Grantee Research Project Results
2007 Progress Report: The Future of Harmful Algal Blooms: An Empirical Approach to Predicting the Combined Impacts of Rising CO2, Temperature, and Eutrophication
EPA Grant Number: R833221Title: The Future of Harmful Algal Blooms: An Empirical Approach to Predicting the Combined Impacts of Rising CO2, Temperature, and Eutrophication
Investigators: Hutchins, David A. , Warner, Mark A , Coyne, Kathryn J.
Current Investigators: Hutchins, David A. , Coyne, Kathryn J. , Warner, Mark A
Institution: University of Delaware , University of Southern California
EPA Project Officer: Packard, Benjamin H
Project Period: March 15, 2007 through March 14, 2010
Project Period Covered by this Report: March 15, 2007 through March 14,2008
Project Amount: $549,993
RFA: Ecology and Oceanography of Harmful Algal Blooms (2006) RFA Text | Recipients Lists
Research Category: Aquatic Ecosystems , Water
Progress Summary:
D. Hutchins, USC:
Overview
The nature of our participation has changed somewhat from what was originally planned, due to the PI moving from the University of Delaware to a new position at the University of Southern California early in the project. Obviously, this has limited our ability to carry out extensive fieldwork in the Delaware Inlands Bays (DIB), the original focus of the project. However, we returned to Delaware for two weeks in the early summer of 2007 for joint DIB field experiments with coPIs Coyne and Warner, and we have carried out extensive physiological work using DIB cultures to examine their responses to changing CO2 and nutrients in our new laboratory at USC. The move to the West Coast also afforded us with an opportunity to extend our project’s scope to examine CO2 and nutrient effects on HAB species of concern along the California coast, especially the toxic diatom Pseudo-nitzschia. Other opportunistic additions to the project include a manipulative CO2 experiment on a cruise of opportunity in the Gulf of Mexico during a Karenia brevis bloom with coPI Warner. All of these projects are detailed below. We are currently continuing with experimental work, as well as with writing our results for publication. Spending on our separate USC budget is on track.
| Principal investigator | Dave Hutchins (USC) |
| Research scientist | Feixue Fu (USC) |
| Postdoctoral researcher | Andrew King (USC) |
| Graduate students |
Elif Demir (UD, PhD completed 2007) Nathan Garcia (USC, current) |
| Undergraduate students |
Kevin Lyu (USC) Tony Rzeteljski (USC) |
| Outside collaborators |
Allen Place (University of Maryland Biotechnology Institute) Cindy Heil (Florida Fish and Wildlife Research Institute) Jun Sun (Key Laboratory of Marine Ecology & Environmental Science, Institute of Oceanology, Chinese Academy of Sciences, Qingdao, PRC) Jed Fuhrman (USC) Allen Place (University of Maryland Biotechnology Institute) Cindy Heil (Florida Fish and Wildlife Research Institute) Jun Sun (Key Laboratory of Marine Ecology & Environmental Science, Institute of Oceanology, Chinese Academy of Sciences, Qingdao, PRC) Jed Fuhrman (USC) |
Recent and current projects
1) DIB natural community CO2 and temperature manipulation experiment. PI Hutchins and supported researcher Fu traveled to Delaware in May/June 2007 to participate in a first try at a DIB natural community CO2/temperature manipulative experiment along with the other PIs. The intention was to incubate a natural DIB community containing HABs and co-existing species in a complete four treatment matrix of pCO2 levels (380 ppm, present day; and 750 ppm, projected year 2100) and temperatures (present day and projected future warming scenario). This experimental design was chosen to allow examination of potential interactions between rising pCO2 and temperature on DIB HAB species within an intact native phytoplankton community.
Details of the outcome of these DIB experiments are given in the narratives of PIs Coyne and Warner. Briefly, QPR results from Coyne’s group showed higher abundance of the HAB dinoflagellates Gyrodinium instriatum, Prorocentrum minimum, and Karlodinium veneficum at higher CO2 (and lower temperature) over the first several days. By late in the experiment however, diatoms had come to dominate in all treatments, perhaps due to reduced grazing pressure in the enclosed experimental bottles. While the results of this trial run natural community experiment were interesting and suggest several potentially fruitful new research avenues, they also were cautionary since they demonstrate the potential for experimental artifacts in work using complex natural communities in enclosed experiments.
|
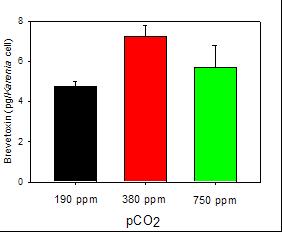
Figure 1. Levels of brevetoxin per K. brevis cell in a shipboard incubation experiment at glacial (190 ppm, black), present day (380 ppm, red) and projected year 2100 (750 ppm, green) pCO2 levels. |
2) GOM Karenia brevis bloom CO2 manipulation experiment. PIs Hutchins and Warner and USC student Garcia had an opportunity to carry out a pCO2 manipulation experiment during an ECOHAB-funded cruise to the Gulf of Mexico with collaborator Cindy Heil in October of 2007. We incubated a dense Karenia brevis bloom collected off Sanibel Island, Florida at 190, 380 and 750 ppm CO2, and followed photosynthetic parameters (Warner), biological and biogeochemical parameters (Hutchins) and K. brevis abundance and brevetoxin production (Heil). Results demonstrated no significant effect of pCO2 changes on K. brevis abundance. Interestingly however, brevetoxin levels did vary with changing pCO2. Significantly higher brevetoxin concentrations (per cell) were found at present day pCO2 (380 ppm) than at low glacial levels (190 ppm). Toxin levels then declined somewhat from 380 to 750 ppm pCO2, so that the highest levels were recorded in the “present day” treatment (Figure 1). Intriguingly, these preliminary results suggest the possibility of declines in K. brevis toxin production with future increases in atmospheric pCO2. These trial experiments will require further work before robust predictions can be made, of course.
|
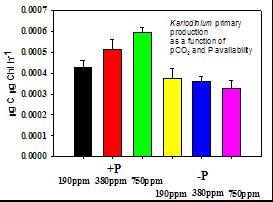
Figure 2. Carbon fixation rates of Karlodinium cultures across a range of pCO2 from 190 to 750 ppm, in P-limited and P-replete cultures. P-replete: Black, 190 ppm; red, 380 ppm; and green, 750 ppm. P-limited: (yellow, 190 ppm; blue, 380 ppm; and pink, 750 ppm). |
3) Laboratory culture studies of DIB Karlodinium veneficum physiological responses and toxin production in response to changing pCO2, light and P availability. USC research assistant professor Fu has led our effort to understand how the toxic DIB dinoflagellate Karlodinium veneficum may respond to changing CO2 and phosphorus availability. These laboratory culture experiments examined both cell physiology and karlotoxin production, in collaboration with UMBI researcher Allen Place. As expected, parameters like carbon fixation and growth rates were higher in P-replete cultures than in P-limited ones. Interestingly though, carbon fixation rates increased significantly with increasing pCO2 in P-replete cultures, but not in P-limited ones (Figure 2). This suggests the possibility of enhanced Karlodinium growth and abundance under future high CO2, acidified conditions, as long as enough P is available. It is also consistent with the early preliminary results from the DIB natural community experiment (above).
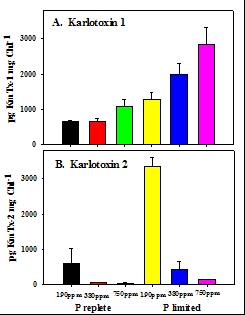
Even more interesting was the pattern of toxin production with these two variables. Total karlotoxin production was greatly enhanced under P-limitation (Figure 3 A and B); this confirms previously published results of Dr Place. Although pCO2 had little effect on total karlotoxin production within each P treatment, it affected the production of the two isomers of karlotoxin produced by this strain quite differently. In P-limited cells, the isoform Karlotoxin 1 responded positively to increasing CO2 across all three concentrations (Figure 3A). In contrast, Karlotoxin 2 production declined dramatically from 190 to 380 ppm, and then even further at 750 ppm (Figure 3 B). The net result is a radically different ratio of the two isoforms at each pCO2, with large increases in the Karlotoxin 1:Karlotoxin 2 ratio with increasing pCO2. The ecological significance of this observation is at present uncertain, since the relative toxicity, lability and residence time of each isoform is at present not fully understood. It does point out a definite trend towards toxin regulation by pCO2 in this DIB HAB species though, and warrants further investigation. A manuscript is currently in preparation from this work.
|
Figure 3. Production of Karlotoxin 1 (panel A) and Karlotoxin 2 (panel B) across a range of pCO2 from 190 to 750 ppm, in P-limited and P-replete cultures. P-replete: Black, 190 ppm; red, 380 ppm; and green, 750 ppm. P-limited: (yellow, 190 ppm; blue, 380 ppm; and pink, 750 ppm). |
USC undergraduate Kevin Lyu also completed a research project with Karlodinium in our lab, supported by this grant and the USC SOAR (Sophomore Opportunities for Academic Research) program. This project looked at growth and physiology of Karlodinium at high and low light levels, under both present day (380 ppm) and future (750 ppm) pCO2. Although in general karlotoxin levels in this experiment were lower than in the P-limited cultures discussed above, Kevin’s results suggested an enhancement of toxin production by both high light and high CO2. These results are currently being prepared for publication along with Dr. Fu’s results discussed above; Kevin will be a coauthor on this paper.
4) Laboratory culture studies examining competition between DIB Karlodinium veneficum and Prorocentrum minimum across a range of pCO2. Dr Fu in collaboration with visiting scientist Dr. Jun Sun just finished a series of experiments in which they grew mixed cultures of DIB isolates of Karlodinium veneficum and Prorocentrum minimum across a range of pCO2 values. These experiments were begun by USC undergraduate Tony Rzeteljski, who unfortunately had to withdraw from the project midway through due to family concerns. The object of these experiments is to complement and enhance the DIB natural community experiments by conducting controlled lab competition experiments with mixed cultures of DIB HAB species. Although we are still working up these data, preliminary cell count data (Figure 4) suggest that although Karlodinium was the “winner “ over Prorocentrum in all treatments, Prorocentrum became increasingly abundant (lower Karlodinium to Prorocentrum cell ratios) as pCO2 concentrations increased. These types of experiments could thus give insights into possible changes in interspecific competitive interactions under rising CO2 and global change scenarios, and we plan to do more of these artificial community competition experiments, including ones with larger species matrices (3 or even 4 species).
|
Figure 4. Ratio of Karlodinium to Prorocentrum cells at the end of a mixed culture competition experiment in which the two species were grown together at 190, 380, and 750 ppm CO2 |
5) Laboratory culture studies of Pseudo-nitzschia physiological responses and toxin production in response to changing pCO2 and P availability. Visiting scientist Jun Sun led these experiments in collaboration with Dr Fu and USC researcher Dave Caron. We subjected cultures of the toxic diatom Pseudo-nitzschia to growth at a combination of high (750 ppm) or low (380 ppm) CO2, and high or low P, in much the same manner as the laboratory Karlodinium culture experiments discussed above. As with the other work, these Pseudo-nitzschia experiments were intended to determine interactive effects of CO2 and nutrients on growth, physiology and toxin production. These experiments have been completed, and we are beginning to analyze the samples. Domoic acid samples are being run using a standard ELISA method in Dr Caron’s laboratory; we expect to have these results in the next two weeks or so.
6) California coastal natural community CO2 manipulation experiment. PI Hutchins and postdoc King just finished (July 2008) carrying out a perturbation experiment using a natural mixed phytoplankton community collected from the Southern California bight. We incubated this community for a week at 190, 380 and 750 ppm CO2 at the USC Wrigley Institute Facility on Catalina Island, in collaboration with researchers from the labs of Jed Fuhrman and Dave Caron (USC) who took samples for molecular determinations (ARISA) of phytoplankton and bacterioplankton community structure. Our interest for this ECOHAB project was in the fact that this mixed community included potentially toxic Pseudo-nitzschia diatoms; accordingly, we took samples to determine any changes in their abundance, as well as domoic acid samples to look for any toxicity changes with pCO2. Along with a host of other biological and biogeochemical parameters, these samples are currently being prepared for analysis, and we hope to have early results in the next month or two. Hopefully, these natural community experiments will serve as a perfect comparison to the laboratory Pseudo-nitzschia culture experiments described above.
K.Coyne, UD:
Results of climate change experiment from May, 2007:
DNA was extracted from samples on days 2-10 and the eukaryotic community structure was evaluated by denaturing gradient gel electrophoresis (DGGE). The results indicate a diverse community initially which became dominated by a single species (most likely Skeletonema costatum) in all treatments by Day 6. PCR analysis using species specific primers indicated the presence of 5 HAB species initially: Chattonella subsalsa, C. cf. verruculosa¸ Heterosigma akashiwo, Prorocentrum minimum and Karlodinium veneficum. Gyrodinium instriatum was also present in the initial water sample. By Day 2, the raphidophyte species were no longer observed. Raphidophytes are very fragile so that increased turbulence by CO2 bubbling and sample mixing prior to daily sampling may have had a negative effect on these species.
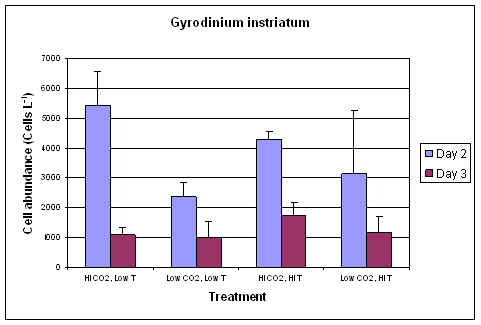
Using primers and probes for quantitative real-time PCR (QPCR), we evaluated the abundance of each of the dinoflagellate species on Days 2 and 3 (Fig. 1-3). Results suggest that the dinoflagellate species all responded positively to increased CO2 so that on Day 2, cell densities were higher in the 750 ppm CO2 treatments than in ambient 375 ppm CO2 conditions. When comparing cell abundance at different temperatures, however, the dinoflagellates all achieved higher cell densities at ambient temperature than at the higher temperature. By Day 3, abundances of all three dinoflagellate species decreased in all treatments except for P. minimum, which increased cell densities in the ambient CO2, high temperature treatment (Fig. 2). Our results suggest that dinoflagellate blooms may be stimulated by increased CO2 under anticipated conditions associated with global warming, but that they may not be competitive with other species such as diatoms. In future experiments, we plan to examine this hypothesis more closely.
|
Fig. 1 QPCR analysis of G. instriatum cell abundance in mixed community experiments at Day 2 and Day 3. Error bars represent standard deviation of triplicate samples. |
Nitrate reductase sequence analysis and enzyme activities:
One of our objectives during this project is to measure expression of nutrient- and CO2- regulated genes in response to climate change. We recently finished sequencing the nitrate reductase (NR) gene from the HAB species Heterosigma akashiwo and we are evaluating changes in nitrate reductase gene expression and enzyme activities in response to increased temperatures, increased CO2, and nitrogen source and concentrations. It was found that NR enzyme activity declines and transcript abundance increases upon initial exposure to high temperature. This is similar to observed diatom Reponses to high light/high temperature conditions in which NR activity decreases and RUBISCO activity increases. After an acclimation period of four days, however, NR activity in H. akashiwo returned to control levels under high temperature and was not significantly different across the temperature and CO2 treatments (Fig. 4). In an experiment conducted previously, we found that there was no difference in NR transcript abundance after H. akashiwo was acclimated to temperatures ranging from 18°C to 28°C, suggesting that transcript abundance also returns to control levels with acclimation. There did appear to be a significant interaction effect of temperature and CO2 on NR transcript abundance at four days, however, with transcript abundance significantly higher for all treatments versus control cultures. This increase in transcript abundance may account for the observed return of NR activity to normal levels under high temperature and CO2 conditions. Our results indicate that H. akashiwo can maintain NR transcript abundance and enzyme activity after acclimation to increased temperature and CO2 concentration.
|
Fig. 4 NR activity for H. akashiwo after four days acclimation to temperature, CO2 conditions. LT=Low Temp; HT=High Temp; LC=Low CO2; HC=High CO2. Error bars represent the standard deviation of the mean.
|
In related experiments, we used PCR to amplify a fragment of the NR gene from another HAB raphidophyte, C. subsalsa, that we have in culture. We are sequencing the fragment for confirmation that it is NR and plan to then conduct similar experiments with this species for comparison to H. akashiwo. We also have several gene sequences for the diatom, S. costatum, including nitrate reductase and carbonic anhydrase, and will be evaluating gene expression, cellular growth and enzyme activities in this species in response to conditions of increased temperature and CO2 as described in our proposal.
CNOB Project:
In addition to the traditional NR enzyme assay for experiments described above, recent efforts in our laboratory have also focused on the development and validation of a novel method to evaluate species-specific changes in nitrate reductase (NR) activity. When applied to experimental manipulations of natural communities, this method will allow us to evaluate in vivo NR activity for each species in response to environmental change. This in vivo method employs 6-chloro-9-nitro-5-oxo-5H- benzo[a]phenoxazine (CNOB) as a fluorescent substrate for nitrate reductase and flow cytometry to evaluate cell specific fluorescence. Molecular identification of populations sorted by flow cytometry is used for the analysis of mixed communities and can be applied to future work with environmental samples of unknown composition. CNOB is a fluorescent molecule with a molecular weight of 326.7 g/mol and an emission maximum at 583 nm when excited at 488 nm. It was shown that fluorescence intensity at 583 nm is proportional to CNOB concentration and a decrease in intensity due to reduction of CNOB by the enzyme could be used to measure enzyme activity. To test this hypothesis, the reduction of CNOB by purified yeast nitrate reductase (YNaR) was visualized using a spectrafluorometer and a decrease in fluorescence at 583 nm confirms that NR is capable of utilizing CNOB as a substrate. In a separate experiment, increasing concentrations of CNOB were added to YNaR in the presence of 10 mM nitrate-containing buffer. The reaction was assayed for nitrate reduction by spectrophotometric measurement of nitrite production. As CNOB concentration increases, less nitrate is converted to nitrite, presumably due to CNOB out-competing nitrate for the active site of the NR enzyme. Our results confirm that CNOB is a substrate for NR and that reduction of CNOB can be quantitatively assessed by measuring changes in fluorescence at 583 nm.
An artificial mixed community was constructed by mixing the following species growing in culture: Chatonella subsalsa, Karlodinium venificum, Prorocentrum minimum, Heterosigma akashiwo and Gyrodinium instriatum. Flow cytometry was used to visualize cell-specific fluorescence and fluorescence was plotted as FL3-H (LP 650 nm filter) versus FL2-H (580 +/- 45 nm filter). Autofluorescence of a control sample was visualized and CNOB was added to the community and incubated for 60 minutes in darkness. A shift in fluorescence intensity in the FL2-H channel was observed for all species in the mixed community. Groups were gated and sorted individually by the flow cytometer sorting feature into new media for DNA extraction and amplification by PCR. Denaturing gradient gel electrophoresis (DGGE) of PCR products was used to successfully identify groups of individual species sorted from the mixed community compared to standards. After identification, species-specific reduction of CNOB by nitrate reductase can be calculated from shifts in fluorescence observed in the FL2-H channel. Flow cytometry sorting and DGGE were also successful for an environmental sample collected from the Broadkill River.
While this method is still in development, its application to environmental samples will allow us to identify species-specific changes in NR activity under conditions of increased temperature, CO2, and nutrients.
The effect of carbon dioxide (CO2) on mixotrophic activity of Karlodinium veneficum:
In phytoplankton, growth responses are determined by a number of factors (i.e. dissolved gasses, temperature, light intensity, and nutrients), and the effect of individual variables on growth rate can be strongly influenced by interactions of these factors. As a response to probable environmental changes, we conducted a study to evaluate the effect of carbon dioxide (CO2) on growth rate and grazing activity of Karlodinium veneficum.
K. veneficum is a photosynthetic, mixotrophic dinoflagellate that is capable of ingesting prey to fulfill limitations in energy and nutrients. Previous studies have shown that for K. veneficum, heterotrophic activity is the main mode for obtaining energy and carbon; whereas, phototrophy may be a strategy for survival when environmental conditions for heterotrophic activity are poor. Recent studies of other mixotrophic algal taxa, however, have demonstrated that effects of heterotrophy on growth are diverse.
Although the importance of energy and inorganic nutrients as factors that regulate feeding in mixotrophic flagellates has been recognized, detailed examinations of how other factors influence the physiological state of cells and, therefore, feeding capability are scarce. In particular, the effects of global climate change; including increased atmospheric CO2, on heterotrophic activities in mixotrophic dinoflagellates has never been examined. Preliminary data from our research on K. veneficum indicate that when the prey (Rhodomonas) is present, there is a positive correlation with growth rates in high carbon dioxide systems. This research will help to better our understanding of the physiological dynamics of K. veneficum under global warming conditions and will contribute to projections of trends that may result from predicted environmental changes.
For this project, our objectives were to determine changes in cellular growth of K. veneficum with exposure to ambient and elevated CO2 levels, determine changes in grazing activities as a response to elevated CO2 exposure, and determine changes in RUBISCO gene expression as dissolved CO2 levels are varied. We measured growth rates for Rhodomonas under high and low CO2 conditions with and without Karlodinium present to determine grazing rates. We then measured growth rates for Karlodinium under high and low CO2 conditions with and without Rhodomonas to evaluate the effect of CO2 in conjunction with grazing for Karlodinium.
Results: The growth rates of K. veneficum with and without Rhodomonas is presented in Fig. 5. This experiment was conducted under semi-continuous culturing conditions to ensure that Karlodinium did not become nutrient limited. The difference between growth in Rhodomonas treatments without K. veneficum, and those with K. veneficum, is attributed to grazing (Fig. 6). Our results confirm higher growth for K. veneficum with Rhodomonas under both high and low CO2, and higher growth for K. veneficum under high CO2 conditions both with and without Rhodomonas prey.
Results of this study corroborate our results from the mixed community experiment (Fig. 5), and separate experiments conducted by co-PI Hutchins at USC, suggesting that growth rates and the potential for blooms of K. veneficum are likely to increase under higher CO2 conditions. Interestingly, grazing rates also decreased for K. veneficum. This finding suggests that ambient CO2 conditions are not sufficient for sustained growth of K. veneficum and that it must supplement its carbon requirements by grazing on Rhodomonas. Reduced grazing under conditions of increased CO2 by this and other mixotrophic dinoflagellates may also have severe ecological repercussions.
We are currently in the process of quantifying relative expression of the RUBISCO gene using real-time qPCR, to monitor the molecular physiology of this species as dissolved CO2 levels are varied. We also are investigating mixotrophic activities of K. veneficum in laboratory culture with varied CO2 levels at 30° C to determine if higher CO2 levels cause the same effects in mixotrophic capabilities as they do at 25° C.
M. Warner, UD:
1) Results of climate change experiment from May, 2007: Our initial goal for this experiment was to assess any changes in total photosynthetic productivity and photosynthesis to light responses after a prolonged exposure to elevated CO2. After 10 days of exposure, there was no significant change in photosynthetic activity between control and treatment bottles. However, as mentioned above, by this time in the experimental design, all bottles had become dominated by several species of diatoms and thus these results are not representative of the initial raphidophyte response noted above for transcript abundance and NR activity. While labor intensive and requiring more material, future experiments with natural water samples will require more frequent sampling intervals so as to follow the immediate response of the algal community when it is dominated by raphidophytes.
2) GOM Karenia brevis bloom CO2 manipulation experiment. As detailed by Hutchins above, we conducted a short-term ship-board CO2 addition experiment on freshly collected populations of Karenia brevis during a natural bloom in the eastern Gulf of Mexico in the Fall of 2007. Photosynthetic response was measured at the beginning and end of the CO2 exposure by measuring the active chlorophyll a fluorescence using the fluorescence induction and relaxation technique with a FIRe fluorometer (Satlantic). No significant differences were noted in the quantum yield of photosystem II charge separation (Fv/Fm) between gas treatments (data not shown), and all algae appeared photosynthetically competent by chlorophyll fluorescence. This result was not surprising considering the short-term nature of the incubation.
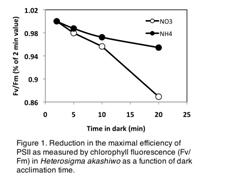
3) The effect of CO2 and temperature on the photobiology of Heterosigma. We have been collaborating with Coyne’s laboratory in an effort to maintain multiple cultures of Heterosigma at both elevated temperature and CO2 over short (days) and longer time intervals (weeks) with different forms of inorganic nitrogen. Our methods include both traditional photosynthesis to irradiance measurements (P:E curves) as well as detailed measurements of chl a fluorescence. These same samples are analyzed for NR activity and transcript abundance as mentioned by Coyne above. An early finding by our laboratory was that traditional protocols for measuring dark acclimated quantum yields in order to assess photosystem II function (Fv/Fm) yielded artificially low values for nutrient replete cells. Specifically, dark acclimation times typically used for other phytoplankton (e.g. 15–30 min.) result in a significant reduction of Fv/Fm, most likely the result of chlororespiratory electron flow, similar to that noted in some other algal species (Fig. 1). This problem is circumvented by maintaining cells in low light (< 5µmol photons•m-2•s-1) for 15 min, followed by 2 min or dark acclimation prior to reading fluorescence.
The Delaware Inland Bays isolate of H. akashiwo was grown semi-continuously (µ=0.35) in modified f/2 media containing either 150 µM NO3- or NH4+ and acclimated to 25 or 30°C, with bubbling with filtered air. Once cultures reached steady-state growth, bubbling was switched to a controlled gas mixture of air containing 375 or 750 ppm CO2 and cultures were maintained for 10 days. Similar to our previously published work (Fu et al. 2007), the addition of elevated CO2 and temperature resulted in a slightly higher maximal photosynthetic rate in NO3- grown cells, however, no such increase was noted for cells grown on NH4+. In addition, maximal primary productivity was consistently higher in NH4+ grown cells relative to NO3- grown cells despite growth conditions (results not shown).
Preliminary work with measuring chlorophyll fluorescence in these algae has provided evidence that traditional dark acclimation protocols, typically used with other microalgae (e.g. placing samples in the dark for 20 min. or longer), results in an artificially low value for the maximal efficiency of photosystem II (Fv/Fm) (Fig. 1). This is most likely due to enhanced chlororespiration while the samples are held in the dark. Thus, we have modified our measurement protocols accordingly to avoid this confounding factor by placing cells in low light (<10 µmol photons•m-2•s-1), followed by 2 min. dark acclimation.
Chlorophyll fluorescence revealed no significant change in the maximal efficiency of photosystem II (Fv/Fm) in any treatment, however some differences were noted for PSII efficiency in the light (Fv’/Fm’) (Fig. 2A), with NO3- grown cells having a slight, yet significant decline in Fv’/Fm’ when grown under elevated temperature, while NH4+ grown cells showed an opposite trend with increasing PSII efficiency in the light as temperature and CO2 are raised (Fig. 2A). These results provide evidence for a gradual additive effect of temperature and CO2 on Fv’/Fm’ in NH4+ grown cells. There appears to be minimal change in the functional absorption cross section in the dark or light acclimated state (σPSII or σPSII’), with both NO3- and NH4+ grown cells showing a small decline that is dependent on the elevated temperature and not CO2 (Fig. 2B). These results are in contrast to previous findings with some photosynthetic prokaryotes that show significant changes in light harvesting structure (e.g. Synechococcus) when grown at elevated temperature. However, we did several interesting differences in the connectivity of photosystem II (PSII) reaction centers (i.e. the degree of light energy transfer between PSII reaction centers) within and between Heterosigma cultures grown on NO3- and NH4+ (Fig. 2 C). In particular there was a decline in PSII connectivity in NO3- and NH4+ grown cells when shifted to elevated CO2 at ambient temperature (25°C), while no change occurred at 30°C or 30°C + elevated CO2 in NO3- grown cells, yet NH4+ grown cells did show an equal and significant rise in PSII connectivity when comparing control to greenhouse conditions (Fig. 2C, closed circles) Further, temperature and CO2 had little impact on the change in PSII connectivity when the cells were exposed to light, however, NH4+ grown cells consistently showed a significant increase in connectivity in the light while NO3- grown cells showed no change or a slight decline in p’ (Fig. 2C, open circles). While these results are preliminary, they may provide further evidence for different photoprotective pathways in this genus of algae when grown under different sources of inorganic N. Further work with continuous cultures as well as possible differences in static and changing light fields would be the next logical steps in this work. Likewise, a closer comparison of photosynthetic electron transport, carbon fixation, and transcript analysis of NR and other proteins (Rubisco) will be useful in this work.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 19 publications | 8 publications in selected types | All 8 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Demir E, Coyne KJ, Doblin MA, Handy SM, Hutchins DA. Assessment of microzooplankton grazing on Heterosigma akashiwo using a species-specific approach combining quantitative real-time PCR (QPCR) and dilution methods. Microbial Ecology 2008;55(4):583-594. |
R833221 (2007) R833221 (Final) R831041 (Final) |
Exit Exit |
|
|
Demir-Hilton E, Hutchins DA, Czymmek KJ, Coyne KJ. Description of Viridilobus marinus (gen. et sp. nov.), a new raphidophyte from Delaware’s Inland Bays. Journal of Phycology 2012;48(5):1220-1231. |
R833221 (2007) |
Exit |
|
|
Fu F-X, Place AR, Garcia NS, Hutchins DA. CO2 and phosphate availability control the toxicity of the harmful bloom dinoflagellate Karlodinium veneficum. Aquatic Microbial Ecology 2010;59(1):55-65. |
R833221 (2007) R833221 (Final) |
Exit |
Supplemental Keywords:
RFA, Scientific Discipline, Ecosystem Protection/Environmental Exposure & Risk, Water, algal blooms, Ecological Risk Assessment, Oceanography, Environmental Monitoring, water quality, HAB ecology, bloom dynamics, cyanobacteria, eutrophication, algal bloom detectionProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.