Grantee Research Project Results
Final Report: Biocatalytic Polymerization of Naturally Occurring Green Tea Flavonoids for Cancer Therapy
EPA Grant Number: SU832474Title: Biocatalytic Polymerization of Naturally Occurring Green Tea Flavonoids for Cancer Therapy
Investigators: Nagarajan, Subhalakshmi , Kumar, Jayant , Braunhut, Susan J.
Institution: University of Massachusetts - Lowell
EPA Project Officer: Page, Angela
Phase: I
Project Period: September 30, 2005 through May 30, 2006
Project Amount: $10,000
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet (2005) RFA Text | Recipients Lists
Research Category: Nanotechnology , Pollution Prevention/Sustainable Development , P3 Awards , P3 Challenge Area - Chemical Safety , Sustainable and Healthy Communities
Objective:
The primary objective of the proposed research is to develop a simple eco-friendly technology for the polymerization of naturally occurring compounds like "green tea catechins" resulting in a new class of compounds that would be effective in treating cancer. To accomplish this goal, various isomers of catechins were polymerized using naturally occurring plant peroxidases like Horseradish peroxidase (HRP) in biocompatible solvents such as water-ethanol mixtures. This one-pot synthesis is carried out at room temperature and the polymers obtained can be used as such with minimal purification. Another important objective of this effort is to evaluate the efficacy of these polymers on arresting the growth of various types of cancer cells. The inhibitory activities of these polymeric catechins are compared to the monomeric forms as well as other green tea flavanols like epigallocatechin gallate (EGCG).
Summary/Accomplishments (Outputs/Outcomes):
Key findings from Phase I
- Various stereoisomers were biocatalytically polymerized using the enzyme HRP in the presence of biocompatible templates like PEO and in water-ethanol mixtures.
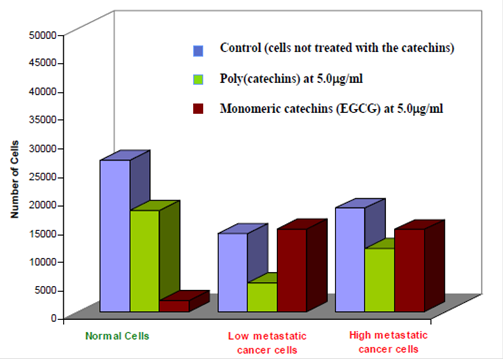
- The polymer of (-)-epicatechin, was identified to exhibit the best specific inhibitory activity for cancer cells. In addition, compared with other known green-tea based compounds poly(epicatechin) does not significantly affect the growth of normal cells( Figure 1).
- At effective doses in which the polycatechin inhibits the growth of cancer cells, it does not affect the growth of normal cells.
- At similar doses, the monomers themselves did not inhibit the growth of cancer cells. One of the most widely studied monomers in the green tea family, EGCG, was found to be less inhibitory than the poly (catechins) against the low and high metastatic cancer cells. In addition, EGCG also arrested the growth of normal cells as well.
- The active catalytic site in HRP is heme porphyrin. Hematin is a heme containing porphyrin and considered to be a promising, cost-effective biomimetic alternative to HRP. We have recently found that Hematin was able to efficiently catalyze the polymerization of catechins.
Figure 1. Effectiveness of the poly(catechins) against normal cells and high and low metastatic cancer cells
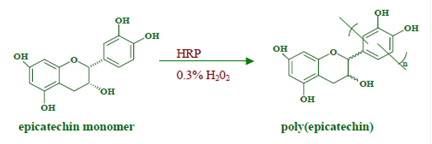
The approach employed here is inherently simple yet extremely powerful. No toxic chemicals are involved at any stage in the process. Figure 2 depicts the polymerization route to obtain water-soluble polymers/oligomers. The starting materials are entirely derived from nature and are biocompatible. The polymerization process does not involve any toxic chemicals/solvents, does not generate toxic by-products and the polymers obtained can be used with minimal purification.
Figure 2. Scheme for the polymerization of catechins in 5% ethanol
Summary of Accomplishments:
- A provisional patent was filed in April 2005.
- The graduate student (P3 Phase I awardee) involved has presented recent results at the International Material Research Conference in Boston in November 2005.
- Initial results of this research have been published in a peer reviewed journal (Journal of Macromolecular Science - Pure Applied Chemistry 2005;42(11):1547-1554.
- An exclusive website on “Green-tea based therapeutics” was developed by the P3 awardee. This website is dedicated to provide information on latest developments on environmentally benign methods for the treatment of cancer ( www.uml.edu/centers/cam/greentea).
Leverage of EPA funds:
We have been successful in obtaining additional funds for this project from the Office of the Congressionally Directed Medical Research Programs (CDMRP) ($ 114,750) in September 2005 and will end by September 2006. The novelty and multidisciplinary approach of this research effort also has been appreciated by the Office of the Chancellor at UMASS Lowell, resulting in a $85,000 grant, which will be used to support personnel involved in this project.
While these additional funds clearly indicate the novelty, interest and effectiveness of this new class of “green” therapeutics, the Phase II would be crucial in taking this project to the next step.
We strongly believe that support from EPA is vital for promoting this new philosophy of using completely “green” methods for treating and preventing one of the most dreadful diseases known to mankind.
Sustainability and P3:
The project directly benefits the three pillars of sustainability - people, prosperity and the planet. The method proposed here could greatly reduce the high costs associated with waste disposal in most countries, besides creating healthier work environments. More importantly, this project is of enormous significance to the better part of the underdeveloped and developing world, which cannot afford most of the drugs that are effective in treating cancer owing to the exorbitant costs associated with them. The simplicity of the synthetic route combined with the wide availability of raw materials and low waste generation make it ideal for implementation in most countries in the developing world. This route will definitely prove to be much more economical and will use renewable resources for treatment of cancer.
Conclusions:
A simple one-pot synthetic route has been developed for producing water-soluble polymers of green tea catechins effective in the treatment of cancer. The polymers have a specific inhibitory effect for cancer cells and do not significantly affect the growth of normal cells. The monomers, HRP tested in parallel, did not exhibit any inhibition. In addition, the polymers are stable in excess of 3 months under normal conditions.
Our initial studies of the anti-cancer activity were performed from a polymeric solution that could be a mixture of polymers/oligomers. The next key step is the separation of the polymers through chromatographic techniques and this leads directly into our Phase II objectives
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 5 publications | 1 publications in selected types | All 1 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Bruno FF, Nagarajan S, Nagarajan R, Kumar J, Samuelson LA. Biocatalytic synthesis of water-soluble oligo(catechins). Journal of Macromolecular Science - Pure and Applied Chemistry 2005;42A(11):1547-1554. |
SU832474 (Final) |
Exit |
Supplemental Keywords:
Enzymatic polymerization, plant peroxidases, green tea catechins, affordable therapy, environmentally friendly, polymeric flavonoids, bio-based feedstocks, cancer therapy, toxic use reduction, green chemistry, sustainable environment, technology for sustainable environment, anti-cancer drugs, biocatalytic polymerization, green cancer compounds, Sustainable Industry/Business, RFA, Technology for Sustainable Environment, Sustainable Environment, green tea flavonoids, green cancer compounds, anti-cancer drugRelevant Websites:
Center for Advanced Materials Exit
Susan Braunhut Bio Exit
P3 Phase II:
Biocatalytic Polymerization of Naturally Occurring Green Tea Flavonoids for Cancer Therapy | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.