Grantee Research Project Results
Final Report: Biocatalytic Polymerization of Naturally Occurring Green Tea Flavonoids for Cancer Therapy
EPA Grant Number: SU833204Title: Biocatalytic Polymerization of Naturally Occurring Green Tea Flavonoids for Cancer Therapy
Investigators: Kumar, Jayant , Nagarajan, Subhalakshmi , Braunhut, Susan J.
Institution: University of Massachusetts - Lowell
EPA Project Officer: Page, Angela
Phase: II
Project Period: September 1, 2006 through August 31, 2008
Project Amount: $75,000
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet - Phase 2 (2006) Recipients Lists
Research Category: Nanotechnology , Pollution Prevention/Sustainable Development , P3 Awards , P3 Challenge Area - Chemical Safety , Sustainable and Healthy Communities
Objective:
The primary objective of the proposed research is to explore the possibility of oligomerizing naturally occurring flavonoids using simple eco-friendly methods to yield oligo (catechins) that would prove to be effective in the treatment of cancer. To accomplish this goal, various isomers of catechins were oligomerized using plant peroxidases in biocompatible solvents such as water-ethanol mixtures. This one-pot synthesis is carried out under ambient conditions.
Another important objective of this effort was to evaluate the effectiveness of these oligomerized catechins on the inhibition of growth of breast cancer cells. The inhibitory effect of these oligo(catechins) on the growth of human breast cancer cells and normal mammary cells was compared with that of other naturally occurring flavonoids such as EGCG.
Summary/Accomplishments (Outputs/Outcomes):
Synthesis of oligo(catechins)
Template assisted enzymatic oligomerization:
To obtained oligomeric products with improved solubility the oligomerization of catechins was initially carried out using a range of macromolecular templates like Sulphonated polystyrene (SPS), sodium dodecylbenzene sulphonate (SDS). The product of oligomerization reaction was characterized using UV-visible and fluorescence spectroscopy.13
Effect of the oligo(catechin)-SPS complex on the proliferation of human colon cancer cells:
We have tested the inhibitory effect of the oligo(catechins) synthesized using different templates on the proliferation of human colon cancer cells. The thresholds for an effect on viability were 25 µg/ml for oligo(+)-catechin/SPS and 50 µg/ml for oligo(-)-catechin/SPS. The most effective compound was oligo(-)-catechin/SDS which reduced viability by 88 and 93% at 24 and 48 h, respectively at the highest concentration tested. In contrast, oligo(-)catechin/SPS had no effect at 24 h, but reduced the viability of cells by 66% after 48 h exposure. The oligo(+)-catechin/SPS synthesized using similar procedures caused a small reduction (21%) in cell viability after 24 h incubation, which was enhanced to 48% after an additional 24 h.
In addition, polymeric templates like Poly (ethylene oxide) [PEO] have been used for oligomerization, since PEO could possibly help in aligning the monomers through hydrogen bonding interactions.
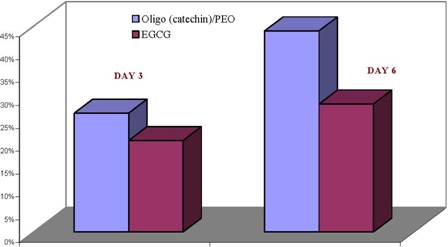
These oligomeric (catechin)/PEO complex were tested for their ability to inhibit the growth of high metastatic human breast cancer cells (figure 2). These results indicate that the water-soluble oligo (catechins) have greater growth inhibitory effects than the EGCG for low and metastatic human breast cancer cells.
While macromolecular templates promote solubility, the bioavailability of the oligocatechins may be restricted due to the size constraints created by the templates. In order to investigate if a macromolecular template was essential for the oligomerization to occur, the oligomerization of catechins was carried out in presence of small molecules such as p-toluenesulphonic acid [PTSA]. It was then concluded that the presence of a polyelectrolytic environment may not be absolutely required for the oligomerization to take place.
Figure 2: Effect of oligo(catechin)/PEO and EGCG on highly metastatic human cancer cells
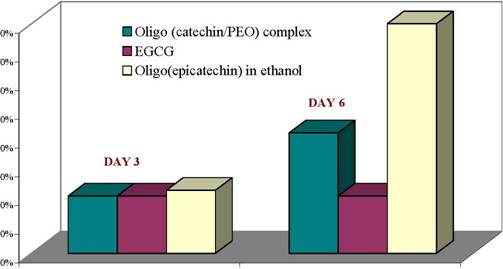
Figure 3: Effect of oligo(catechin)/PEO Oligo (epicatechin),EGCG on growth of low metastatic human cancer cellsOligomerization in eco-friendly water-ethanol mixtures:
A major breakthrough in this research was achieved when the oligomerization was carried out in biocompatible mixed solvent systems. Various isomers of catechins were then biocatalytically oligomerized in benign water-ethanol mixtures. A typical reaction occurs in aqueous media buffered at pH 7 with only the monomeric catechin, a catalytic amount of the enzyme and hydrogen peroxide, to initiate the reaction (figure 4). The resulting oligomer remains soluble in ethanol-water mixtures and can be purified by simple techniques such as dialysis and centrifugation.
Figure 4: Schematic for the oligomerization of (-)-epicatechin
The presence of chiral centers in these catechins results in the oligomers adopting unique conformations/ macro-asymmetry. Circular dichroism polarimetry has been used to study the stereochemical properties and secondary structure of the oligomers. Initial studies indicate that the CD spectra of oligomeric forms of (+) and (-) catechins have a unique secondary structures. These oligo(catechins) with distinct secondary structures are capable of mimicking a specific ligand(s), accessing growth control pathways.
Oligomerization of Epicatechin
In the past, studies on the stability of the catechins found (-)-epicatechin to be the most promising monomer of the catechin family. Epicatechin is more stable than other naturally occurring monomers in the green tea family14.
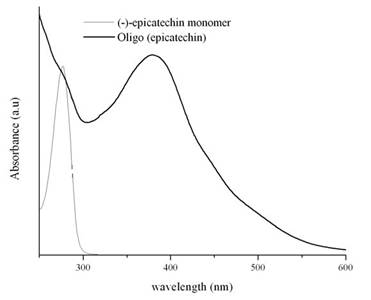
Among all the naturally occurring catechins, (-)-epicatechin, when oligomerized was found to possess excellent anti-proliferative activity. As seen in the fig. 5, the monomer shows significant absorption in the range of 250-300 nm and no absorption beyond 300 nm. The initiation of oligomerization by the addition of H2O2 leads to the appearance of a dark-red brown solution and a new broad absorption peak in the 325-550 nm range with a maxima around 390 nm. The reaction is a simple and is carried out on a bench-top at room temperature. The oligomers that are obtained are water-soluble and stable in excess of three months.
Figure 5: UV-Visible spectra for the oligomerization of (-)-epicatechin
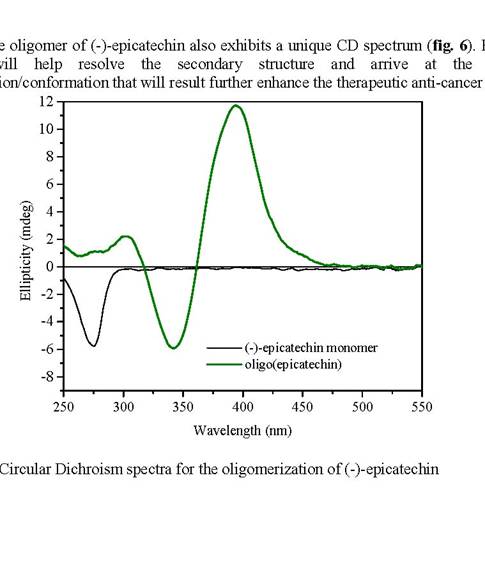
The oligomer of (-)-epicatechin also exhibits a unique CD spectrum (fig. 6). Further CD studies will help resolve the secondary structure and arrive at the distinctive configuration/conformation that will result further enhance the therapeutic anti-cancer activity.
Figure 6: Circular Dichroism spectra for the oligomerization of (-)-epicatechin
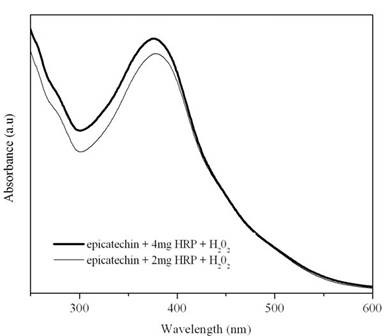
The concentration of the HRP added for the reaction was varied from 1 to 4 mg. Figure 7 shows the UV-visible spectra for the oligomerization of epicatechin using two and four mg of HRP. As seen in the figure, the oligomer obtained in both cases is similar.
Figure 7: UV-Visible spectra for the oligomerization of (-)-epicatechin using different amounts of HRP
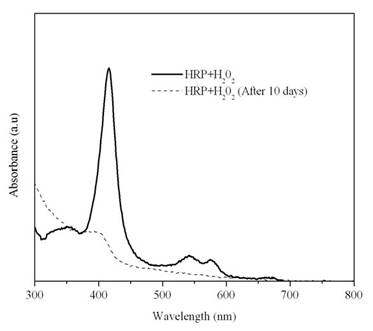
Since native HRP has been reported to possess some anti-cancer properties (15), the activity of HRP on addition of hydrogen peroxide was studied over a time period.
Figure 8: UV-Visible spectra for a mixture of HRP and hydrogen peroxide over time
HRP has a distinct Soret band at around 402 nm. This band was found to almost disappear after 10 days, indicating that most of the enzyme has denatured (fig. 8). The oligomeric epicatechin synthesized was stored for 15-21 days before the anti-proliferative studies were carried out. This ensures that most of the HRP would be denatured and the hydrogen peroxide reduced to water.
MALDI-TOF MS was used to ascertain the molecular weight of the oligomeric epicatechin mixture. Initial MALDI-TOF studies indicate the formation of oligomers (up to 2035 Daltons) indicating the presence of at least 7 repeat units.
Hematin catalyzed oligomerization of catechins
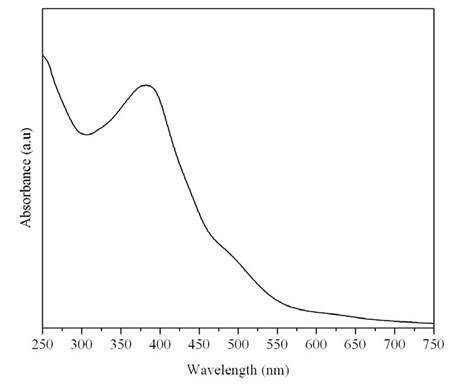
Oligomerization of epicatechin was also carried out using Hematin as a catalyst. As seen in fig. 9, the oligomer obtained from hematin catalysis is similar to shows the UV-visible spectrum for the oligomerization of epicatechin. These oligomers were then tested for their ability to inhibit the growth of high and low metastatic human breast cancer cells. However, the oligomeric epicatechins obtained using hematin catalysis was not very effective in arresting the growth of cancer cells. Further investigations are underway to vary reaction conditions to amplify the anti-proliferative activity.
Figure 9: UV- visible spectrum for the oligomerization of Hematin.
3.1.a.vi Anti-Cancer activity of the oligomerized catechins synthesized in “green” solvent mixtures:
The most promising and widely studied polyphenol among the green tea flavonoids is Epigallocatechin gallate (EGCG) for its potent anti-tumorigenic applications16,,1718. However as mentioned earlier these compounds are highly unstable and difficult to dissolve in aqueous solutions which greatly limits its utility. In addition, there have been very few studies on the efficacy of the various stereoisomers of catechin [(-)-, (+)-, (±)-] on cancer cells. In our tests, we found that the oligomerized catechins synthesized using the template approach and using benign solvent mixtures clearly outperformed EGCG and the monomeric forms in their selectivity, stability and inhibition. We also found that the monomer, catalyst or hydrogen peroxide under the same conditions do not inhibit the growth of cancer cells
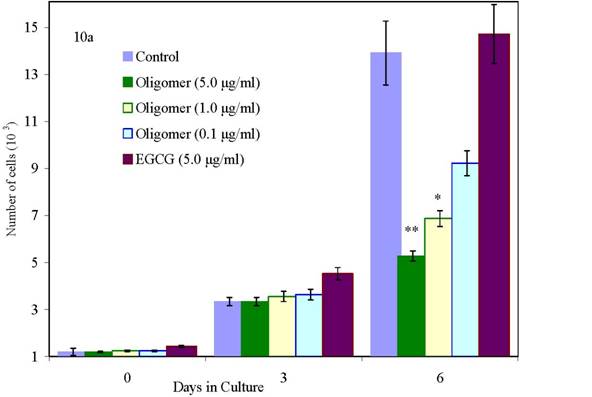
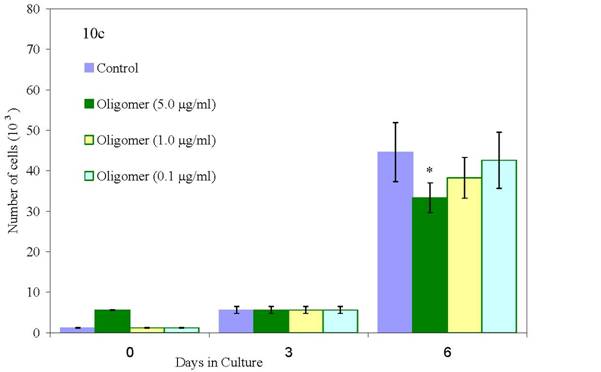
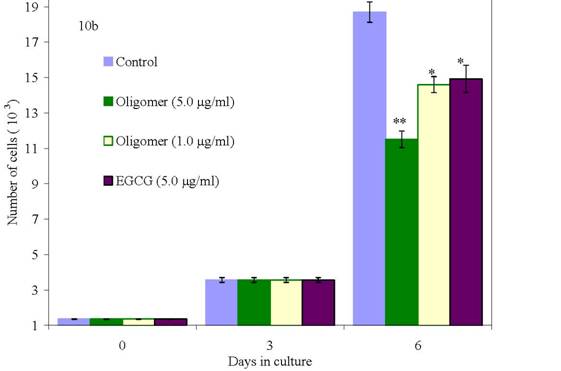
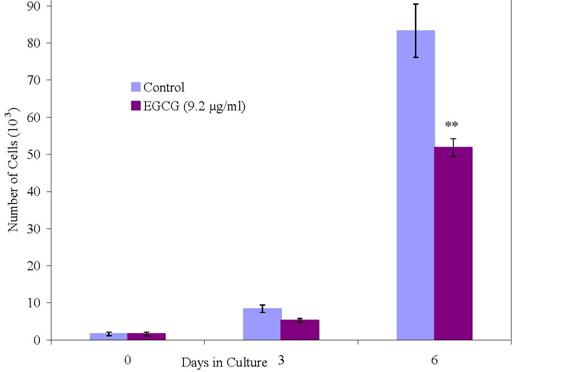
We have conducted proliferation studies of normal and malignant human breast cancer cells using newly synthesized oligo(-) epicatechin/EtOH [ oligo(-)-EC/ETOH], over a dose range of 0.01 to 5 μg/ml in parallel with EGCG at 9.2 μg/ml. As shown in Figure 7c, normal mammary epithelial cells are unaffected in their growth when treated with oligo(–)-EC/EtOH at all doses. At the same doses, MCF-7 cells are 100% growth inhibited (Figure 7a) whereas MDA-MB-231 cells show a dose response at 1.0 and 5.0 μg/ml (Figure 7b). EGCG, included in these studies, and requiring DMSO to be soluble, inhibited MCF-12A cells by 31%, MCF-7 by only 31% and MDA-MB-231 cells by only 19% at a 9.2 μg/ml (20 μM) dose by day 9. Oligo(-)EC/EtOH also proved effective in inhibiting growth of colon adenocarcinoma cells (HT29) at low doses (0.1 ug/ml) (data not shown).
Figure 10: Effects of oligo(epicatechin) on the growth of (10a) low metastatic human breast cancer cells (10b) high metastatic human breast cancer cells (10c) normal cells [p values <0.05(*) or <0.001(**)]
Figure 11: Effect of EGCG on the growth of normal cells.
NS: Not significant
Table 1: Anti-proliferative activity of the monomer, oligomer HRP and hydrogen peroxide
The inhibitory activity of the oligomer was compared with the monomer, enzyme and hydrogen peroxide. As seen in table 1, at effective doses at which the oligo (EC/EtOH) inhibits the growth of cancer cells, the monomer, HRP and hydrogen peroxide did not show any activity.
Separation of the oligomeric catechins using HPLC
Oxidative polymerization of phenol containing compounds can potentially lead to a mixture of polymeric products (19). Preliminary investigations of anti-proliferative activity were carried out using oligo (epicatechin) that could possibly have a mixture of various oligomers.
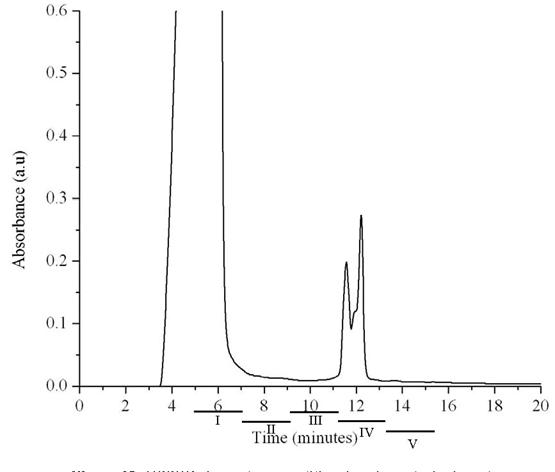
High Performance Size Exclusion Chromatography (HPSEC) has been used for separating the mixture of oligo (epicatechins). A mobile phase of water: ethanol (90:10) was used. As seen in fig. 12, three major peaks can be identified. These were collected at two minute intervals from 5 minutes (e.g. fraction collected between 5 minutes to 7 minutes was designated as fraction I).
Figure 12: HPSEC chromatogram of the oligo (epicatechin) mixture.
These fractions were then concentrated through lyophilization and were subsequently tested for the anti-proliferative activity.
Anti-Proliferative activity of the individual fractions
The anti-proliferative studies were performed on the five fractions tested in parallel with the oligomeric epicatechin mixture.
Inhibition of growth when compared to the control
Day 9
-21%
-17%
-26%
-46%
- 43%
Table 2: Effect of the individual fractions on the growth of high metastatic human breast cancer cells.
The control in all experiments was pH 7 phosphate buffer containing 5% ethanol. Among the five different fractions, fraction IV and V was found to significantly inhibit the growth of the high metastatic human breast cancer cells by day 9.
Conclusions:
This EPA P3 award funded research provided the opportunity to develop a simple one-pot ‘green’ route for producing water-soluble oligomers of green tea catechins. The oligomers synthesized using enzyme catalysis have been shown to be effective in inhibiting the growth of breast cancer cells. An HPLC instrument was acquired during the course of the project and used for separating various oligomeric products formed. The oligomers have a specific inhibitory effect for cancer cells and do not significantly affect the growth of normal cells. The monomers nor HRP tested in parallel did not exhibit any inhibition. In addition, the oligomers are stable in excess of three months under normal conditions. The enzyme catalyzed oligomerization of naturally occurring materials in non-toxic solvents opens new possibilities for developing therapeutic materials using ‘green’ and eco-friendly methods. This project is expected to prompt a paradigm shift in drug discovery and synthesis toward naturally occurring starting materials and ‘greener’ synthetic strategies.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 4 publications | 1 publications in selected types | All 1 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Magarajan S, Nagarajan R, Braunhut S, Bruno F, McIntosh D, Samuelson L, Kumar J. Biocatalytically Oligomerized Epicatechin with Potent andpecific Anti-proliferative Activity for Human Breast Cancer Cells. MOLECULES 2008;13(11):2704-2716 |
SU833204 (Final) |
Exit |
Supplemental Keywords:
Sustainable Industry/Business, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Technology for Sustainable Environment, Sustainable Environment, Environmental Chemistry, Chemicals Management, environmentally-friendly chemical synthesis, alternative chemical synthesis, pollution prevention, green cancer compounds, biocatalytic polymer synthesis, sustainable development, alternative products, clean manufacturing, green chemistry, biocatalytic polymerizationProgress and Final Reports:
Original AbstractP3 Phase I:
Biocatalytic Polymerization of Naturally Occurring Green Tea Flavonoids for Cancer Therapy | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.