Grantee Research Project Results
Final Report: The Future of Harmful Algal Blooms: An Empirical Approach to Predicting the Combined Impacts of Rising CO2, Temperature, and Eutrophication
EPA Grant Number: R833221Title: The Future of Harmful Algal Blooms: An Empirical Approach to Predicting the Combined Impacts of Rising CO2, Temperature, and Eutrophication
Investigators: Hutchins, David A. , Coyne, Kathryn J. , Warner, Mark A
Institution: University of Delaware , University of Southern California
EPA Project Officer: Packard, Benjamin H
Project Period: March 15, 2007 through March 14, 2010
Project Amount: $549,993
RFA: Ecology and Oceanography of Harmful Algal Blooms (2006) RFA Text | Recipients Lists
Research Category: Aquatic Ecosystems , Water
Objective:
Our goals for this project are to evaluate the cumulative impacts of increasing CO2, temperature and nutrients on HAB raphidophytes and dinoflagellates that co-occur in the Delaware Inland Bays (DIB). We will test the hypotheses that 1) Rising CO2 and temperature in concert with increased eutrophication will favor the dominance of raphidophytes and dinoflagellates over competing non-harmful algal species; and 2) These effects will be manifested through changes in gene expression, cell physiology, and ecological dominance. The specific objectives are to (i) quantitatively assess the effects of increases in CO2, temperature and nutrients on the growth rates and photosynthetic physiology of HAB species, relative to non-HAB species; (ii) evaluate differential expression of critical nutrient and CO2 -regulated genes; and (iii) carry out manipulative experiments with natural algal communities containing HAB species to determine their likely responses to global change.
Summary/Accomplishments (Outputs/Outcomes):
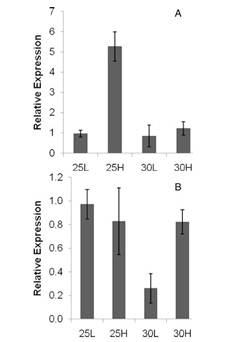
We recently sequenced the gene for acetyl-coA carboxylase (ACCase) from Heterosigma akashiwo and examined the effects of elevated temperature and CO2 concentration on its expression. ACCase catalyzes the first committed step in fatty acid synthesis and is a target for the regulation of lipid metabolism. The storage of energy-rich lipids is often increased under stress, such as nutrient limitation. Temperature also alters the lipid composition of cellular membranes, and future increases in temperature due to global warming may have a pronounced effect on membrane structure and lipid content in algae. By using semi-continuous cultures, we eliminated nutrient stress, and could evaluate strain-specific responses associated with changes in temperature and pCO2. Strain CCMP 2808 is a cold water toxic strain isolated on the west coast, and strain CCMP 2393 is a warm water nontoxic strain isolated from Delaware’s inland bays. The effect of experimental conditions on ACCase expression varied between isolates of H. akashiwo, showing that greenhouse ocean conditions may differentially affect strains of a single HAB species. In this experiment, for the cold water strain, ACCase expression increased with increasing CO2 concentration at 25°C only, and expression under high temperatures remained at control levels (Fig. 1). Conversely, for the warm water strain, expression decreased from control levels for the 30°C treatment, but recovered back to control levels when CO2 was added at 30°C.
Figure 1. Relative expression of acetyl-coA
carbonxylase in Heterosigma akashiwo WA
strain (A) and DE strain (B). 25L, (25oC + 375
ppm CO2); 25H, (25oC + 750 ppm CO2); 30L,
(30oC + 375ppm CO2);30H,(30oC + 750 ppm CO2)
Analysis of community structure with respect to temperature and nutrients
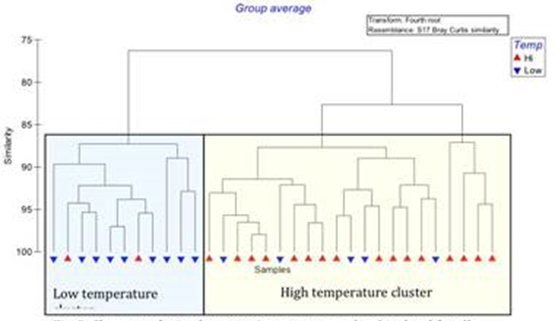
We collected weekly water samples from 3 dead-end canals and evaluated changes in community composition using class-level QPCR assays for diatoms, dinoflagellates and raphidophytes developed in Coyne’s lab. We evaluated the relative abundances of each class of phytoplankton with respect to site, temperature, salinity, and nutrient concentrations. Cluster analysis shows a significant clustering of the community structure combined from all 3 sites due to temperature (Fig. 2). The temperature threshold for this difference was identified as 27°C, such that samples collected below that temperature were more similar in terms of relative abundances of diatoms, dinoflagellates and raphidophytes, while temperatures at or above 27°C were more similar to each other (Fig. 2).
Figure 2. Cluster analysis of community structure at the class level for all three sites. Low and
high temperature communities are significantly.
Within each cluster, dinoflagellates and raphidophytes significantly contributed to the community structure, with greater relative abundances of both groups at higher temperatures (above 26°C) compared to lower temperatures. Diatoms as a group did not appear to be affected by temperature, suggesting that differences in their relative abundances within each sample were due to species-specific responses or to environmental factors that were not evaluated here.
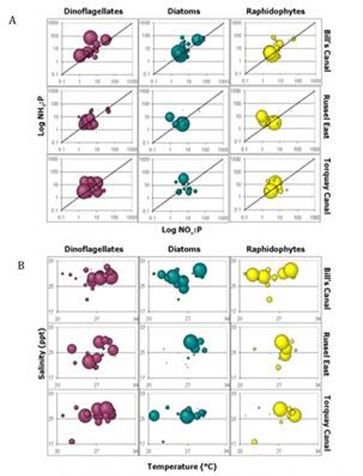
We then evaluated site-specific changes in the relative abundances of each algal class with respect to nutrients (specifically N-source and N:P ratios), temperature and salinity (Fig.3). Our results show site-specific differences in nitrogen utilization among dinoflagellates in that dinoflagellates as a group are more abundant at the Bill’s Canal site when NH4 was the dominant nitrogen source, while dinoflagellates bloom over a smaller range of N:P ratios at the other 2 sites and there was no distinct preference for nitrogen source (Fig. 3a). Site-specific differences in the relative abundances of each class of phytoplankton were also apparent when evaluated with respect to temperature and salinity (Fig. 3b). Here, diatoms were more abundant over a broader range of temperatures at the Bill’s Canal and Torquay Canal sites compared to the Russel East site, and raphidophytes bloomed over a broader range of temperatures but narrower range of salinities at the Bill’s Canal site compared to the Russel East location. Temperature appears to have had a greater impact on dinoflagellates at Bill’s Canal, with blooms occurring above 26°C, compared to the other 2 sites. With this data, we can begin to predict how the phytoplankton community as a whole will respond to environmental factors at each site. Future work will focus on identifying key species that are driving these differences.
Figure 3. RElative abundances of dinoflagellates, diatoms and
raphidophytes are indicated by bubble size at three dead end
canals in Delaware's inland bays, evauated with respect to
(A) N-source and (B) temperature and salinity.
Climate change impacts on toxic Karlodinium veneficum
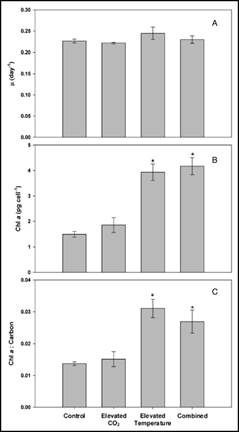
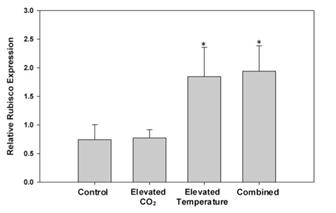
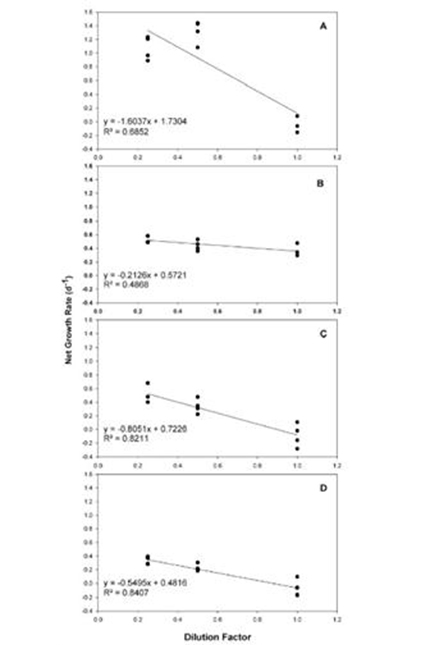
In previous experiments, we evaluated Karlodinium veneficum growth and heterotrophic activities in response to ambient and elevated temperature crossed with ambient and elevated CO2. These experiments were conducted after short-term exposure of K. veneficum and the prey species, Rhodomonas, to elevated temperatures. We repeated these experiments after these species had been fully acclimated to experimental temperatures (25 and 30°C for more than 1 year) and CO2 (375 and 750 ppm for 3 weeks). In unialgal cultures, K. veneficum had higher cellular carbon content at elevated temperature and/or elevated CO2 (Fig. 5c), but these conditions did not result in increased growth rate for this species (Fig. 4a). Chlorophyll a, Rubisco expression, and chl:C ratios all increased significantly with the increase in temperature, but showed no significant changes with the increase in pCO2 (Fig. 4b,c - 6). Together, these data suggest that elevated temperature alone, or in conjunction with elevated pCO2 will have a positive effect on the physiological state and productivity of this species, but may not have a significant impact on its cell densities. Grazing by K. veneficum on Rhodomonas decreased with elevated temperature and/or pCO2 (Fig. 7), consistent with the apparent enhancement in physiological state of the dinoflagellate when cultured under these conditions in unialgal culture. Our results suggest that anticipated climate change conditions will reduce dependence on grazing for carbon acquisition by K. veneficum, potentially impacting phytoplankton community composition and dynamics.
Figure 6. Relative expression of Rubisco (normalized to
actin) in K. veneficum at ambient and elevated temperature
and CO2 levels. Asterisks indicate significant differences
compared to control (n=4±SD).
Figure 7. Grazing rates of K. veneficum on Rhodomonas sp. at
ambient temperature and CO2 (A), elevated CO2 (B), elevated
temperature (C), and elevated temperature and CO2 (D). The
grazing experiment was conducted over 24 hours using the
dilution method to take into account differences in growth rates
for the prey species under each condition. Grazing rates are indicated
by the slope of each line (n=4)
Conclusions:
We anticipate future work with harmful raphidophytes and dinoflagellates in a pH controlled continuous culture system. This experimental design will allow us to maintain algae at higher concentrations, while still maintaining the desired constant carbonate chemistry of the growth media. Such a system should allow us to sample cultures for several cellular assays that require large amounts of biomass, and will allow us to monitor algal response over an extended period of time. We anticipate at least one more year of environmental and algal population data collection at our three canal sites as well.
Journal Articles on this Report : 6 Displayed | Download in RIS Format
| Other project views: | All 19 publications | 8 publications in selected types | All 8 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Boyd PW, Strzepek R, Fu F, Hutchins DA. Environmental control of open-ocean phytoplankton groups:now and in the future. Limnology and Oceanography 2010;55(3):1353-1376. |
R833221 (Final) |
Exit Exit |
|
|
Demir E, Coyne KJ, Doblin MA, Handy SM, Hutchins DA. Assessment of microzooplankton grazing on Heterosigma akashiwo using a species-specific approach combining quantitative real-time PCR (QPCR) and dilution methods. Microbial Ecology 2008;55(4):583-594. |
R833221 (2007) R833221 (Final) R831041 (Final) |
Exit Exit |
|
|
Fu F-X, Place AR, Garcia NS, Hutchins DA. CO2 and phosphate availability control the toxicity of the harmful bloom dinoflagellate Karlodinium veneficum. Aquatic Microbial Ecology 2010;59(1):55-65. |
R833221 (2007) R833221 (Final) |
Exit |
|
|
Fu F-X, Zhang Y, Warner ME, Feng Y, Sun J, Hutchins DA. A comparison of future increased CO2 and temperature effects on sympatric Heterosigma akashiwo and Prorocentrum minimum. Harmful Algae 2008;7(1):76-90. |
R833221 (Final) R831041 (Final) |
Exit Exit Exit |
|
|
Hutchins DA, Mulholland MR, Fu F-X. Nutrient cycles and marine microbes in a CO2-enriched ocean. Oceanography 2009;22(4):128-145. |
R833221 (Final) |
Exit Exit |
|
|
Sun J, Hutchins DA, Feng Y, Seubert EL, Caron DA, Fu F-X. Effects of changing pCO2 and phosphate availability on domoic acid production and physiology of the marine harmful bloom diatom Pseudo-nitzschia multiseries. Limnology and Oceanography 2011;56(3):829-840. |
R833221 (Final) |
Exit Exit |
Supplemental Keywords:
Climate change, harmful algae, toxicity, RFA, Scientific Discipline, Ecosystem Protection/Environmental Exposure & Risk, Water, algal blooms, Ecological Risk Assessment, Oceanography, Environmental Monitoring, water quality, HAB ecology, bloom dynamics, cyanobacteria, eutrophication, algal bloom detectionRelevant Websites:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.