Grantee Research Project Results
Final Report: Conducting-Polymer Nanowire Immunosensor Arrays for Microbial Pathogens
EPA Grant Number: GR832375Title: Conducting-Polymer Nanowire Immunosensor Arrays for Microbial Pathogens
Investigators: Mulchandani, Ashok , Chen, Wilfred , Yates, Marylynn V. , Myung, Nosang V.
Institution: University of California - Riverside
EPA Project Officer: Hahn, Intaek
Project Period: June 1, 2005 through March 31, 2008 (Extended to March 31, 2009)
Project Amount: $320,000
RFA: Greater Research Opportunities: Research in Nanoscale Science Engineering and Technology (2004) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
A promising approach for the direct (label-free) electrical detection of biological macromolecules uses one-dimensional (1-D) nanostructures such as nanowires and nanotubes, configured as field-effect transistors that change conductance upon binding of charged macromolecules to receptors linked to the device surfaces. Combined with simple, rapid and label-free detection, potentially to single molecule, these nanosensors also are attractive due to the small size, low power requirement and most of all the possibility of developing high density arrays for simultaneous analyses of multiple species.
The overall objective of the research is to develop a method for fabrication of a bioreceptor (antibody)-functionalized conducting polymer nanowire based immunosensor and to demonstrate its application for label-free, real-time, rapid, sensitive and cost-effective detection of multiple pathogens in water/environment.
Summary/Accomplishments (Outputs/Outcomes):
1. Fabrication of single conducting polymer nanowire based field-effect transistor:
1.1. Synthesis of conducting polymer nanowire: Conducting polymer polypyrrole (Ppy) nanowires were electrochemically synthesized using a well established template directed electrodeposition technique (Figure 1).
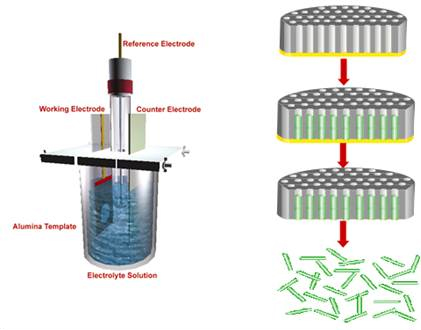
Figure 1. Schematic of template method for nanowire synthesis.
Alumina membrane of 200 nm pore size and 60 µm thickness (Whatman International Ltd., Maidstone, England) was used as a scaffold for nanowire fabrication. Seed layer was deposited by sputtering ~200 nm thick gold using the Emitech K550 (Emitech Ltd., Kent, England) sputter coater on one side of the alumina template. Electrodeposition was carried out in a three electrode cell with the gold-coated alumina template as the working electrode, Ag/AgCl as reference and platinum coated titanium strip as the counter electrode using a CH Instrument Model 760C (CH Instruments, Austin, TX, USA) electrochemical analyzer. 0.5 M pyrrole (Sigma Aldrich) in 0.2 M LiClO4 (Aldrich, Milwaukee WI USA) was used as an electrolyte solution. Chronocoulometry method was used for electrodepositing Ppy nanowires by passing 0.7 C charge at 0.9 V. The gold seed layer was removed using 0.15 M KI in 0.1 N I2 gold-etchant solution. After washing the alumina template with water, it was dissolved in 30% H3PO4 acid (Acros organic) and briefly sonicated to free the nanowires to form a suspension. Nanowires were washed with nanopure water and resuspended in nanopure water. The suspension was diluted 10-fold for further use.
1.2. Fabrication of single nanowire field-effect transistor/chemiresistor: A chromium (Cr) adhesion layer of 200 A° and 1800 A° thick gold (Au) contact layer was deposited on (100) oriented Silicon wafer with a 300 nm SiO2 layer. An array of 16 pairs of gold microelectrodes with ~70 µm separation between adjacent pairs with each pair containing rectangular electrodes of ~55 µm width separated by 3 µm gap were used as contact electrodes for device assembly as shown in Figure 2 (a).
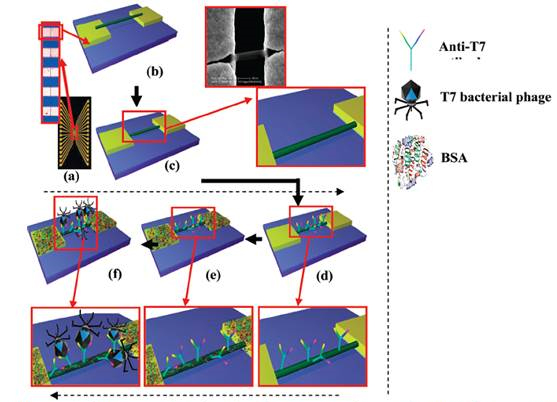
Figure 2. a) photo image of 16 electrodes chip (inset optical image showing gap between two electrodes).
Schematic of b) alighed and c) ancholred Ply nanowire on the gold electrodes with three µm gap (inset:
blown up schematic and SEM inage). Schmatics with blown up of d) aniti-T7 finctionalized Ppy nanowire.
e) BSA blockign after antibody-functionalization and f) T7 phage interactes with anti-T7 antibody on the nanowire surface.
These electrodes were cleaned with piranha solution (3.5 ml of concentrated H2SO4 + 1.5 ml of H2O2) (Fisher Scientific, Fair Lawn, New Jersey, USA). The two sides of 16-pairs were shorted to form two terminals. Ppy nanowires were aligned in the electrode gap by passing alternating current field of 4 MHz frequency and 3 V peak to peak between the two terminals. A 2 µl drop of Ppy nanowire suspension was dispensed on it and alignment was carried out for about a minute. To achieve single nanowire connection between a pair of contact electrodes as in Figure 2 (b), excess nanowires were physically/manually removed using a probe tip made out of 25 µm diameter gold wire under a 1000x magnification optical microscope. In order to secure the Ppy nanowire on to the gold electrodes, the nanowire was anchored with maskless electrodeposition (Figure 2 (c)), using chronoamperometry method (CH Instrument Model 760C Electrochemical Analyzer). A three-electrodes electrochemical cell consisting of the 16-pairs of contact electrodes with single Ppy nanowire connections as the working electrode, Ag/AgCl as a reference electrode and a platinum coated metal strip as a counter electrode were used. The electrolyte was Technigold (Technic Inc., California, USA) at pH of 7.0 and the deposition potential used was -0.5 V vs Ag/AgCl at room temperature for 10 min for build up of a ~300 nm thick gold layer on the gold electrode surface.
1.3. Biological functionalization: Single Ppy nanowire devices were incubated for 3 hrs in 10 µl of 60 mM 1-Ethyl-3-[3-dimethylaminopropyl]carbodiimide hydrochloride (EDC) (Sigma Chemical Co. St Louis, MO, USA) solution in 0.1 M MES buffer at a resulting pH of ∼5.5, mixed with 100 µl N-hydrosuccinimide (NHS) in dimethyl sulfoxide (DMSO) (2 mg/mL) (Thermo Fisher Scientific, Rockford, IL, USA) and 10 µl anti-T7 (Millipore Corporate Billerica, MA, USA) or anti-MS2 antibody (Tetracore, Inc., Rockville, MD USA) as shown in Figure 1 (d). The excess reagent was subsequently removed and the devices were washed with 1% Tween-20 in PB buffer (PBT) followed by PB. To block nonspecific interactions between biomolecules and the nanowire, devices were incubated in BSA (1 mg/mL) (Sigma-Aldrich Inc., Missouri, USA) solution in PB (Figure 2 (e)). The excess BSA was removed, and the devices were washed with PBT followed by PB.
2. Single conducting polymer nanowire based immunosensors
Application of nanowire as chemiresistive immunosensors for sensing bacteriophages (as a model virus) and spores.
2.1. Nanoimmunosensor for bacteriophages: Bactriophages T7 and MS2 were used as targets. Ppy nanowire bridging the gap between the gold electrodes was functionalized with ant-T7 or anti-MS2 antibodies according to the protocol described above.
A single Ppy nanowire based sensor works on the principle of gating effect wherein, the semiconducting nanowire connected between the two contact electrodes is extremely sensitive to its surface environment and shows changes in its electrical properties to even slight perturbations on its surface such as changes in ions/pH or adsorption of charged molecules on the surface. The binding of the negatively charged moieties such as antibodies and phage particles on the Ppy, a well-known p-type semiconductor, nanowire surface decreases the number of charge carriers (holes), thus increasing the nanowire resistance. Figures 3A and 3B (trace (a)) show the I-V curves for bare Ppy nanowires. Functionalization of the Ppy nanowire with anti-T7 (Figure 3A, trace (b)) and anti-MS2 (Figure 3B, trace (b)) (and blocking step) increased the device resistance.
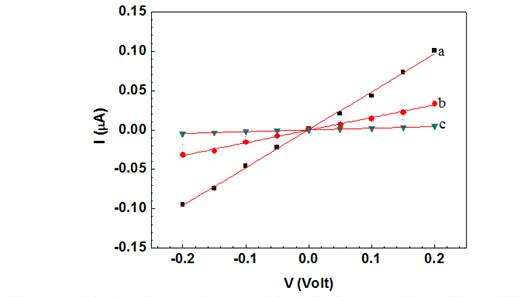
Figure 3A. Wet I-V characteristic curves of a a) bare Ppy nanowire b) after anti T7
immobilization and BAS blocking and c) after subsequent incubation with T7 phage
(109 PFU). Solid lines represent the linear fit.
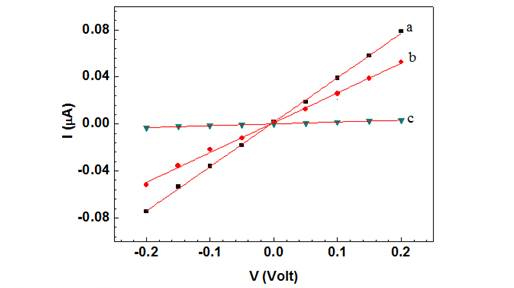
Figure 3B. Wet I-V chanracteristic curves of a a) bare Ppy nanowire b) after anti MS2
immobilization and BSA blocking and c) after subsequent incubation with MS2 phage
(109 PFU). Solid lines represent the linear fit.
This increase could arise due to chemical gating effect from the presence of negatively charged antibodies (and BSA) on p-type semiconducting Ppy nanowire surface The increase of nanowire resistance upon antibody immobilization (and blocking) indicated successful functionalization (and blocking). Subsequent incubation of these anti-T7 and anti-MS2 functionalized nanowire with the corresponding target bactriophage particles produced a dramatic increase in resistance (Figure 3A (c) and 3B (c)), illustrating the conductometric sensing.
Figures 4 and 5 show the anti-T7 and anti-MS2 functionalized Ppy nanowire sensor responses [(R - R0)/R0, where R is the resistance after exposure to T7 or MS2 phage and R0 is the initial sensor resistance], respectively, as a function of logarithmic T7 or MS2 phage concentration in 10 mM PB.
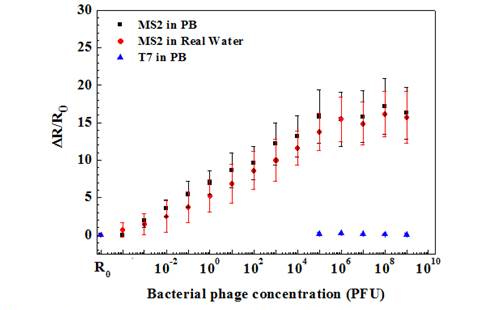
Figure 4. Sernsing response of anti-T7 immobilized Ppy nanowire towards various
concentrations of bacteriophages in PB and lake water samples.
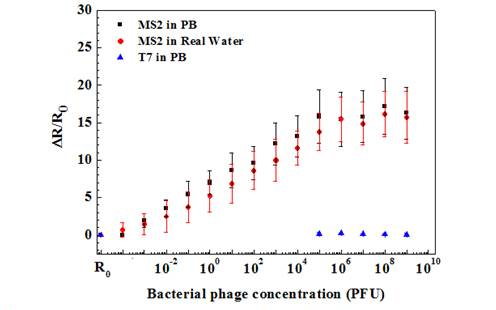
Figure 5. Sensing response of anit-T7 immobilized Ppy nanowire towards various
concentrations of bacterophages in PB and lake wate samples.
As shown, anti-T7 and anti-MS2 functionalized Ppy nanowire sensors were extremely sensitive with a response of 0.74 ± 0.05 and 1.96 ± 0.9 even at the lowest tested concentration of 10-3 PFU, respectively. Further, sensors had a wide dynamic range spanning from 10-3 to 106 PFU. A 10-3 value of detected PFU can be attributed to the probability that all the phage particles in the stock culture are not infective and are hence not accounted by the infectivity assay technique used for standardizing the phage stock and/or that there can be up to 103 viral particles/PFU. Also, the MS2 phage sensor showed almost 3-fold greater response compared to that of T7 phage sensor at the saturation, which could be attributed to the extent of individual antibody immobilization, which controls the binding capacity of the sensor or the difference in the surface charge density of the two phages or both. The LOD for our single Ppy nanowire immunosensor is comparable to that reported for single silicon nanowire-based immunosensor for influenza type A virus and paramyxovirus.
Specificity is a critical issue for successful application of a sensor. A very low response of 0.18 ± 0.03 for 108 PFU of T7 phage was recorded on nanowire sensor functionalized with monoclonal anti-MS2 antibody. On the other hand, nanowire functionalized with polyclonal anti-T7 showed a response of 0.55 ± 0.3 when exposed to 108 PFU MS2. This cross-reactivity was, however, very small compared to the affinity between the antibodies and the respective target phages. Negative controls, nanowires without antibodies but blocked with BSA, showed a response of only 0.02 ± 0.07 even for very high (108) PFU of both phages. The above results confirm the high selectivity of both phage immunosensors.
The utility of the developed sensor for detection of T7 and MS2 in untreated field samples was further evaluated by measuring different T7 and MS2 concentrations spiked in untreated lake water samples (Figures 4 and 5). As shown, the lower detection limit, dynamic range and sensitivity of the biosensor for T7 and MS2 in lake water sample were quantitatively comparable to those in PB. This indicates that the sensors are highly specific towards their respective target and also show no response towards the other interferants present in the field water sample.
2.2. Nanoimmunosensor for spores: Bacillus globigii was used as simulant of the threatening bioterrorism agent B. anthracis in this work. The Ppy nanowire was functionalized with antibody against the target spore using the zero-length linker as described above.
Figure 6 shows the calibration plot of the nanoimmunosensor for spores. The fabricated biosensor showed a lower detection limit of 1 CFU/mL (3 times the standard deviation of the response obtained for blanks/buffer) and a linear dynamic range between 1 and 100 CFU/mL (r2= 0.992) following the equation y = 5.49x - 0.065, with a response time of 30 min. The sensor response reached a plateau/saturation above 100 CFU/mL and no further increase in resistance was registered. Additionally, the biosensor responses were highly reproducible with small standard deviations.
The specificity of the biosensor also was assessed by measuring the response in the absence of anti-B. globigii antibodies (negative control) (Figure 6) and in the presence of non-target bacterial cells (E. coli) (positive control) (Figure 7) in solution. None of these controls showed significant levels of nonspecific interactions, even when very high concentrations of bacteria (106 CFU/mL) were used.
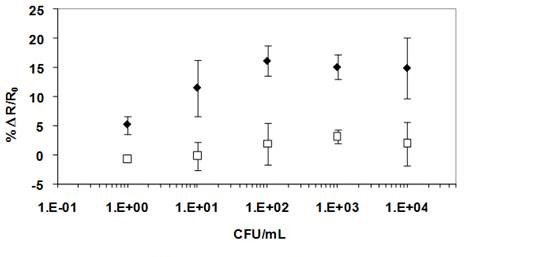
Figure 6. Calibration of plot ofr B. globigi spores biosensing at Ppy nanowires covalently
functionalized with anti-B. gloigii Abs (♦) versus Ppy nanowires with no Abs, blocked
with BSA (▢). The fidderences in response confirm the apore detection is acheived through
Ab-spore specific immuno-capture and not due to spore non-specific binding.
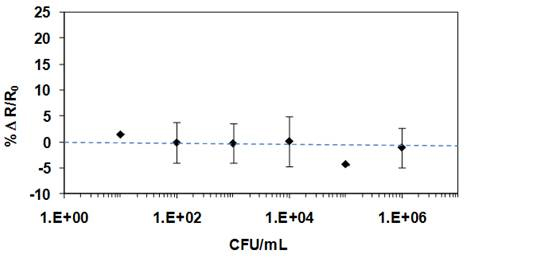
Figure 7. Negative control experiment performed at Ppy nanowires covalently
functionalised with anti-B-glogigii Abs in the presence of E. coli. The non-target
bacterial cells generated undetectable levels of non-specific adsorption into
the sensing surface up to titters of 106 CFU/mL.
Human Resources Development: Funding from this project was used to train the graduate students Ms. Sandra Harnandez, Mangesh A. Bangar, Carlos Hangarter, Ms. Miso Park and Ms. Lakshmi Cella and Postdoctoral Fellows Dr. C.L. Aravinda, Dr. Cristina Garcia-Aljaro, Dr. Debansu Chaudhuri, Dr. D. J. Shirale and Dr. Adam Wanekaya in the interdisciplinary areas of nanobiosensors and nanobiotechnology.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 9 publications | 9 publications in selected types | All 7 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Shirale D, Bangar M, Chen W, Myung N, Mulchandani A. Effect of Aspect Ratio (Length:Diameter) on a Single Polypyrrole Nanowire FET Device. THE JOURNAL OF PHYSICAL CHEMISTRY C 2010;114(31):13375-13380 |
GR832375 (Final) |
Exit |
Supplemental Keywords:
nanosensors, viruses, bacteria, monitoring, water, nanotechnology, biosensors, nanobiosensors, nanobiotechnology, Sustainable Industry/Business, RFA, Ecosystem Protection/Environmental Exposure & Risk, Scientific Discipline, Water, POLLUTANTS/TOXICS, Chemical Engineering, Environmental Chemistry, Engineering, Chemistry, & Physics, Monitoring/Modeling, New/Innovative technologies, Environmental Engineering, Environmental Monitoring, Water Pollutants, homeland security, electrochemical polymerization, environmental measurement, carbon nanotubes, nanowires, nanotechnology, nanosensors, nanoengineering, microbial pathogens, analytical chemistry, continous monitoring, continuous monitoring, nanocontact sensor, bioterrorism, immunosensor arraysProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
