles produced a significant increase in cellular death compared to non exposed animals. To further investigate the differential responses, the nanoparticle solutions were evaluated at two time points to determine the lead concentration using ICP- OES. Both PbS-MT and PbS-DT, solutions consist of relatively similar concentration of lead. Both nanoparticle solutions destabilized and precipitated five days after oxidation where clear supernatant consist of ionic lead. Supernatant measurements revealed that less solubilized lead in the PbS-DT supernatant compared to the PbS-MT. This demonstrates that PbS-DT nanoparticles are more stable than the PbS-MT. The differential biological response observed for monothiol vs dithiol capped PbS nanoparticles could be explained by the different nanoparticle stability and the propensity for the lead core material to leach from the less stable (PbS-MT) NP.
We also tested two gold nanoparticles with the same core size (1.5nm) and with two different surface group [positive charge = N,N,N-trimethylammoniumethanethiol (TMAT), and negative charge = 2-mercaptoethanesulfonate (MES)] (Truong et al., in press). We found that the positively charged nanoparticle produced embryo lethality, while the negatively charged induced a wide range of sublethal resposnes at the same concentration. To determine if differential particle uptake could explain these differential biological responses, tissue gold concentration were quantified at two different developmental time points. Embryos were exposed to five-fold serial dilution of 0-250 µg/mL and samples were collected at 24 and 48 hpf. In general, gold (regardless of form) was quantitatively detected in animals at both developmental time points. There was a dose-dependent relationship and a relatively consistent level of gold delivered. Therefore, we conclude that differential particle uptake cannot explain the differential toxicity. Since there were similar levels of gold taken up into embryos regardless of surface functionalization we next wanted to determine if exposure to the AuNP differentially lead to in vivo cellular death. Embryos were exposed to 10 µg/mL of 1.5nm TMAT, and 50 µg/mL for 1.5nm MES. A plate-based method was used to quantify cellular death in the whole animal at both 24 and 48 hours post fertilization. 1.5nm MES, and TMAT had a protective effect on exposed embryos, and this response declined at 48 hpf. Therefore, it has become evident that the cellular death response does not directly correlate with tissue concentration or to the incident of adverse biological responses.
These two surface functionalize gold nanoparticles became ideal to investigate the global gene expression pattern (tier 3). NimbleGen whole genome microarrays were used. Embryos were dechorionated and exposed from 6 to either 24 or 48 hpf in three biological replicates. The transcripts that were misexpressed relative to controls for both treatments and at both time points are depicted as heat maps (data not shown) and venn diagram illustrates the number of differential expressed transcripts at 24 and 48 hpf for both of the 2 treatments. MES functionalized AuNPs misregulated more transcripts at both 24 and 48 hpf (35 to 497, respectively) than TMAT functionalized (29 to 239). To investigate the role of these misregulated transcripts, we used pathway analysis to begin to dissect the differential pathways impacted by the functionalized AuNPs. We found that for MES functionalized AuNPs, at both time points, the top five most significantly impacted biological functions were cellular and embryonic development, genetic disorder, cellular death and cellular assembly and organization. TMAT functionalized AuNPs impacted cancer, cardiovascular system development and function, cell morphology, cell-to-cell signaling and interactions and cellular compromise. This pathway analysis on the misregulated transcripts suggest that the differential toxic responses observed is mediated by two modes of actions. The pathway analysis suggests that MES-AuNPs disrupts cellular and embryonic development leading to future system failures, while TMAT-AuNPs causes uncontrolled development and cellular promise, resulting in immediate system shut down, which ultimately leads to embryo lethality. Collectively, this gene expression data provides us the stepping-stone necessary to identify key structure-activity relationships to design safer nanomaterials.
Objective 3: Develop a Nanomaterials Effects Database to serve as a repository for annotated data and computational tools for comparison across species and among disparate exposure scenarios.
The Nanomaterial Effects Database has been renamed to the Nanomaterial-Biological Interactions knowledgebase to more accurately represent the type of data integration systems that we envision. A website has been established at Oregon State University (OSU) as a portal for the NBI knowledgebase (www.oregonstate.edu/nbi  ) housed at OSU. This website was utilized recently when formulating a collaborative Informatics Science Project proposal for the NanoHealth Enterprise.
) housed at OSU. This website was utilized recently when formulating a collaborative Informatics Science Project proposal for the NanoHealth Enterprise.
Mathematical metrics for evaluating nanomaterial-biological interactions using the embryonic zebrafish experimental platform (Embryonic Zebrafish Metrics for Nanomaterial Toxicity, EZ-Metric) were derived from primary data. Such data compression allows for a single metric representative of the morbidity and mortality elicited from exposure. Briefly, control and nanomaterial-exposed groups are statistically compared using proportionality analysis. For each exposure concentration, the # of animals that exhibit morbidity and/or mortality is multiplied by a hierarchical ranking of effects and is then normalized to a common scale.
Dr. Stacey Harper, who was a former postdoc, is leading this effort as an independent Assistant Professor at Oregon State University. She has established significant collaborations with government, industry and academic developers of similar data/knowledgebases to ensure compatibility, develop standard ontologies and cross-searching capabilities, and promote data sharing and future planning for an integrated, federated network of knowledgebase systems. Integral members of the nanobioinformatics community that have been engaged include InterNano: Nanomanufacturing Information Clearinghouse of the University of Massachusettes, Amherst (Mark Tuominen and Rebecca Reznik-Zellen), the EPA Exposure and Dose Research Branch (Curtis Dary), the National Center for Computational Toxicology (Bob Kavlock and Jerry Blancato), the Nanotechnology Characterization Laboratory of the National Cancer Institute (Marty Fritts), the Nanoparticle Information Library (NIL) of the National Institute for Occupational Safety and Health (NIOSH) (Art Miller and Mark Hoover), and the Advanced Biomedical Computing Center of the National Cancer Institute (Raul Cachau).
Conclusions:
Defined Measures of Success
- One measure of success will be the completion of screening the nanomaterials proposed herein. Novel data generated in the screening portion of these studies will be an important first step at developing generalizations about the effects of structural indices on toxicity. We have made significant progress toward screening nanomaterials and identifying their relative toxic potential using the embryonic zebrafish model.
- A second measure of success will be the validation of the embryonic zebrafish model for evaluating the toxicity of nanomaterials in vivo. This entails the capability to discern between toxic and non-toxic nanomaterials based on effects. We have established the embryonic zebrafish model as an ideal biological platform in which to rapidly interrogate the biological activity and toxic potential of nanomaterials.
- The Nanomaterial-Biological Interactions knowledgebase will be established through collaborations with industry, academia and government researchers. We will consider this effort successful when the NBI is functional and the dynamics of the dose-response can be probed statistically with confidence. We have the foundational framework for determining dose (concentration)-responses to nanomaterials. The NBI system is operational and will soon be upgraded to include functional links to other databases on Nanomaterial EHS.
Journal Articles on this Report : 12 Displayed | Download in RIS Format
| Other project views: | All 41 publications | 12 publications in selected types | All 12 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Harper SL, Dahl JA, Maddux BLS, Tanguay RL, Hutchison JE. Proactively designing nanomaterials to enhance performance and minimise hazard. International Journal of Nanotechnology 2008;5(1):124-142. |
R833320 (2008) R833320 (Final) |
Exit |
|
|
Harper SL, Carriere JL, Miller JM, Hutchison JE, Maddux BLS, Tanguay RL. Systematic evaluation of nanomaterial toxicity: utility of standardized materials and rapid assays. ACS Nano 2011;5(6):4688-4697. |
R833320 (Final) |
Exit Exit Exit |
|
|
Harper S, Usenko C, Hutchison JE, Maddux BLS, Tanguay RL. In vivo biodistribution and toxicity depends on nanomaterial composition, size, surface functionalisation and route of exposure. Journal of Experimental Nanoscience 2008;3(3):195-206. |
R833320 (Final) |
Exit Exit |
|
|
Isaacson CW, Usenko CY, Tanguay RL, Field JA. Quantification of fullerenes by LC/ESI-MS and its application to in vivo toxicity assays. Analytical Chemistry 2007;79(23):9091-9097. |
R833320 (2008) R833320 (Final) |
Exit Exit Exit |
|
|
Thomas DG, Klaessig F, Harper SL, Fritts M, Hoover MD, Gaheen S, Stokes TH, Reznik-Zellen R, Freund ET, Klemm JD, Paik DS, Baker NA. Informatics and standards for nanomedicine technology. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology 2011;3(5):511-532. |
R833320 (Final) |
Exit |
|
|
Truong L, Moody IS, Stankus DP, Nason JA, Lonergan MC, Tanguay RL. Differential stability of lead sulfide nanoparticles influences biological responses in embryonic zebrafish. Archives of Toxicology 2011;85(7):787-798. |
R833320 (Final) |
Exit |
|
|
Truong L, Harper SL, Tanguay RL. Evaluation of embryotoxicity using the zebrafish model. Methods in Molecular Biology 2011;691(Part 6):271-279. |
R833320 (2008) R833320 (Final) |
Exit |
|
|
Truong L, Zaikova T, Richman EK, Hutchison JE, Tanguay RL. Media ionic strength impacts embryonic responses to engineered nanoparticle exposure. Nanotoxicology 2012;6(7):691-699. |
R833320 (Final) |
Exit |
|
|
Truong L, Saili KS, Miller JM, Hutchison JE, Tanguay RL. Persistent adult zebrafish behavioral deficits results from acute embryonic exposure to gold nanoparticles. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 2012;155(2):269-274. |
R833320 (Final) |
Exit Exit Exit |
|
|
Truong L, Tilton SC, Zaikova T, Richman E, Waters KM, Hutchison JE, Tanguay RL. Surface functionalities of gold nanoparticles impact embryonic gene expression responses. Nanotoxicology 2013;7(2):192-201. |
R833320 (Final) |
Exit |
|
|
Usenko CY, Harper SL, Tanguay RL. In vivo evaluation of carbon fullerene toxicity using embryonic zebrafish. Carbon 2007;45(9):1891-1898. |
R833320 (2008) R833320 (Final) |
Exit |
|
|
Usenko CY, Harper SL, Tanguay RL. Fullerene C60 exposure elicits an oxidative stress response in embryonic zebrafish. Toxicology and Applied Pharmacology 2008;229(1):44-55. |
R833320 (2008) R833320 (Final) |
Exit Exit Exit |
Supplemental Keywords:
dose-response, teratogen, animal, stressor, toxics, particulates, nanotechnology, nanotoxicology, environmental chemistry, Northwest, Oregon, OR, industry, scientific discipline, health, physical aspects, risk assessments, Health Risk Assessment, physical processes, biochemistry, nanotechnology, toxicity assay, human health risk, nanomaterials, nanoparticle toxicity, Health, Scientific Discipline, PHYSICAL ASPECTS, Health Risk Assessment, Physical Processes, Risk Assessments, Biochemistry, nanomaterials, toxicity assay, exposure, nanoparticle toxicity, nanotechnologyProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.