Grantee Research Project Results
Final Report: Bioavailability of Haloacetates in Human Subjects
EPA Grant Number: R828044Title: Bioavailability of Haloacetates in Human Subjects
Investigators: Schultz, Irvin R. , Shangraw, Robert
Institution: Pacific Northwest National Laboratory , Oregon Health & Sciences University
EPA Project Officer: Hahn, Intaek
Project Period: September 30, 2000 through September 29, 2003 (Extended to April 1, 2005)
Project Amount: $524,928
RFA: Drinking Water (1999) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
This project characterized the absorption, disposition, and oral bioavailability of dichloroacetic acid (DCAA) in human volunteers after consumption of drinking water containing a known amount of DCAA. In controlled dosing experiments, we directly tested the hypothesis that prolonged exposure to low concentrations of dihaloacetic acids reduces their elimination and increases their systemic bioavailability in humans. DCAA was used as a model dihaloacetic acid in human volunteer studies because of its safe history as a therapeutic agent. A parallel experiment was performed using mixtures of chlorinated and brominated trihaloacetic acids in rhesus monkeys.
Overall, the project consists of three Specific Aims. In Specific Aim 1, volunteers (or monkeys) consumed only purified bottled water for 2 weeks. Volunteers then were given an oral dose of 12C-DCAA (2 mg/kg subject weight) in 1 pint of water. Monkeys were dosed with a mixture of 12C/13C trihaloacetic acids. After 5 minutes, 13C-labeled DCAA was administered by intravenous (i.v.) injection (via a catheter placed in the arm) and 12C/13C-DCAA measured in plasma over a 12-hour time period. In Specific Aim 2, Aim 1 was repeated in the same volunteers after a 2-week period of consuming a daily 0.02 mg/kg dose of DCAA. Specific Aim 3 studied the absorption of haloacetic acids occurring naturally in a finished municipal drinking water supply.
Approach:
Dichloroacetate (2 mg-haloacetate /Kg) will be given to volunteers within a pint of water. After 5 minutes, 13C-labeled dichloroacetate will be given by intravenous injection (via a catheter placed in the arm). A similar experiment will be performed using mixtures of chlorinated and brominated haloacetates in rhesus monkeys. In a second experiment, volunteers will consume a pint of tap water previously verified to contain the seven haloacetates of interest. For all experiments, serial blood samples will be removed using the intravenous catheter and the blood plasma analyzed simultaneously for both the 13C- and 12C haloacetates (using GC-MS or LC-MS/MS techniques). The area-under-the-curve ratio for the oral and intravenous doses will be determined to estimate the oral bioavailability.
Summary/Accomplishments (Outputs/Outcomes):
Human Volunteer Study-Aim 1
A total of 18 volunteers were recruited for the project. Two volunteers, subjects 4 and 14, withdrew from the study before completion and are not included in the analysis. Volunteers were provided with purified bottled water (Costco brand) and asked to cook and drink using only this water for 14 consecutive days prior to the DCAA study. On the day of the experiment, each volunteer arrived at the Oregon Health Sciences University (OHSU) Clinical Research Center (CRC) and was asked to consume 0.5 L of bottled water spiked with DCAA at a corresponding dose of 2 mg/kg subject weight. After 5 minutes, the volunteer was administered a 0.3 mg/kg 13C1-DCAA i.v. dose via a catheter placed in the arm). Blood was sampled repetitively via a second catheter not used for 13C1-DCAA injection, and the plasma collected and stored at -80°C until analysis. The blood sampling schedule was based on the results from the initial three volunteers (subjects 1,2,3). Initially, sampling occurred at 0, 10, 20, and 40 minutes, and 1, 1.5, 2, 4, 8, and 12 hours after oral dosing. Analysis of the plasma levels of DCAA in these volunteers indicated absorption and elimination were much more rapid than expected, with plasma concentrations in all subjects dropping below the DCAA method detection limit of 1 ng/mL within 2 hours after dosing. Therefore, the sampling schedule was modified to increase sampling frequency during the initial 1.5 hours after dosing. The final sampling schedule used in all subsequent studies was 0, 5, 10, 15, 20, 25, 30, 45, 60, 75, 90 120, and 180 minutes. Analysis of 12C-DCAA and 13C-DCAA in the plasma samples was by gas chromatography-mass spectrometry (GC-MS) operated in selected ion monitoring mode, with the mass-to-charge ratio (m/z) = 59 (12C-DCAA) and m/z = 60 (13C-DCAA) ions used for quantification. Fluorochloro acetic acid (FCAA) was used as the internal standard. Peak identification was confirmed from matching retention times of known analytical standards and presence of secondary ions (m/z = 83 for DCAA and m/z = 67 for FCAA). Prior to GC-MS analysis, DCAA and FCAA was methylated using diazomethane.
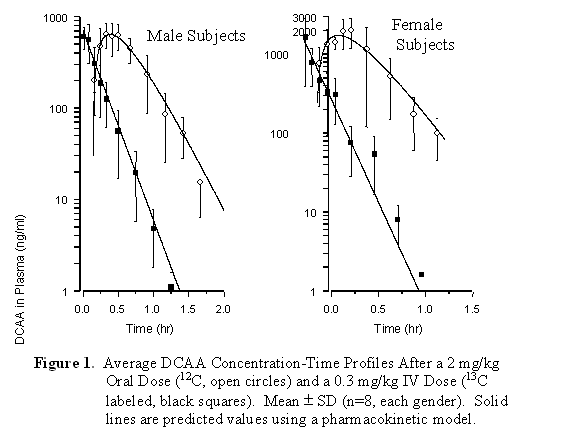
The plasma concentration-time profiles for the oral and i.v. doses in each subject were analyzed using a clearance-volume compartmental pharmacokinetic model and non-compartmental analysis. The latter was used to obtain estimates of the area-under-the-curve (AUC0→µ), apparent volume of distribution (Vss), and total body clearance (Clb). The pharmacokinetic model was used to estimate the oral absorption rate (Ka) and peak plasma levels (Cmax) after the simultaneous nonlinear least squares fitting of the observed oral and i.v. data to model-based differential equations. The pharmacokinetic model included bioavailability as a fitted parameter which permitted direct estimation of oral bioavailability in addition to its determination based on AUC ratios. A summary of the mean (± SD) plasma concentration-time profiles for the male and female volunteers is shown in Figure 1. Table 1 summarizes the findings from the pharmacokinetic analysis. In general, the shape of the oral and i.v. DCAA profiles was qualitatively similar for all subjects. Peak DCAA levels were achieved rapidly after oral dosing, typically occurring between 15 and 30 minutes after consumption of the dose. Model-predicted peak plasma levels varied substantially between subjects, ranging between 600-3,423 ng/mL (Table 1). Total body clearance estimated from the i.v. dose also varied to a similar extent, ranging from 716-4,300 (mL/h/kg). In contrast, the Vss and Ka were less variable among the volunteers (Table 1). In all subjects, plasma levels of DCAA declined below method detection limits by 2 hours. With regard to estimation of oral bioavailability, there was generally close agreement between estimates made by both analysis methods (Table 1). Based on these results, the oral bioavailability of DCAA in the volunteers varied widely, ranging from 28-100 percent.
With regard to gender differences, statistical analysis of the pharmacokinetic data was performed using the Mann-Whitney test, which is a non-parametric statistic. This analysis indicated there were no significant differences (p > 0.05) in all parameters between male and females volunteers. Although not statistically significant, there appeared to be a trend in the results suggesting that in women, DCAA was absorbed more rapidly (increased Ka) while the capacity for excretion was less (decreased Clb) compared to men (Table 1). Correspondingly, the observed and model predicted peak levels of DCAA were higher in women (Table 1 and Figure 1) along with a small increase in bioavailability in women.
Human Volunteer Study-Aim 2
For this part of the study, the same volunteers who participated in Aim 1 were asked to participate again. In this experiment, volunteers consumed 0.02 mg/kg DCAA daily for 14 consecutive days. The DCAA was spiked into 0.5 L of purified water, which the volunteers were asked to consume each morning. They were asked to use purified bottled water as before for cooking and drinking. This was intended to control the exposure of DCAA (and other haloacetic acids) and approximate consumption of drinking water containing 1 mg/L DCAA. On day 15, each volunteer arrived at the OHSU-CRC and participated in the DCAA oral and i.v. dosing experiments as described in Aim 1. Blood sampling and DCAA analysis along with the pharmacokinetic analysis was as described in Aim 1. A summary of the pharmacokinetic analysis is provided in Table 2. The same general description of DCAA pharmacokinetics observed in Aim 1 also applies to the results observed after the 14-day DCAA exposure. As observed before, there were no statistical differences in DCAA pharmacokinetics between men and women. Analysis of the pharmacokinetic data before and after the prolonged DCAA exposure using the Wilcoxon signed rank test, however, indicated women, but not men, were affected by the treatment. The AUC0→µ for both oral and i.v. DCAA doses was increased significantly (p<0.04, 0.014, respectively) after the 14-day DCAA exposure (Table 1 and Table 2). Consistent with the increased AUC, the Clb was decreased significantly (p<0.014) in women
Table 1. Summary of Pharmacokinetic Parameters After Simultaneous Administration of a 2 mg/kg Dose of DCAA in Drinking Water and 0.3 mg/kg Intravenous Dose of 13C-labeled DCAA
|
|
Intravenous Dosing |
|
Oral Dosing |
Bioavailability* |
||||||
|
Subject |
AUC0→µ |
Vss |
Clb |
t½, β |
|
AUC0→µ |
Cmax |
Ka |
PKmodel |
Ncomp |
|
No. |
(ng/mL/h) |
(mL kg-1) |
(mL/h/kg) |
(h) |
|
(ng/mL/h) |
(ng/mL) |
(hr-1) |
% |
% |
|
Male Volunteers |
|
|
|
|
|
|
|
|
||
|
1 |
221 |
339 |
1,360 |
0.21 |
|
986.0 |
n.d. |
n.d. |
n.d. |
67 |
|
5 |
85 |
651 |
3,527 |
0.13 |
|
577.0 |
768 |
3.17 |
98 |
100 |
|
7 |
154 |
541 |
1,949 |
0.21 |
|
490.6 |
606 |
2.34 |
64 |
48 |
|
9 |
165 |
354 |
1,822 |
0.14 |
|
402.2 |
618 |
4.27 |
34 |
37 |
|
12 |
122 |
446 |
2,458 |
0.13 |
|
302.3 |
600 |
7.10 |
35 |
37 |
|
13 |
374 |
164 |
802 |
0.12 |
|
1,084.1 |
2,432 |
6.74 |
50 |
43 |
|
16 |
258 |
267 |
1,164 |
0.12 |
|
489.3 |
978 |
10.4 |
27 |
28 |
|
18 |
316 |
227 |
950 |
0.16 |
|
1,710.3 |
3,338 |
5.93 |
86 |
81 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Average |
212 |
374 |
1,754 |
0.15 |
|
755 |
1,334 |
5.71 |
56 |
54 |
|
SD |
99 |
164 |
905 |
0.04 |
|
474 |
1,099 |
2.74 |
28 |
25 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Female Volunteers |
|
|
|
|
|
|
|
|
||
|
2 |
220 |
235 |
1,365 |
0.19 |
|
1315 |
n.d |
n.d. |
n.d |
90 |
|
3 |
205 |
171 |
1,461 |
0.13 |
|
766 |
n.d |
n.d. |
n.d |
56 |
|
6 |
153 |
1010 |
1,948 |
0.27 |
|
742 |
3,423 |
6.31 |
100 |
73 |
|
8 |
278 |
268 |
1,079 |
0.16 |
|
853 |
1,316 |
3.33 |
51 |
46 |
|
10 |
70 |
745 |
4,300 |
0.12 |
|
225 |
1,230 |
4.63 |
54 |
48 |
|
11 |
419 |
164 |
716 |
0.16 |
|
1558 |
759 |
3.02 |
60 |
56 |
|
15 |
318 |
270 |
942 |
0.16 |
|
1280 |
2,180 |
5.24 |
67 |
60 |
|
17 |
280 |
151 |
1,070 |
0.10 |
|
739 |
2,323 |
14.1 |
38 |
40 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Average |
243 |
377 |
1,610 |
0.16 |
|
935 |
1,872 |
6.11 |
62 |
59 |
|
SD |
107 |
320 |
1,150 |
0.05 |
|
425 |
966 |
4.10 |
21 |
16 |
|
|
|
|
|
|
|
|
|
|
|
|
Note: The AUC0→µ, Vss, Clb and t½, β were calculated using non-compartmental methods. The Cmax and Ka were determined from a simultaneous nonlinear least squares fit of the oral and i.v. data using the pharmacokinetic model and the WinNonlin program.
*Oral bioavailability was calculated from the ratio of the AUC0→µ values for the oral and i.v. doses (with adjustment for the differences in dose) and as a fitted parameter from the in the pharmacokinetic model.
n.d. = not determined.
after the DCAA exposure. Although the clearance was reduced in women, the oral bioavailability for both men and women was not significantly different after the 14-day DCAA exposure.
Rhesus Monkey Study
In these experiments, rhesus monkeys were used as surrogates for human volunteers to study the pharmacokinetics of chlorinated and brominated trihaloacetates: trichloracetic acid (TCAA), bromodichloroacetic acid (BDCAA), chlorodibromoacetic acid (CDBAA) and tribromoacetic acid (TBAA). The experimental approach was identical to the human volunteer studies: female
Table 2. Summary of Pharmacokinetic Parameters After Simultaneous Administration of Oral and Intravenous Doses of DCAA in Volunteers After 14-Day Consumption of 20 μg/kg DCAA in Their Drinking Water
|
|
Intravenous Dosing |
|
Oral Dosing |
Bioavailability* |
||||||||||||||||||||||||||
|
Subject |
AUC0→µ |
Vss |
Clb |
t½, β |
|
AUC0→µ |
Cmax |
Ka |
PKmodel |
Ncomp |
||||||||||||||||||||
|
number |
(ng/mL/h) |
(mL kg-1) |
(mL/h/kg) |
(h) |
|
(ng/mL/h) |
(ng/mL) |
(hr-1) |
% |
% |
||||||||||||||||||||
|
Male Volunteers |
|
|
|
|
|
|
|
|
||||||||||||||||||||||
|
1 |
428 |
145 |
701 |
0.16 |
|
1,616 |
n.d |
n.d. |
n.d. |
57 |
||||||||||||||||||||
|
5 |
202 |
395 |
1,487 |
0.18 |
|
569 |
904 |
3.02 |
51 |
42 |
||||||||||||||||||||
|
7 |
116 |
945 |
2,582 |
0.15 |
|
363 |
579 |
4.81 |
51 |
47 |
||||||||||||||||||||
|
9 |
490 |
153 |
611 |
0.17 |
|
1,800 |
3,539 |
7.97 |
56 |
55 |
||||||||||||||||||||
|
12 |
276 |
175 |
1,087 |
0.1 |
|
792 |
1,398 |
3.54 |
46 |
43 |
||||||||||||||||||||
|
13 |
212 |
443 |
1,413 |
0.18 |
|
1,188 |
2,246 |
5.85 |
98 |
84 |
||||||||||||||||||||
|
16 |
161 |
526 |
1,868 |
0.18 |
|
555 |
938 |
7.94 |
55 |
52 |
||||||||||||||||||||
|
18 |
366 |
239 |
820 |
0.19 |
|
2,104 |
3,245 |
3.72 |
99 |
86 |
||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||
|
Average |
281 |
377 |
1,321 |
0.16 |
|
1,123 |
1,835 |
5.26 |
65 |
58 |
||||||||||||||||||||
|
SD |
133 |
271 |
668 |
0.03 |
|
653 |
1,190 |
2.06 |
23 |
17 |
||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||||||||
|
Female Volunteers |
|
|
|
|
|
|
|
Expected Results:This project will provide critical data needed to make accurate and reliable exposure estimates of haloacetates to humans consuming municipal drinking water supplies. This project will identify the consequences of low level exposure to haloacetates on their subsequent metabolism and disposition. This information is needed to assess whether individuals who consume water containing high levels of by-products experience greater than predicted exposure due to decreased elimination of haloacetates. This project will also allow for the direct testing of physiologically based pharmacokinetic model predictions of haloacetate dosimetry in humans. The increased understanding of haloacetate pharmacokinetics obtained in this study will allow for improvements in PBPK modeling accuracy, thus providing credible, scientifically defensible measures of effective dose estimates of haloacetates to humans. Journal Articles on this Report : 3 Displayed | Download in RIS Format
Supplemental Keywords:drinking water, bioavailability, mixtures, halogenated acetic acids, toxicokinetics, mass spectroscopy, stable isotopes,, Health, RFA, Ecosystem Protection/Environmental Exposure & Risk, Scientific Discipline, PHYSICAL ASPECTS, Waste, Water, Toxics, National Recommended Water Quality, Health Risk Assessment, Physical Processes, Fate & Transport, Risk Assessments, Biology, Monitoring/Modeling, Bioavailability, Disease & Cumulative Effects, Toxicology, Drinking Water, Environmental Monitoring, metabolism, DBPs, PBPK modeling, health effects, stable isotope, cellular physiology, treatment, dose response, water quality, haloacetates, risk assessment, human exposure, DBPs , PBPK, dosimetry, brominated DPBs, pharmacokinetics, monitoring, physiologically based pharmacokinetic model, drinking water contaminants, exposure, dose estimates, absorption, elimination, microbial exposure, human health risk, disinfection byproducts (DPBs), dose-response, chlorinated DBPs, DBP exposure, renal eliminatio, dichloroacetate, exposure and effectsProgress and Final Reports:Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency. Project Research ResultsSite NavigationRelated InformationContact Us to ask a question, provide feedback, or report a problem.
Last updated April 28, 2023
| ||||||||||||||||||||||