Grantee Research Project Results
Final Report: Transport/Fate/Ecological Effects of Steroids from Poultry Litter & Evaluations of Existing/Novel Management Strategies
EPA Grant Number: R833418Title: Transport/Fate/Ecological Effects of Steroids from Poultry Litter & Evaluations of Existing/Novel Management Strategies
Investigators: Fisher, Daniel J. , Kane, Andrew S. , Staver, Kenneth , Yonkos, Lance T. , VanVeld, Peter , Klauda, Ronald J.
Institution: Wye Research and Education Center , Maryland Department of Natural Resources , Virginia Institute of Marine Science , School of Medicine at the University of Maryland
EPA Project Officer: Aja, Hayley
Project Period: August 1, 2007 through June 30, 2010 (Extended to July 31, 2012)
Project Amount: $694,736
RFA: Fate and Effects of Hormones in Waste from Concentrated Animal Feeding Operations (CAFOS) (2006) RFA Text | Recipients Lists
Research Category: Endocrine Disruptors , Human Health , Safer Chemicals
Objective:
The objectives of the proposed study were to determine the environmental transport, fate and ecological effects of fecal sex steroids following agricultural field application of poultry manure. Previous studies at our Wye Research and Education Center (WREC) laboratory have demonstrated that: (1) fecal steroids in poultry manure reach receiving waters via rain-induced runoff; (2) this runoff is sufficiently estrogenic to feminize male fish; and (3) differences in agricultural management strategies can affect steroid concentrations in runoff and receiving waters.
Our work under the 2007 EPA CAFO research grants addressed remaining questions concerning environmental persistence and bioactivity of steroids upon reaching surface waters and further investigated effects of agronomic practices on mitigating resultant environmental steroid loads. This was accomplished using laboratory, controlled research field, and in situ watershed investigations. Steroids and estrogenicity in aqueous manure mixtures (lab-generated and field-collected) were monitored over time to determine degradation rates. In all analyses, particular attention was paid to determining proportions of fecal steroids, most importantly 17β-estradiol (E2) and estrone (E1). Fish were exposed to aqueous manure mixtures in laboratory assays to determine the effects of steroid degradation on bioactivity as measured by two vitellogenin (VTG) assays and two novel in-vitro estrogen-inducible reporter-gene assays.
A final multi-litter fish exposure was conducted to compare various analytical techniques for measuring estrogens and estrogenicity (GC/MS/MS, LC/MS/MS, UPLC/MS/MS, VTG blood plasma ELISA, VTG mRNA, KBLUC, and E-Screen). Effects of agricultural management practices on rates of transport of fecal steroids and nutrients to surface waters via rain-induced runoff were investigated using adjacent 33-acre research fields cropped variously under standard No-till practices of direct surface manure application, or by a novel Sub-surface Manure Injection (SS) technique and a currently used Turbo-Till® (TT) (Great Plains, Salina, KA) manure application practice. Turbo-Till is the name of the particular tillage equipment used at the WREC for this study. The genralized terminology for this type of tillage is “verticle tillage.” Finally, Maryland Biological Stream Survey (MBSS) protocols were applied to agriculturally influenced watersheds to assess community and population level disturbances resulting from fecal steroid exposure. Based on results from the early MBSS sampling effort, largemouth bass sampling for intersex (testicular oocytes) was conducted in lakes on the Delmarva Peninsula in Maryland and Delaware.
Summary/Accomplishments (Outputs/Outcomes):
Maryland Biological Stream Survey (MBSS)
| Creek/Run | Year | MD DNR MBSS Site Designation | Fish IBI | Condition | Benthic IBI | Condition |
| Herring Run | 2007 | UPCK-211-X-2007 | 5.00 | Good | 4.43 | Good |
| Watts Creek | 2007 | UPCK-212-X-2007 | 4.67 | Good | 5.00 | Good |
| Tuckahoe Creek | 2009 | TUCK-450-H-2009 | 3.33 | Fair | 5.00 | Good |
| Skeleton Creek | 2000 | UPCK-113-S-2000 | 2.00 | Poor | 3.00 | Fair |
| Skeleton Creek | 2001 | UPCK-113-S-2001 | 2.67 | Poor | 4.43 | Good |
| Skeleton Creek | 2002 | UPCK-113-S-2002 | 3.33 | Fair | 3.86 | Fair |
| Skeleton Creek | 2003 | UPCK-113-S-2003 | 2.33 | Poor | 4.14 | Good |
| Skeleton Creek | 2004 | UPCK-113-S-2004 | 4.00 | Good | 4.14 | Good |
| Skeleton Creek | 2007 | UPCK-113-S-2007 | 3.00 | Fair | 3.86 | Fair |
| Skeleton Creek | 2009 | UPCK-113-S-2009 | 3.00 | Fair | 3.29 | Fair |
| Site | Sample Date | n | Testicular Oocyte | |
| Prevalence | Severity Index | |||
| 2008 | | |||
| Hearns Pond, DE | 04/30/08 | 8 | 88% | 0.22 ± 0.16 |
| Moore’s Lake, DE | 04/30/08 | 10 | 80% | 0.37 ± 0.38 |
| McColley Pond, DE | 05/01/08 | 9 | 67% | 0.25 ± 0.26 |
| Williston Lake, MD | 05/15/08 | 11 | 73% | 0.24 ± 0.31 |
| Smithville Lake, MD | 05/22/08 | 10 | 40% | 0.11 ± 0.23 |
| Tuckahoe Lake, MD | 05/29/08 | 12 | 42% | 0.22 ± 0.48 |
| 2009 | | |||
| Tuckahoe Lake, MD | 05/12/09 | 10 | 50% | 0.16 ± 0.21 |
| Tuckahoe Lake, MD | 08/19/09 | 9 | 33% | 0.25 ± 0.42 |
| Pocomoke River, MD | 05/14/09 | 10 | 40% | 0.33 ± 0.56 |
| Pocomoke River, MD | 08/20/09 | 5 | 80% | 0.53 ± 0.40 |
| Year | Tillage Practice | % Reduction in fecal estrogens from No-till | % Reduction in estrogenicity from No-till | |
| 2002 | No Till | Conventional Till | 40% | Not measured |
| 2008 | No Till | Turbo Till | 58% | 66% |
| 2009 | No Till | Turbo Till | 28% | 71% |
| No Till | Turbo Till | Sample lost | 66% | |
| 2010 | No Till | Turbo Till | 38% | 43% |
| 2011 | No Till | Sub-Surface Injection (Grab) | 70% | Not measured |
| No Till | Sub-Surface Injection (Composite) | 84% | Not measured | |
| |||||
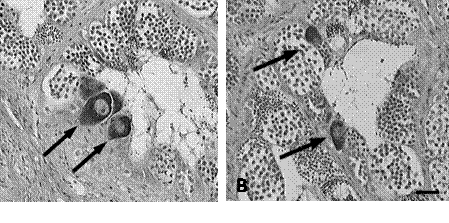
Figure 1. Testicular oocytes (TO) in largemouth bass (Micropterus salmoides) from the Delmarva Peninsula, USA. Clustered (A) and diffuse (B) previtellogenic oocytes (arrows) within epithelium of testicular tubules (Bar = 50 mm; H&E stain)
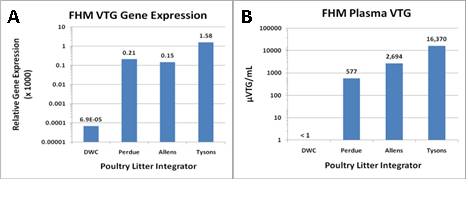 |
Figure 3. Fathead minnow VTG mRNA (A) and plasma protein (B) induction following 9-d 2010 exposures to aqueous solutions of three different poultry litters.
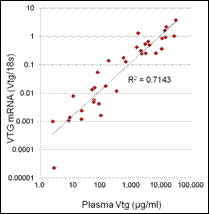
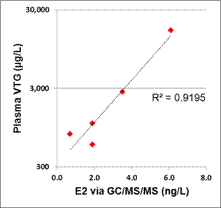
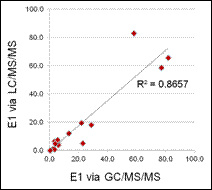
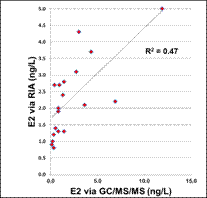
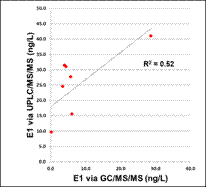
Figure 7. Comparison of E2 as measured by GC/MS/MS (ng/L) and RIA (ng/L) as measured in fish exposures.
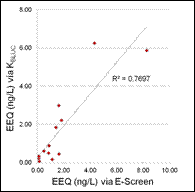
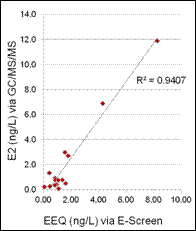
Conclusions:
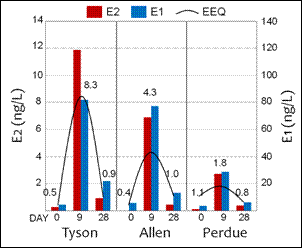
Runoff from poultry litter amended fields and from aqueous litter exposures generated in the laboratory were found to be estrogenic to both adult and larval fish. Vitellogenin (VTG), a potent protein biomarker for endocrine disruption due to estrogens, was induced in male fathead minnows in both exposure scenarios. Male fish do not produce VTG unless exposed to some external source of estrogens. VTG can be measured directly in blood plasma or by measuring the induction of VTG-mRNA in fish livers. Both techniques proved to be equally sensitive in detecting VTG. In addition, larval fathead minnows exposed to poultry litter from runoff produced adult fish whose sex was harder to determine than fish from control treatments. Consistently, measured 17β-estradiol (E2) and estrone (E1) levels in poultry litter solutions were low at exposure initiation and increased during fish exposure periods before decreasing by day 28. Estrogenicity followed an identical pattern with activity increasing before decreasing at day 28. Estrogenicity was still measureable at day 28. In each instance, estrogenicity was explained entirely by measured E2 levels. This is not surprising given that E2 is the most potent vertebrate estrogen.
Of the estrogen and estrogenicity analytical methods compared in this study using split samples, Gas Chromatography Dual Mass Spectrometry and Liquid Chromatography Dual Mass Spectrometry proved to be equally sensitive for measuring environmental samples containing poultry litter. In contrast, Radioimmunoassay and Ultra Performance Liquid Chromatography methods had difficulty with measuring poultry litter samples due to matrix interferences. VTG analytical methods measuring fish responses were sensitive in detecting estrogenicity at the very low levels detected by the more traditional chemistry methods. Likewise, In-vitro Estrogenicity Analysis (KBLUC Reporter-Gene Assay and E-screen Proliferation Assay) using estrogen-inducible reporter-gene cell lines also proved equally sensitive to the chemical methods. Both VTG and In-vitro Estrogenicity Analysis can yield indications of exposure to estrogens at or near chemical analytical limits of quantitation and the In-vitro method can do this without requiring fish exposures.
A large part of this project involved field sampling to detect possible endocrine disruptive effects in poultry litter influenced streams and lakes on the Delmarva Peninsula. Rigorous sampling of four small streams in agriculturally dominated watersheds did not show degraded systems when compared to reference streams. Many components of agriculture runoff may not drastically impact lotic (flowing systems) because any contaminants are transient there. In contrast, such runoff could have greater impacts to lentic (lake) environments where the biota are exposed for a longer time period (contaminants do not flow away quickly). Largemouth bass sampled from six lakes and ponds in Maryland and Delaware and a large river in Maryland showed male fish with gonads containing eggs (intersex). Intersex prevalences ranged from 40% to 88% similar to prevalence values from other parts of the country. These are the first reported findings of intersex in waters of the Delmarva Peninsula.
Finally, our investigations into the benefits of using alternative poultry litter tillage practices to reduce nutrient and steroid runoff from litter amended fields found significant reductions in these contaminants from practices that incorporated the litter into the soil compared to No-till litter applications. Both vertical tillage (Turbo-till) and subsurface injection (Poultry Litter Subsurfer) dramatically reduced runoff or nutrients, especially dissolved phosphorous, and steroids. On October 15, 2012 Maryland’s New Nutrient Management Regulations went into effect. The new management regulations are part of Maryland’s Watershed Implementation Plan required by EPA to meet EPA’s goals to avoid future consequences of nutrient runoff. The regulations state that ...”organic nutrient sources shall be injected or incorporated as soon as possible, but no later than 48 hours after application.” Organic nutrient sources include animal manure. The data generated by Dr. Ken Staver and the other Co-PIs at WREC for this EPA CAFO study on the efficacy of alternative tillage practices were used to help provide the scientific basis for this decision.
References:
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 9 publications | 1 publications in selected types | All 1 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Yonkos LT, Friedel EA, Fisher DJ. Intersex (testicular oocytes) in largemouth bass (Micropterus salmoides) on the Delmarva Peninsula, USA. Environmental Toxicology and Chemistry 2014;33(5):1163-1169. |
R833418 (2011) R833418 (Final) |
Exit |
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- 2011 Progress Report
- 2010 Progress Report
- 2009 Progress Report
- 2008 Progress Report
- Original Abstract
1 journal articles for this project