Grantee Research Project Results
Final Report: Endothelial Cell Responses to PM—In Vitro and In Vivo
EPA Grant Number: R832414C002Subproject: this is subproject number 002 , established and managed by the Center Director under grant R832414
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: UC Davis Center for Children's Environmental Health and Disease Prevention
Center Director: Van de Water, Judith
Title: Endothelial Cell Responses to PM—In Vitro and In Vivo
Investigators: Wilson, Dennis , Barakat, Abdul , Anastasio, Cort , Tablin, Fern , Rutledge, John
Institution: University of California - Davis
EPA Project Officer: Chung, Serena
Project Period: October 1, 2005 through September 30, 2010 (Extended to September 30, 2011)
RFA: Particulate Matter Research Centers (2004) RFA Text | Recipients Lists
Research Category: Human Health , Air
Objective:
The overall goal of this project is to determine the relationship between vascular disease and systemic effects of particulate matter (PM).
Summary/Accomplishments (Outputs/Outcomes):
Our overall approach was to characterize gene and functional responses of target cell populations to collected ambient PM in vitro and compare these responses to those elicited by field exposures of mice to criteria air pollutants (CAPs). Our overall results demonstrate induction of pro-inflammatory and xenobiotic metabolism responses as consistent markers of PM exposure in pulmonary epithelial cells, aortic and pulmonary microvascular endothelial cells and monocytes in culture. Results of CAPs studies demonstrate consistent systemic pro-coagulant responses as evidenced by platelet priming in blood from animals given 2 week CAPs exposures in both rural and urban settings and in both winter and summer seasons. Systemic inflammatory responses as measured by a panel of cytokine assays were inconsistently present with significant elevations in cytokines resulting from the winter rural CAPs study but not in other locations or seasons. We asked whether pulmonary expression of pro-inflammatory adhesion molecules could be detected by immunohistochemistry in CAPs exposed mice. While we demonstrated enhanced staining in positive control mice from environmental tobacco smoke (ETS) experiments, no significant differences were found in CAPs exposed mice. We expanded our evaluation of inflammatory and xenobiotic responses in the last two CAPs studies using laser capture microdissection (LCM) to determine whether specific regions of the lung expressed transcripts of these genes. We found upregulation of CYP 1A1, a PAH inducible activity, clearly associated with CAPs exposure in both the airways and pulmonary vessels from winter urban but not summer urban exposures. We also found NOX-2, an inducible component of the reactive oxygen species (ROS) generating NADPH (nicotinamide adenine dinucleotide phosphate) oxidase system, and ICAM-1, an endothelial cell derived leukocyte adhesion molecule upregulated in the pulmonary vasculature suggesting a possible mechanism for pulmonary activation of systemic inflammatory and coagulant responses.
Our initial studies used cultured human aortic endothelial cells (HAEC) to demonstrate that collected PM2.5 stimulates pro-inflammatory and polycyclic aromatic hydrocarbon (PAH) metabolizing genes. We further examined PAH response elements and demonstrated that HAEC respond to PM2.5 by activating aryl hydrocarbon receptor (AhR) signaling. Finally, we examined the hypothesis that TGF-β family signaling was elicited by PM as it is by several other endothelial cell stimuli. We determined that PM2.5 does not stimulate TGF-β family second messenger responses. We then extended gene response studies to cultured human bronchiolar cells and determined that similar proinflammatory and PAH response genes were upregulated but that signaling activites were less evident in epithelial cells. Overall, both cell types had only modest gene responses compared with treatments with endothelial cell toxins or lipolysis products of blood lipids. Based on evidence that napthoquinone (NQ) is a photo-oxidation product of vehicular emissions, we evaluated its ROS generating capacity as free compound or bound to proteins. We found a modest ROS generation in cultured cells that was markedly enhanced by pre-binding NQ to a sulfhydryl containing protein. These results suggest that protein binding by reactive intermediates of PAH metabolism are not necessarily detoxifying reactions and that bound intermediates can retain ROS generating activity. This also implies that binding of reactive intermediates in organic fractions of airborne particulates may have a stabilizing effect that enhances their toxicity.
We next asked whether PM interaction with monocytes might contribute to systemic cytokine secretion. We examined the effects of urban source PM on activation of human monocytes and their interaction with vascular endothelium, a key event in atherogenesis. PM exposure increased cytokine expression and secretion from monocytes and enhanced monocyte adhesion to HAEC, both of which were attenuated by neutralizing endotoxin. PM also increased monocyte CYP1a1 expression, and inhibition of the AhR reduced the CYP1a1 and inflammatory responses. PM-treated monocytes accumulated intracellular ROS, and antioxidants attenuated inflammatory and xenobiotic responses. Finally, supernatants from PM-treated pulmonary microvascular endothelial cells induced monocyte inflammatory responses that were not a consequence of endotoxin transfer. These results suggest that certain components of urban PM, namely endotoxin and PAH, activate circulating monocytes directly or indirectly by first stimulating other cells such as pulmonary endothelial cells, providing several mechanisms by which PM inhalation could induce pulmonary and/or systemic inflammation.
Finally, we measured the ability of ambient fine and coarse particles to chemically generate ROS in a cell-free medium. Normalized to the volume of air sampled, fine particles generated much more hydrogen peroxide (HOOH) and hydroxyl radical (OH) than the coarse particles in the aerosol. In addition, particles from the urban (Fresno) site formed much more HOOH and OH than particles from the rural (Westside) site. Based on diagnostic tests, soluble copper was the dominant source of HOOH and OH in our particle extracts, while soluble iron was an appreciable source of OH. The rate of HOOH formation in a given extract was typically approximately 40 times greater than the rate of OH formation; however, because OH is much more reactive than HOOH, both of these oxidants could be important sources of pulmonary oxidative stress in aerosols with high mass loadings of copper and iron.
Accomplishments by specific aim:
Specific Aim 1: To characterize human endothelial cell culture responses to direct concentrated ambient PM2.5 exposure.
Microarray gene analysis in cultured HAEC exposed to PM2.5.
Characterize PAH receptor signaling in response to PM2.5
Evaluate ROS generation and associated Nrf2 signaling in PM2.5 treated HAEC
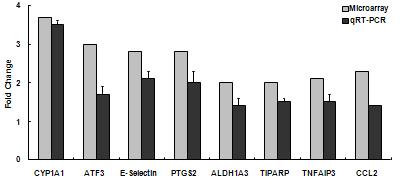
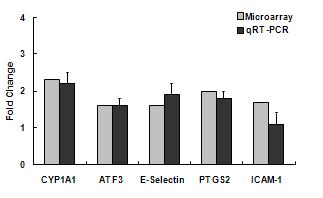
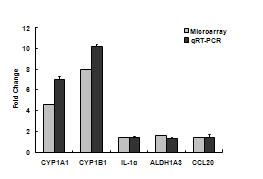
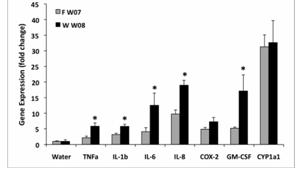
We completed a comparison of the gene responses to collected samples of summer and winter urban PM in cell cultures of aortic endothelium and bronchial epithelial cells. The number of gene responses and their magnitude varied significantly between ambient particles collected in summer or winter. Samples collected in summer up-regulated significantly more genes in both target cells than did particles from winter. In general, the fold changes resulting from particles collected in summer were more robust than those from winter. The exception to this was the responses related to xenobiotic metabolism. Responses in endothelial cells included genes associated with inflammation and xenobiotic metabolism, specifically CYP1A1, an aryl hydrocarbon response element related gene (Figure 1a and b). Associated xenobiotic response genes included aldehyde dehydrogenase and TCCD induced polyribosomal polymerase. Endothelial inflammatory responses included upregulation of E selectin and CCL2 (MCP-1). Endothelial cells also consistently responded by up-regulation of activating transcription factor 3, a transcription factor associated with the stress associated protein kinase/JNK subset of map kinase signaling.
Multiplex protein assays showed secretion of IL-6 and MCP-1 by HAEC. Because induction of CYP1A1 is mediated through the ligand-activated AhR, we demonstrated APM induced AhR nuclear translocation by immunofluorescence and western blotting and activation of the AhR response element using a luciferase reporter construct. Inhibitor studies suggest differential influences of PAH signaling, ROS mediated responses and endotoxin alter stress and pro-inflammatory endothelial cell responses (Aung, et al., Physiological Genomics, in press).
Given the unique signature of xenobiotic metabolism induction in response to PM, we further characterized activation of the AhR response element in assays demonstrating its nuclear translocation and transcriptional activation of a luciferase based AhR response element. Interestingly, both fold changes of CYP 1A1 and 1B1 as well as induction of the AhR luciferase response element (Figure 3) were higher in winter samples than those collected in summer. This is in contrast to genes more directly associated with inflammation that had more genes with significant responses and greater fold changes in cultures treated with summer source PM. This suggests that differing seasonal composition of PM alters the nature of cellular responses. Indeed, winter sample PM characterization demonstrates a higher concentration of PAH-related compounds relative to summer samples (see report for Grant No. R832414C003 for details).
Figure 1a. Gene responses in human aortic endothelial cells treated with ambient particulate matter collected from urban Fresno in Summer 2006. Initial microarray results on pooled culture treatment samples (n=4) were verified by individual RT-PCR analysis.
Figure 1b. Gene responses in human aortic endothelial cells treated with ambient particulate matter collected from urban Fresno in Winter 2007. Initial microarray results on pooled culture treatment samples (n=4) were verified by individual RT-PCR analysis.
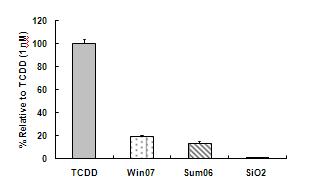
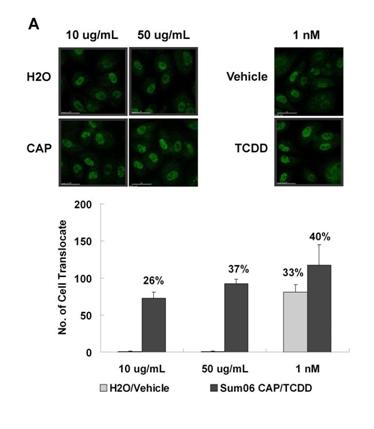
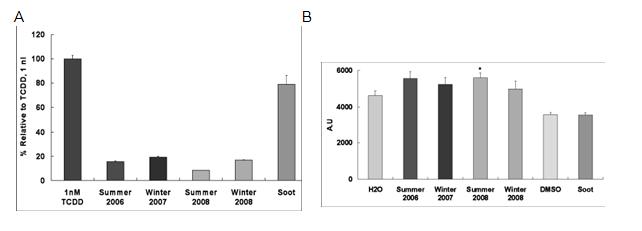
The prominent and consistent xenobiotic metabolism response in our gene transcription studies led us to further examine the role of PAH metabolism and ask whether this was a function of particulates of any sort or rather more specific to ambient PM. We compared PM from various sources with known inducers of the PAH receptor AhR using an established luciferase construct created in hepatoma cells. We found that induction by ambient PM was significantly more than that associated with similar concentrations of similarly sized synthetic silicates (Figure 2). To determine whether this response occurred through activation of AhR in endothelium, we evaluated nuclear accumulation of AhR in PM treated endothelial cell cultures (Figure 3). Further work outlined below and additionally in the progress report for R832414C004 examines the functional and toxicologic aspects of endothelial cell responses to synthetic ultrafine silicate particles as well as synthetic soot containing high levels of PAHs.
Figure 2. Activation of AhR response element by ambient particulate matter collected from urban Fresno in Summer 2006 or Winter 2007. Recombinant mouse hepatoma (H1L1.1c2) containing a stably integrated DRE-driven firefly luciferase reporter gene plasmid whose transcriptional activation occurs in an AhR-dependent manner treated with APM or TCDD (positive control). Comparison with equivalent concentration of 30 nm SiO2 particles
Figure 3. Nuclear translocation assays for AhR. HAEC treated with either summer or winter urban source APM were stained with Immunofluorescent antibodies to AhR and the percent of cells with prominent nuclear staining determined by visual counts. TCDD (1nM in EtOH) was used as a positive control. A significant response in the vehicle control for TCDD was attributed to the reported EtOH activation of AhR. A) Representative images for cells treated with summer APM. B) Percent nuclear staining for summer APM treated cells.
Pulmonary Microvascular Endothelium
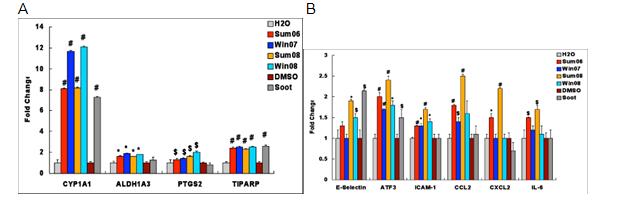
Because of our ongoing interest in the interface between the pulmonary microvasculature and systemic inflammatory responses, we did additional experiments on cultured human pulmonary microvascular endothelial cells (HPMVEC) as in vitro targets for toxicity. We used HPMVEC for comparative response studies between ambient PM collected in different sites and seasons as well as synthetic fine and ultrafine particulates. We also used laboratory generated carbon particles containing PAHs (soot) for comparison. We find the trends for summer pro-inflammatory responses and winter PAH responses to be consistent between sites (Figure 4A and B). Soot treatment also upregulated AhR responses but was less potent in induction of inflammatory responses. In addition to PAH related metabolism, ambient PM upregulated prostagladin S2 synthase (COX-2) and the antioxidant response enzyme Aldehyde dehydrogenase.
Figure 4A. Induction of CYP1A1, ALDH1A3, PTGS2 and TIPARP gene expression by urban and rural APM from different seasons and soot. Urban and rural APM were compared to H2O (control) and Soot was compared to DMSO (control). N = 4, # = P≤0.0005, * = P ≤0.005, $ = P ≤0.05.
B. Induction of E-Selectin, ATF3, ICAM-1, CCL2, CXCL2 (MIP-2α) and IL-6 by urban and rural APM for different season and Soot. Urban and rural APM were compared to H2O (control) and Soot was compared to DMSO (control). N = 4, # = P ≤0.0005, * = P ≤0.005, $ = P ≤0.05.
To confirm AhR selectivity and promotional activation, we used the luciferase based assay in a hepatocyte tumor line construct (Figure 5A). Relative to the inactivity we reported above for similar concentrations of silica, soot was more active in stimulating AhR responses than ambient PM from all sources. This finding is somewhat tempered by compositional differences in that 100% of the soot was carbon particles containing PAHs while ambient source material contains a large proportion of salts that likely are soluble under in vitro conditions. To determine whether APM or soot preparations were likely to induce apoptosis, we evaluated caspase 3 activation as a marker of apoptosis (Figure 5B). APM was significantly more inductive of apoptosis than soot.
Figure 5A. Induction of luciferase in H1L1.1c2 cells containing a stably integrated AhR responsive DRE-driven firefly luciferase reporter gene plasmid. Response to a 3 hour incubation of 10 µg/mL APM collected in Summer 2006 or Winter 2007 or Summer 2008 or Winter 2008 or Soot was compared to TCDD (100% as positive control). N=3.
B. APM induced caspase3/7 activity was increased significantly by APM collected in Summer 2008. Urban and rural APM were compared to H2O (control) and Soot was compared to DMSO (control). N = 5, * = P ≤0.05
Role of Composition in Selective Endothelial Responses to Collected Ambient PM:
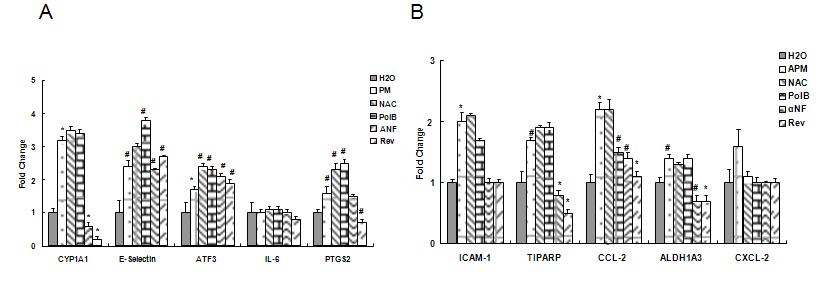
We examined the potential role of compostition in driving endothelial responses using inhibitors to determine whether PAH, metal, ROS generation or endotoxin components of PM drive the pattern of gene responses we have described. Based on experiments with monocytes, described below, we used inhibitors of AhR activation (α-napthoflavone) endotoxin (polymyxin B) and ROS generation (n-acetyl cysteine and resveratrol) to determine whether gene responses in endothelial cells were selectively attenuated by any of these treatments. As expected α-naphthoflavone treatment inhibited AhR mediated CYP 1A1 upregulation. This upregulation also was inhibited by antioxidant treatment with resveratrol but not n-acetyl cysteine. In general, antioxidant treatment effectively reduced all inflammatory responses. Binding endotoxin with polymyxin B prevented cytokine synthesis but not transcription of adhesion molecules (Figure 6 A and B) (Aung, et al., Physiological Genomics, in press).
Figure 6. Effect of AhR inhibition (α-napthoflavone, α-NF) endotoxin binding (polymyxin B, PolB) or antioxidant treatment (n-acetyl cysteine and resveratrol) on gene responses (RT-PCR) to collected urban summer source ambient particulate matter.
Bronchial Epithelial Cell Responses in Vitro:
While the initial aim of this subproject was to evaluate endothelial responses, our growing perspective that there was an interface between pulmonary inflammation and systemic responses led us to ask whether correlative patterns of gene responses were elicited from cultured bronchial epithelium exposed to collected ambient particulates.
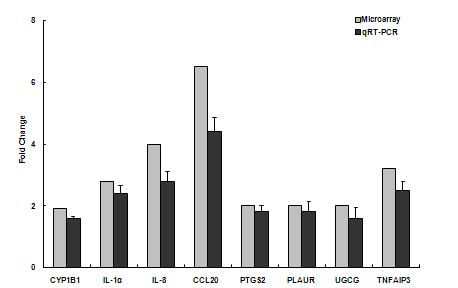
Human bronchial epithelial cells also responded with a correlative mixture of pro-inflammatory genes and those involved with xenobiotic metabolism. In contrast to endothelium, HBEC upregulated CYP1B1 and 1A1. Of particular importance relative to responses associated with asthma, HBEC had highest fold changes for the dendritic cell chemotactant, CCL20. Interleukin 8 and IL-1a also were prominent in the HBEC response (Figure 7 A and B).
Figure 7A. Gene responses in human bronchial epithelial cells treated with ambient particulate matter collected from urban Fresno in Summer 2006. Initial microarray results on pooled culture treatment samples (n=4) were verified by individual RT-PCR analysis.
Figure 7B. Gene responses in human bronchial epithelial cells treated with ambient particulate matter collected from urban Fresno in Winter 2007. Initial microarray results on pooled culture treatment samples (n=4) were verified by individual RT-PCR analysis.
Again, we asked whether these responses were ambient PM specific, this time comparing urban source PM with soot from wildfire collected during a significant fire-based air pollution event. We examined the effects of urban ambient particulate matter (APM) collected from Fresno, California, and wildfire (WF) particulate matter collected from Escalon, California on early transcriptional responses in human bronchial epithelial cells (HBE). We probed for biomarkers of inflammation, oxidative stress, and xenobiotic metabolism. Initial microarray analysis of APM treated HBE activated genes related to epithelial cell biology or cellular responses to chemical challenge, including CCL20 and CYP1B1. Inhibitor studies targeting endotoxin, ROS, and trace metals found that APM induce an inflammatory profile in addition to biomarkers of oxidative stress. Inhibition of trace metals and endogenous ROS in WF PM treatments also induce a pro-inflammatory gene response, but elicit a more robust xenobiotic profile and oxidative stress induction (Nakayama, et al., Toxicology in Vitro, in press).
Toxicologic Effects of PAH Metabolites on Target Cells in Vitro:
The consistent gene transcription and functional evidence of PAH involvement in PM toxicity stimulated an investigation of the cellular consequences of PAH metabolites on toxicity in target cells. This study was done in correlation with other studies in our laboratory, funded by other sources, investigating the consequence of protein binding of reactive intermediates of the endothelial cell toxin monocrotaline pyrrole. We chose the reactive intermediate of naphthalene, napthoquinone (NQ) as a model PM associated PAH. We asked whether these reactive intermediates were detoxified by binding to cellular proteins and whether differences in their metabolism and cellular interactions resulted in compound specific cellular responses. We treated human pulmonary artery endothelial cells (HPAEC) with NQ, MCTP or preformed NQ or MCTP adducts to the protein galectin-1 (gal-1) and examined indicators of ROS oxidative injury and markers of apoptosis (caspase-3 and annexin V). ROS production was assayed fluorescently using CM-H2DCFDA. NQ adducts to gal-1 (NQ-gal) produced 183% more intracellular ROS than gal-1 alone (p<.0001). Caspase-3 activity and annexin V staining of phosphatidylserine were used to assess apoptotic activity in treated cells. HPAEC exposed to MCTP-gal had increases in both caspase-3 activation and membrane translocation of annexin V relative to gal-1 alone (p<.0001). Direct application of NQ produced significantly more ROS and induced significant caspase-3 activation, whereas MCTP did not. Western blot analysis showed that both NQ and MCTP significantly induced the Nrf2 mediated stress response pathway despite differences in ROS generation. We concluded that protein binding was not a detoxification reaction but rather served to stabilize cytotoxic potential. Furthermore, electrophile adduction to gal-1 produces different cytotoxic effects specific to each reactive intermediate. These results provide new conceptual information about the cellular toxicity of inhaled PM (Nakayama, et al., Chemical Research in Toxicology, 2010).
Specific Aim 2: To determine the effects of direct PM exposure on permeability and pro-coagulant activity in endothelium.
Monolayer Permeability in Endothelial Cell Cultures
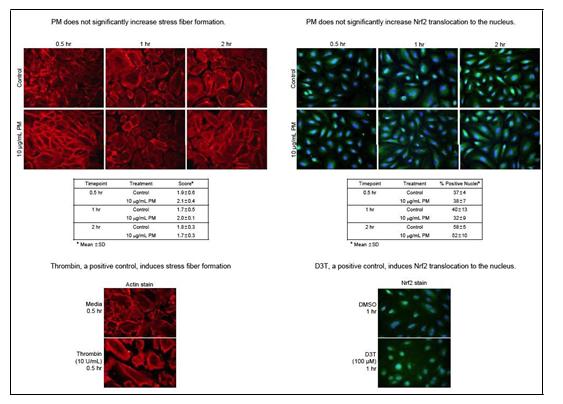
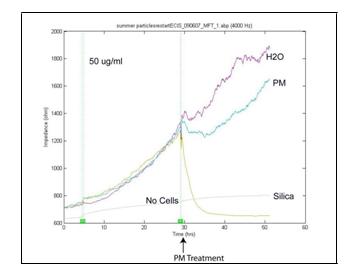
We have performed experiments to determine the effects of PM2.5 on endothelial cell barrier permeability. We examined the effect of rural winter source APM on the actin cytoskelton of HAEC and in related experiments evaluated the activation of the Nrf2 oxidant response element. We found that ambient PM had no effect on either system in vitro (Figure 8). Similarly, we asked whether treatment with ambient PM altered endothelial cell monolayer barrier permeability using a high throughput real time electrical resistance-based permeability system. We compared treatment with rural winter source APM with the response to an equivalent concentration of 30nM SiO2. Surprisingly, the SiO2 treatment had a significant effect on barrier function while APM, even at a relatively high concentration of 50 µg/ml was without effect (Figure 9).
Figure 8. Effects of APM treatment on HAEC monolayer actin cytoskeleton and Nrf-2 nuclear translocation. Positive control for cytoskeletal rearrangement was treatment with Thrombin while Nrf-2 positive control treatment used dithiothreatol (D3T).
Figure 9. Transendothelial impedance measurements in HAEC treated with either 10 or 50 µg/ml summer urban source APM or 10 µg/ml 30 nm SiO2 particles.
Platelet Activation and Systemic Markers of Coagulation and Inflammation
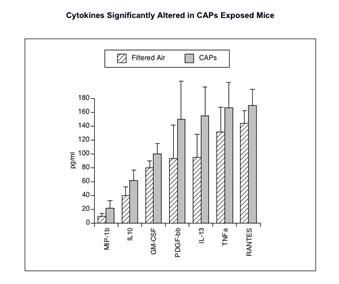
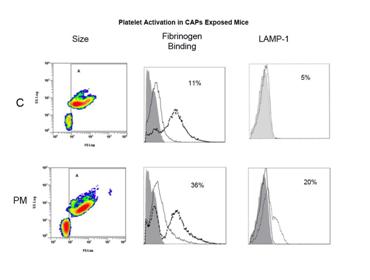
We have accomplished studies of platelet activation and systemic markers of coagulation and inflammation in CAPs exposures to mice. Our initial study demonstrated activation of systemic cytokines (Figure 10) and platelet priming (Figure 11) in CAPs exposed mice at our rural location in winter (Wilson, et al., Inhalation Toxicology, 2010). We performed similar analyses for field exposures in urban Fresno in summer 2008 and winter 2009. Although evidence of platelet priming was present in CAPs exposed mice from both these studies, the systemic cytokine response was not evident in either.
Figure 10. Significantly altered cytokine responses in blood of mice exposed to Winter Rural source CAPs for 2 weeks.
Figure 11. Platelet activation in blood of mice exposed to Winter Rural source CAPs for 2 weeks. Platelet from exposed mice had an aggregated population that was absent in control mice and significant increases in fibrinogen binding activity as well as lysosomal secretion (degranulation) as determined by surface expression of Lysosomal Associated Membrane protein 1 (LAMP-1).
Specific Aim 3: To compare the nature and location of endothelial cell responses in pulmonary and cardiac tissue from CAPs exposed mice.
We completed evaluation of standard histology and immunohistochemical analysis of lung and heart sections from mice in the 2007-2009 field studies that included summer and winter rural and urban exposures. There are no evident inflammatory changes in H+E sections and little evidence of endothelial cell activation by immunohistochemistry. We further evaluated the nasal cavities of CAPs exposed mice to determine whether evidence of local inflammation there could possibly contribute to the systemic responses we observed. We found no treatment-associated differences in sections of nasal cavities by histologic evaluation.
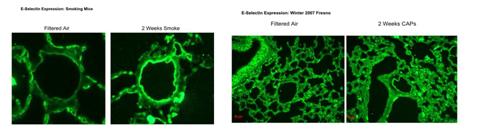
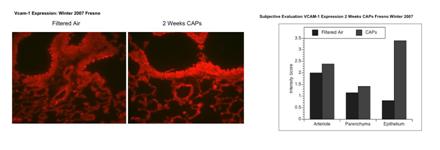
We developed immunofluorescent probes for VCAM-1 and E-selectin and verified their use in lungs of mice exposed to ETS. Figure 12 demonstrates positive responses for E-selectin expression in arterioles of mice exposed for 2 weeks to ETS. Figure 13 shows a relative lack of responses in the lungs of mice exposed for 2 weeks to CAPs. An apparent upreguation of VCAM-1 in airway epithelium of urban CAPs exposed mice from Winter 2007 (Figure 14) was not replicable in exposures from rural exposures in Winter 2008 (data not shown).
Figures 12 and 13. Expression of E-Selectin in lungs of mice exposed to either ETS or CAPs for 2 weeks.
Figure 14. Expression of VCAM-1 in airway epithelium in response to a 2 week CAPs exposure: A) Immunohistochemistry, B) Subjective evaluation of VCAM and E- Selectin Expression.
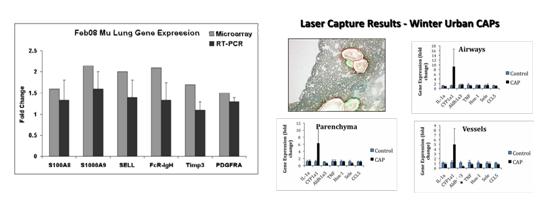
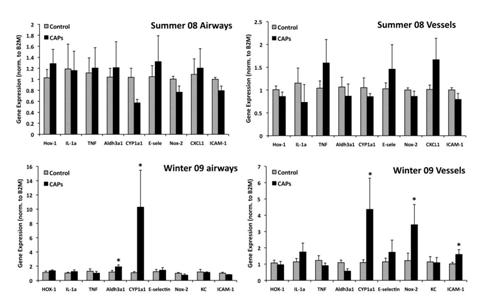
We also performed microarray studies of RNA isolated from these field studies and found inconsistent responses in whole lung gene arrays. PCR analysis showed no significant responses in selected genes between control and CAPs exposed animals and very little response in laser capture studies of gene responses in specific lung sub- compartments was evident (Figure 15).
Figure 15A. Gene expression by microarray and RT-PCR from whole lung of mice exposed to rural winter CAPs.
B. Gene expression by RT-PCR of airways, vessels and alveolar parenchyma isolated from frozen sections of lungs from mice exposed to winter urban CAPs using laser capture microdissection. Upregulation of CYP1A1 was evident in some but not all CAPs exposed animals; this was not statistically significant and no quantitative changes in other genes examined were found.
BAL cytokine assays were performed with material from mice exposed to rural CAPs in Winter 2008. These assays did not detect significant levels of any of the cytokines in the panel in either control or treated mice.
We repeated these studies with additional probes and included samples from mice from urban summer exposures (Summer 2008) (Figure 16). As expected, we found strong induction of CYP1A1 in all segments of lung selected. Interestingly, we found evidence of endothelial cell activation in vessels as shown by upregulation of NOX-2, a component of the NADPH oxidase system, and the endothelial cell specific leukocyte adhesion molecule ICAM-1. These responses were limited to mice exposed to winter source CAPs.
Figure 16. Gene expression by RT-PCR of airways, vessels and alveolar parenchyma isolated from frozen sections of lungs using laser capture microdissection of mice exposed to urban CAPs in either summer or winter.
With additional support from the California Air Resources Board, we have been translating our in vitro findings to in vivo experiments with intra-tracheal instillations of collected ambient PM. To more directly control for dose and time course of changes, we performed intra-tracheal instillations of collected ambient PM. Although results of these experiments are only partially complete, several key findings have emerged. First, intra-tracheal (IT) instillation reliably replicates activation of circulating platelets as we have previously described in CAPs studies. Second, IT experiments elicit regional pulmonary inflammatory responses that center on small pulmonary arterioles.
Specific Aim 4: To determine the effects of CAPs exposure on monocyte activation.
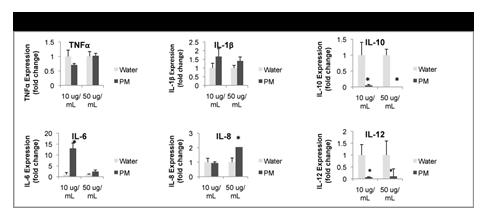
We examined the effects of winter source urban PM on activation of human monocytes and their interaction with vascular endothelium, a key event in atherogenesis. PM exposure increased cytokine expression and secretion from monocytes and enhanced monocyte adhesion to human aortic endothelial cells, both of which were attenuated by neutralizing endotoxin. PM also increased monocyte CYP1a1 expression, and inhibition of the AhR reduced the CYP1a1 and inflammatory responses. PM-treated monocytes accumulated intracellular ROS, and antioxidants attenuated inflammatory and xenobiotic responses. Finally, supernatants from PM-treated pulmonary microvascular endothelial cells induced monocyte inflammatory responses that were not a consequence of endotoxin transfer. These results suggest that certain components of urban PM, namely endotoxin and PAH, activate circulating monocytes directly or indirectly by first stimulating other cells such as pulmonary endothelial cells, providing several mechanisms by which PM inhalation could induce pulmonary and/or systemic inflammation (den Hartig, et al., Toxicology in Vitro, 2009).
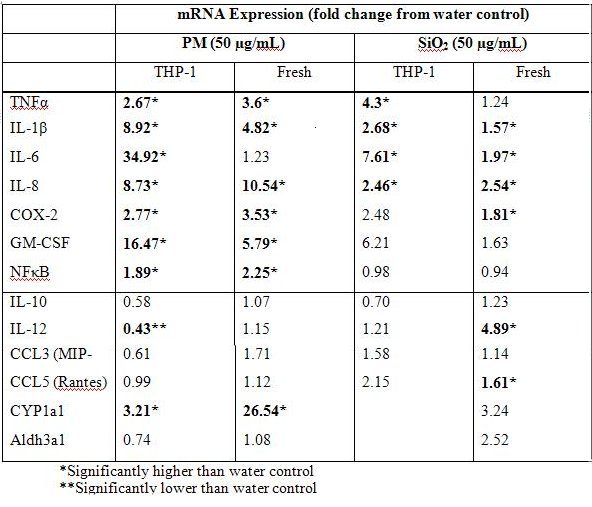
We confirmed these studies with freshly isolated human monocytes and extended them to studies with similarly sized synthetic particulates to ask whether a similar upregulation of AhR related gene responses occurs (Table 1). We found upregulation of CYP1A1 in monocyte cell lines and an even more robust response in freshly isolated human monocytes. Although treatment with SiO2 increased some pro-inflammatory genes, this response was markedly less than in response to PM treatment, especially in fresh monocyte isolations.
Table 1. Summary of gene expression from THP-1 and fresh monocytes treated with APM and SiO2.
In additional experiments, we asked whether pulmonary microvascular endothelium secretes activities that in turn stimulate monocyte responses. We find that monocytes treated with supernatants of PM treated HPMVECs have a similar pro-inflammatory gene response to that resulting from direct PM treatment (Figure 17).
Figure 17. Pro-inflammatory gene activation in monocytes incubated with supernatants from HPMVEC treated with rural winter source ambient PM.
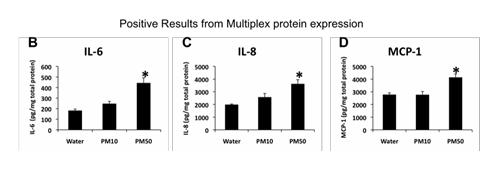
We further asked whether specific pro-inflammatory mediators could be demonstrated in EC supernates using a multiplex protein assay system. Of 32 activities evaluated, only IL6, IL8 and MCP-1 were present in greater than background expression levels (Figure 18).
Figure 18. Positive results of multiplex (Bioplex) assays for cytokines in HPMVEC treated with rural winter source ambient PM.
Monocyte Responses as Biomarkers of Source Specific Changes:
Results of our monocyte experiments suggested that these results could be used to compare location and season source responses. We asked whether differences in monocyte responses occurred between our rural and urban source PM. We found that rural source PM had a greater inflammatory response than urban but that there was no difference in PAH-related responses (Figure 19).
Figure 19. Comparative gene responses to urban (F) or rural (W) winter source ambient PM treatment of monocytes in vitro.
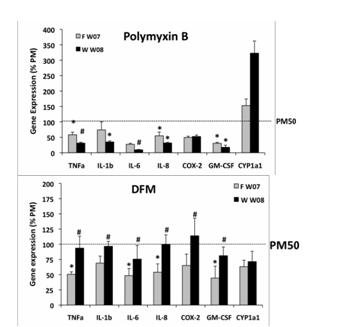
We next asked whether differing components of PM selectively stimulated specific responses using inhibitors of transition metals (DFM chelation) AhR activation (α-napthoflavone), endotoxin (polymixin B) and intracellular and extracellular ROS generation (n-acetyl cysteine and catalase, respectively). As might be predicted, metal chelation and intercellular ROS quenching inhibited inflammatory gene responses but did not alter PAH-related responses. Endotoxin binding by polymixin B significantly inhibited pro-inflammatory responses. Using these inhibitors, we compared winter rural and urban source PM. We found that endotoxin binding was more effective in blunting responses to rural source PM while metal chelation uniquely inhibited urban source PM pro-inflammatory genes (Figure 20 A and B).
Figure 20. Percent inhibition of pro-inflammatory gene upregulation by: A. endotoxin binding (poymixin B treatment) or B. metal chelation (DFM). Monocytes treated with urban source (F) or rural source (W) PM were compared in the presence or absence of indicated inhibitors.
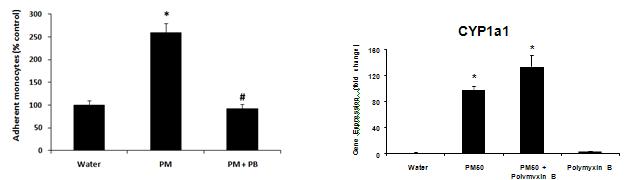
We extended these studies to show that polymyxin B treatment abrogates the PM induced adhesion of monocytes to endothelium. This suggests that LPS is the key component driving pro-inflammatory responses. Conversely, polymyxin B treatment has no effect on CYP 1A1 transcription suggesting that the PAH response pathway is separate from the pro-inflammatory responses (Figure 21).
Figure 21. Comparison of polymyxin B inhibition of endotoxin on monocyte adhesion to endothelium or induction of CYP1A1 gene transcription in response to rural winter source PM.
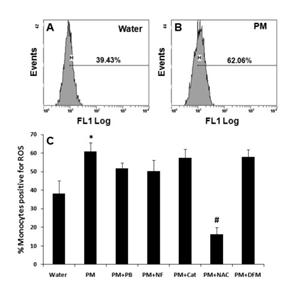
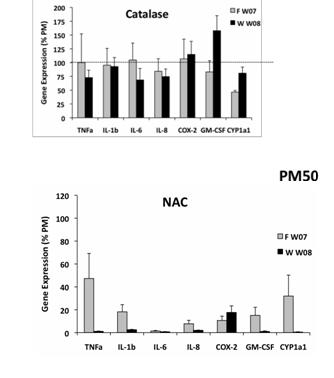
Finally, we asked whether ROS generation occurs in PM-treated monocytes and whether this is an extracellular or intracellular phenomenon. We demonstrated that PM-treated monocytes have increased ROS in a flow cytometric assay (Figure 22). Furthermore, we find that inhibition of extracellular ROS by pretreatment with catalase does not influence PM-induced pro-inflammatory responses but that inhibition of intracellular ROS by NAC greatly blunts inflammatory gene responses (Figure 23).
Figure 22. Flow cytometric analysis of intracellular ROS in monocytes treated with winter rural source PM.
Figure 23. Comparison of inflammatory gene responses in monocytes treated with ambient PM in the presence of catalase or n-acetyl cysteine (NAC).
ROS Generation in Cell-free Assays:
As described above, in several cases the cellular responses to ambient particles apparently are driven by the generation of ROS. As a complement to these cellular studies, we measured the ability of the Fresno and Westside particles to chemically form ROS in cell-free assays. We used two specific, quantitative assays – one for hydrogen peroxide (HOOH) and one for hydroxyl radical (OH). In each assay, we took a filter punch of the high-volume PM sample (collected during the R832414C003 [Project 3] animal exposures from 2006 to 2009) and extracted it in phosphate-buffered saline (PBS), generally in the presence of 50 µM ascorbate.
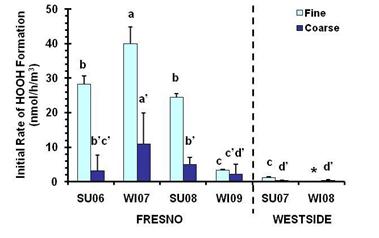
Figure 24 summarizes some of the results from our HOOH measurements (Shen, et al.,2011). We found that Fresno (urban) PM generally generates much more HOOH than Westside (rural) PM and that, normalized by air volume, PM2.5 generally makes more HOOH than the corresponding coarse PM (PMcf, i.e., particles with diameters between 2.5 and 10 µm). PM2.5 dominates primarily because the mass concentration of PM2.5 is much higher than that of PMcf in the ambient aerosol samples.
Figure 24. Air-volume-normalized initial rates of HOOH generation in the presence of 50 µM ascorbate in PBS. Key to sample names: SU = summer, WI = winter, 0x = year (200x). Values are means ± SD, n = 3. Letters above bars indicate statistically different rates: a > b > c for fine PM, while a’ > b’ > c’ > d’ for coarse PM. The asterisk for the Westside Winter 2008 sample indicates that HOOH formation was not statistically different from 0.
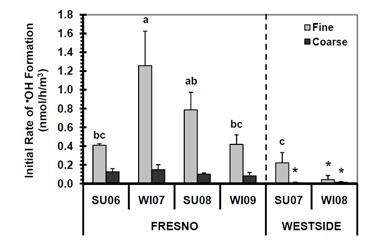
As shown below in Figure 25, OH generation by the particles is broadly similar to HOOH generation (Shen and Anastasio, 2011): on an air-volume-normalized basis, Fresno PM forms much more OH than Westside PM, while at both sites, PM2.5 forms more OH than PMcf. HOOH and OH formation are well correlated in the particle extracts, with an R2 value of 0.75. The major difference in the formation of the two oxidants is that HOOH is much more abundant, with an initial rate of formation that is approximately 40 times greater than that of OH in a given particle extract (Shen and Anastasio, 2012). However, because OH is much more reactive than HOOH, it is difficult to determine from our results which species might be more important for toxicity. Extrapolating our results to expected concentrations of PM-derived HOOH and OH in human lungs suggests that typical daily PM exposures in the San Joaquin Valley are unlikely to cause ROS-mediated acute health effects, but that very high PM events might lead to cytotoxic levels of pulmonary ROS.
Figure 25. Air-volume-normalized initial rates of OH generation in the presence of 50 µM ascorbate in PBS. The sample names and symbol keys are the same as in Figure 24.
We next explored the mechanisms, and particle components, responsible for the formation of ROS in the cell-free assays, starting by examining the impact of 50 μM ascorbate. For the Fresno samples, the addition of ascorbate greatly enhances HOOH and OH generation, by a typical factor of approximately 50 to 100. This indicates that the chemical generation of ROS by PM is much more rapid in the presence of physiological levels of this reductant; it also suggests that assays without ascorbate greatly underestimate the chemical formation of ROS by PM.
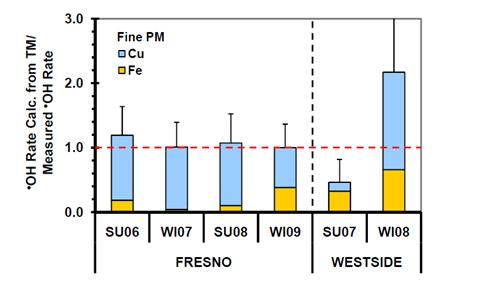
In our second step to explore the mechanisms of ROS generation, we examined the impact of adding a transition metal chelator – desferoxamine (DSF) – to our extract solutions. DSF greatly reduced both HOOH and OH generation from SJV PM: rates of production were, on average, lower by (78±15)% for HOOH and (97±6)% for OH in the presence of DSF. This indicates that transition metals play a dominant role in ROS generation in our extracts. To more specifically probe the roles of individual metals, we measured concentrations of several transition metals (including Fe, Cu, V, Ni, and Mn) in our PM extracts and quantified the formation of OH and HOOH from different concentrations of these metals in laboratory PBS solutions. Combining these data with the measured amounts of ROS formed in each particle extract allows us to estimate the fractions of HOOH and OH that were formed from each metal. As illustrated in Figure 26 for OH formation from fine particles, copper is the dominant source of ROS in both the fine and coarse ambient PM: on average, soluble Cu accounts for (89±18)% of the initial rate of OH generation in the Fresno PM2.5 extracts and (89±23)% in the Fresno PMcf extracts (Shen and Anastasio, 2011). The remainder of the OH reactivity in the Fresno particles is due to soluble Fe. Copper is even more important for HOOH formation, able to account for essentially all of its formation. This dominant role of copper is supported by the fact that of all the transition metals, only Cu was strongly correlated with the rates of HOOH and OH formation. Although we do not know the source of Cu in our samples, brake wear particles are probably important and have been previously shown to contribute significant amounts of copper to both fine and coarse mode aerosols.
Figure 26. Contributions of PBS-soluble Cu (blue bars) and Fe (yellow bars) to the initial rate of OH generation in PBS with 50 μM ascorbate in fine particle extracts. The y-axis value is the fraction of OH generation that can be accounted for by Cu and Fe, i.e., a value of 1 or above indicates that Cu and Fe can account for all of the OH formed. Values are means ± SD, n=3.
Journal Articles on this Report : 9 Displayed | Download in RIS Format
| Other subproject views: | All 15 publications | 9 publications in selected types | All 9 journal articles |
|---|---|---|---|
| Other center views: | All 128 publications | 71 publications in selected types | All 64 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Aung HH, Lame MW, Gohil K, He G, Denison MS, Rutledge JC, Wilson DW. Comparative gene responses to collected ambient particles in vitro:endothelial responses. Physiological Genomics 2011;43(15):917-929. |
R832414 (Final) R832414C002 (Final) |
Exit Exit Exit |
|
|
den Hartigh LJ, Lame MW, Ham W, Kleeman MJ, Tablin F, Wilson DW. Endotoxin and polycyclic aromatic hydrocarbons in ambient fine particulate matter from Fresno, California initiate human monocyte inflammatory responses mediated by reactive oxygen species.Toxicology In Vitro 2010;24(7):1993-2002. |
R832414 (2010) R832414 (Final) R832414C002 (2010) R832414C002 (Final) |
Exit Exit Exit |
|
|
Nakayama Wong LS, Lame MW, Jones AD, Wilson DW. Differential cellular responses to protein adducts of naphthoquinone and monocrotaline pyrrole. Chemical Research in Toxicology 2010;23(9):1504-1513. |
R832414 (2010) R832414 (Final) R832414C002 (2010) R832414C002 (Final) |
Exit Exit Exit |
|
|
Nakayama Wong LS, Aung HH, Lame MW, Wegesser TC, Wilson DW. Fine particulate matter from urban ambient and wildfire sources from California's San Joaquin Valley initiate differential inflammatory, oxidative stress, and xenobiotic responses in human bronchial epithelial cells. Toxicology In Vitro 2011;25(8):1895-1905. |
R832414 (Final) R832414C002 (Final) |
Exit Exit Exit |
|
|
Shen H, Barakat AI, Anastasio C. Generation of hydrogen peroxide from San Joaquin Valley particles in a cell-free solution. Atmospheric Chemistry and Physics 2011;11(2):753-765. |
R832414 (2010) R832414 (Final) R832414C002 (2010) R832414C002 (Final) |
Exit Exit |
|
|
Shen H, Anastasio C. Formation of hydroxyl radical from San Joaquin Valley particles extracted in a cell-free surrogate lung fluid. Atmospheric Chemistry and Physics 2011;11(18):9671-9682. |
R832414 (Final) R832414C002 (Final) |
Exit Exit |
|
|
Shen H, Anastasio C. A comparison of hydroxyl radical and hydrogen peroxide generation in ambient particle extracts and laboratory metal solutions. Atmospheric Environment 2012;46:665-668. |
R832414 (Final) R832414C002 (Final) |
Exit Exit Exit |
|
|
Tablin F, den Hartigh LJ, Aung HH, Lame MW, Kleeman MJ, Ham W, Norris JW, Pombo M, Wilson DW. Seasonal influences on CAPs exposures:differential responses in platelet activation, serum cytokines and xenobiotic gene expression. Inhalation Toxicology 2012;24(8):506-517. |
R832414 (Final) R832414C002 (Final) |
Exit |
|
|
Wilson DW, Aung HH, Lame MW, Plummer L, Pinkerton KE, Ham W, Kleeman M, Norris JW, Tablin F. Exposure of mice to concentrated ambient particulate matter results in platelet and systemic cytokine activation. Inhalation Toxicology 2010;22(4):267-276. |
R832414 (2010) R832414 (Final) R832414C002 (Final) R832414C003 (2010) R832414C003 (Final) |
Exit |
Supplemental Keywords:
Health, RFA, Scientific Discipline, Air, Health Risk Assessment, Risk Assessments, particulate matter, Environmental Chemistry, Epidemiology, ambient aerosol, lung disease, human exposure, long term exposure, airborne particulate matter, concentrated air particles, epidemiological studies, PM, ambient particle health effects, toxicology, inhalation toxicology, vascular dysfunction, cardiovascular disease, human health risk, lung injury, acute cardiovascular effects, microarray studies, air toxicsProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R832414 UC Davis Center for Children's Environmental Health and Disease Prevention Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R832414C001 Project 1 -- Pulmonary Metabolic Response
R832414C002 Endothelial Cell Responses to PM—In Vitro and In Vivo
R832414C003 Project 3 -- Inhalation Exposure Assessment of San Joaquin Valley Aerosol
R832414C004 Project 4 -- Transport and Fate Particles
R832414C005 Project 5 -- Architecture Development and Particle Deposition
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- 2010 Progress Report
- 2009 Progress Report
- 2008 Progress Report
- 2007 Progress Report
- 2006 Progress Report
- Original Abstract
9 journal articles for this subproject
Main Center: R832414
128 publications for this center
64 journal articles for this center