Grantee Research Project Results
Final Report: Assessment of Human Dietary Ingestion Exposures to Water Disinfection Byproducts via Food
EPA Grant Number: R826836Title: Assessment of Human Dietary Ingestion Exposures to Water Disinfection Byproducts via Food
Investigators: Raymer, James H. , Hu, Ye A. , Pellizzari, Edo D. , Akland, Gerald G. , Michael, Lisa J. , Weinberg, Howard S. , Marrero, Thomas , Unnam, Vasu
Institution: Desert Research Institute
EPA Project Officer: Hahn, Intaek
Project Period: October 1, 1998 through September 30, 2003
Project Amount: $446,468
RFA: Drinking Water (1998) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
The overall objective of this research project is to estimate the magnitude of exposure to disinfection by-products (DBPs) in drinking water provided by municipal water treatment facilities via ingestion after uptake into food during cooking. It has been shown in our laboratory that foods can become contaminated with chemicals originating in water used in the home during food preparation (e.g., cooking). The magnitude of this contamination process has not been studied. This research specifically will address the uptake of compounds known to arise from the process of water disinfection (ozonation in conjunction with a secondary process such as chloramination and chlorination), including nonhalogenated aldehydes, ketones and acids, trihalomethanes, haloacetic acids (HAAs), bromate, chloropicrin, and haloacetonitriles. The main hypotheses to be tested are: (1) foods prepared using contaminated water become contaminated; (2) food is a significant source of DBP exposure; (3) DBP concentrations in food can be predicted with knowledge of DBP concentrations in drinking water and foods consumed; and (4) dietary exposures of children are higher than for an adult living in the same household.
Approach:
Analytical methods will be developed for ozonation DBPs in foods and beverages. Controlled, laboratory experiments will be conducted to determine how selected DBPs, especially those produced during ozonation, are adsorbed by food during the cooking process. Other DBP methods for foods and beverages for halogenated compounds (currently under development) will be brought in as needed. The relation between DBP concentrations in water and in foods following cooking in contaminated water will be modeled. This research will specifically address food items commonly eaten by children, in addition to food items eaten by adults, so that estimates of ingestion of the study compounds to this subgroup can be determined. A field study will be conducted in two cities each having different factors in water disinfection, e.g., secondary disinfection, bromide concentration, high dissolved organic carbon, in order to test the validity of the model for predicting potential exposures and to estimate the human exposures to DBPs from food and water.Summary/Accomplishments (Outputs/Outcomes):
Nonhalogenated DBPs
An analytical method for the determination of nonhalogenated water DBPs in foods and beverages was investigated. As a starting point, Standard Method 6252 (DBPs: Aldehydes) for aldehydes in drinking water was modified. The method, which relied on the derivatization of carbonyl compounds with o-(2,3,4,5,6-pentafluorobenzyl)-hydroxylamine to form the corresponding oxime with subsequent determination using gas chromatography with electron capture detection, was optimized with regard to reduced reagent volumes and derivatization time and temperature. The modified method was applied to the determination of formaldehyde, acetaldehyde, propanal, butanal, pentanal, hexanal, heptanal, octanal, benzaldehyde, nonanal, glyoxal, and methyl glyoxal in homogenized medium-fat food composite (1 ppm each) and composite beverage (200 ppb each). The results suggested that nonhalogenated aldehydes, that arise from disinfection via ozonation, can be derivatized and extracted, but that some of the analytes are retained by the matrix or cannot be detected because of substantial interferences. A cleanup step consisted of the extraction of the hexane extract containing the derivatized carbonyl compounds with aqueous bicarbonate to remove potentially interfering acidic compounds; this resulted in minimal improvement. Mass spectrometric analysis showed that the derivatives were present in water and could be determined.
The developed method was applied to foods and beverages. The sample extracts were very complex and contained many of the target analytes without the addition of any known DBPs. Given that the spike levels for the nonhalogenated compounds in food and water were 1 ppm and 200 ppb, respectively, small increases for most of the analytes were measured for the spiked samples relative to the unspiked samples. The additional contributions to exposure as a result of uptake from drinking water during cooking that contained lower concentrations of the nonhalogenated DBPs were concluded to be minimal. No additional work was done on the nonhalogenated DBPs.
Haloacetonitriles and Other DBPs
It was demonstrated that haloacetonitriles (trichloroacetonitrile, dichloroacetonitrile, and bromochloroacetonitrile) could be recovered from a variety of cooked foods when spiked to contain 30 ppb of each analyte (Raymer, et al., 2000a). In addition, 1,2-dichloro-2-propanone and 1,1,1-trichloro-2-propanone could be recovered at the same concentration. Chloropicrin could not be recovered from foods. Excess compounds taken into foods during home processing is possible. Recoveries from beverages, spiked to contain 6 ppb, generally were acceptable for dichloroacetonitrile, 1,1-dichloro-2-propanone, and 1,1,1-trichloro-2-propanone. It is not known whether the availability of these compounds when consumed in beverages made from drinking water is different than when consumed in drinking water. An estimate of exposures based on all drinking water used to prepare beverages or drinking water consumed directly would provide an upper bound. Actual exposures could be less if the analytes are not bioavailable from foods and beverages.
Haloacetonitriles were lost very rapidly from boiling water. In fact, spiked water brought to a boil and then cooled and analyzed showed no detectable residues. Similarly, the haloacetonitriles added to canned soup could be recovered before heating, but were not detectable after the soup had been heated to a boil and then cooled. It is not clear whether the haloacetonitriles were lost to volatilization or were decomposed during the heating process. Exposures to haloacetonitriles via foods prepared with drinking water that are boiled during preparation likely are to be minimal. However, exposures to possible degradation products of haloacetonitriles were not investigated.
HAAs
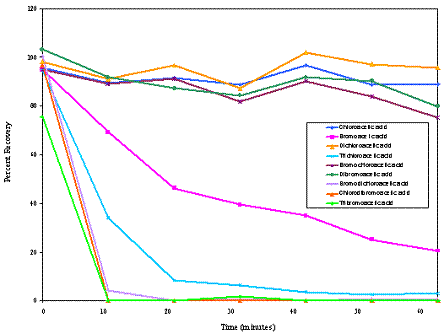
The stability of HAAs (chloroacetic, bromoacetic, dichloroacetic, trichloroacetic, bromochloroacetic, dibromoacetic, bromodichloroacetic, chlorodibromoacetic, and tribromoacetic acids) in an aqueous solution brought to a boil showed that bromodichloroacetic, chlorodibromoacetic, and tribromoacetic acids were not recovered, but the other analytes remained and could contribute to human exposure via ingestion. Recovery as a function of time of boiling in spiked reagent water (initial concentrations ranged from 50-500 ppb each) is shown in Figure 1. Thus, it is clear that food cooked for long periods of time can reduce the excess exposures associated with the uptake of certain HAAs during cooking.
Figure 1. Recovery of HAAs From Water as a Function of Boiling Time
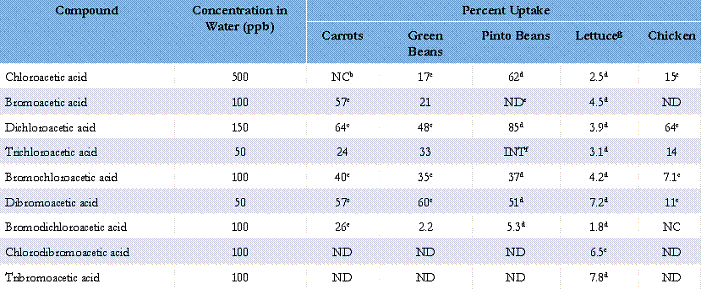
The partition of HAA DBPs into carrots, green beans, pinto beans, chicken, spaghetti, and lettuce following cooking or contact with water was studied. We evaluated the HAAs listed above. Foods were chosen based on different cooking conditions, different chemical compositions (i.e., starch, vegetable, protein), and those commonly consumed by children. Spaghetti and dried beans are prepared with large volumes of water. In the case of spaghetti, multiple contacts with drinking water are possible if the spaghetti is rinsed following cooking. Dried beans usually are soaked overnight, drained, and then cooked. Lettuce was selected because it is commonly rinsed with water during preparation. All foods were prepared according to package directions using reagent water spiked with known concentrations of HAAs. Foods processed in the same manner using unspiked reagent water served as controls. Cooked spaghetti was subjected to rinsing with both reagent and spiked water to determine if rinsing could cause additional HAA uptake. Beans were cooked in spiked water following soaking in either spiked or reagent water. Chicken was cooked in spiked water following a brief soaking in either reagent or spiked water. Lettuce was soaked briefly (~ 5 minutes) then drained prior to homogenization. Foods were homogenized and extracted after preparation. The methyl esters of the HAAs were formed and analyzed by gas chromatography in conjunction with electron capture detection. The results are shown in Table 1.
In some cases, more than 60 percent of the HAAs in the cooking water were taken up by the food during cooking. The HAAs showing the greatest extent of uptake were bromoacetic, dichloroacetic, bromochloroacetic, and dibromoacetic acids. Initial rinsing of the chicken and soaking of the pinto beans in spiked water did not result in any significant increases in HAA concentration in the foods cooked in spiked water compared to the case where the initial rinsing or soaking was performed with reagent water. In general, chlorodibromoacetic and tribromoacetic acids are not detected in the food following cooking. This is consistent with the rapid loss of the these compounds during boiling as shown in earlier studies. The soaking of lettuce also resulted in uptake of the HAAs (1.8 to 7.8 percent of the total available).
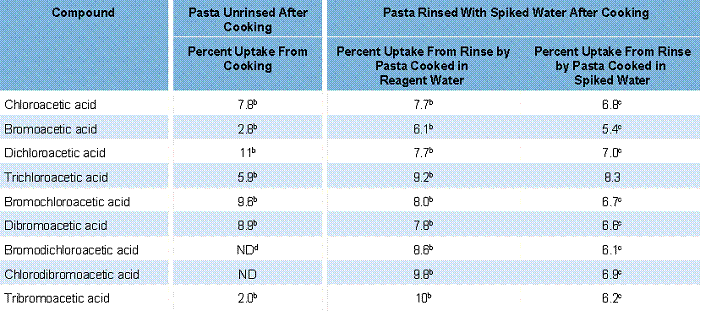
The results for spaghetti indicated that as much as 11 percent of the available HAAs were adsorbed/absorbed during cooking in spiked water (see Table 2). Given the large volumes of water used to cook pasta, the masses absorbed could be much higher than for other types of food. In addition, the data on uptake following rinsing with spiked water indicate that additional HAAs, up to 10 percent, are taken up from the rinsing water. This was true whether the spaghetti was cooked in spiked reagent water or in spiked drinking water. It also is clear that the HAAs that typically show low recovery following cooking (bromodichloroacetic acid, chlorodibromacetic acid, and tribromoacetic acid) become available to contribute to exposure following contact of the spaghetti with fresh DBP-containing water.
To develop a model to predict uptake of the HAA DBPs during cooking, experiments were conducted in which carrots and spaghetti were cooked for different times to characterize accumulation of the DBPs into food. A model was developed for carrots that showed uptake and losses that resulted from increasing uptake as the food structure broke down as well as loss from the overcooked food and decomposition of HAAs. Data for the spaghetti showed a great deal of variability and could not be fit.
Bromate in Foods
Bromate is a component of drinking water that is formed following ozonation of water that contains bromide. This aspect of the study was undertaken to determine whether bromate in drinking water used for cooking would partition into and concentrate in cooked food. Bromate in drinking water currently is regulated in the United States at a maximum contaminant level of 10 μg/L based on drinking water consumption. If bromate in drinking water is concentrated in foods during cooking, then an additional path of exposure occurs, leading to an underestimation of human exposure to bromate. A method that utilized an accelerated solvent extractor was developed to extract bromate from cooked foods. An ion chromatographic method was modified to measure bromate (as formed tribromide) in food extracts and cooking waters. These methods then were applied to different types of spaghetti and carrots that were cooked in various water matrices, including both synthetic and chlorinated drinking water matrices containing elevated bromate levels. Although experimental results illustrated no net bromate uptake in the foods examined during the cooking process, a relatively high level of bromate was found in the cooked products originating from the raw materials.
Table 1. Percent Uptake of HAAs Into Foods During Cookinga
a Percent uptake of HAAs from available cooking water; values corrected for foods cooked in reagent water.
b NC = Not Calculated. A decrease in concentration was measured following cooking in spiked water.
c Significant at P < 0.05.
d P value not calculated since n = 1 for control case; a substantial increase was noted for these compounds relative to the food cooked in reagent water.
e ND = Not Detected.
f INT = Interferent. Very high trichloroacetic acid concentration in the blank made estimation of the uptake difficult.
g Lettuce soaked for 5 minutes in water containing HAAs.
Table 2. Uptake of HAAs Following Cooking in Spiked Water and the Impact of Rinsinga
aUptake calculations corrected for background (i.e., concentrations of HAAs measured following cooking in reagent water). N = 3 in each case.
bP value not calculated since n = 1 for control case; a substantial increase was noted for these compounds relative to the food cooked in reagent water.
cSignificant uptake at P < 0.05.
dND = Not Detected.
Field Study for HAAs
Samples of food and beverages (using the duplicate diet approach) were collected from adults and children between 2 and 8 years of age in homes that utilized disinfected drinking water. A criterion for inclusion was that foods prepared by boiling would be used during the study. A total of 10 homes were selected from residents served by a water treatment plan that utilized ozonation/chloramination, and a total of 10 homes were selected that were served by a utility that used chlorination. Twenty adults and 15 children participated in the study. Food and beverages were collected during 2 days. Water both before and after cooking (recovered water) were collected in addition to food before (raw) and after cooking. Participants recorded the food, the cooking duration, the amounts of food and water used, and the amount of food consumed by participating adults and children. Both sentinel (foods boiled in water) and nonsentinel foods (all other foods) were collected. Because of resource limitations, only the water samples (before and after cooking) were analyzed. Uptakes of HAAs into foods were estimated by the masses of HAAs in water before and after cooking. HAAs were the focus, given that the other DBPs did not persist during boiling.
The percentage uptakes were calculated, including only the measurable values calculated according to [(mass in water before cooking - mass in water after cooking) x 100] ÷ (mass in water before cooking). In each case, the calibration check standards verified in control instrument operation. Calculated uptakes varied by HAA and food type. Even within food types, uptakes ranged from negative values, suggesting that the food released HAA into the cooking water, to greater than 80 percent, even after taking into account those HAAs that decompose with cooking. In those cases, very large positive uptakes would be calculated, and those values would be misleading.
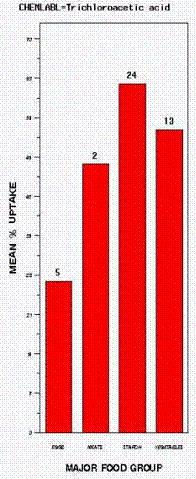
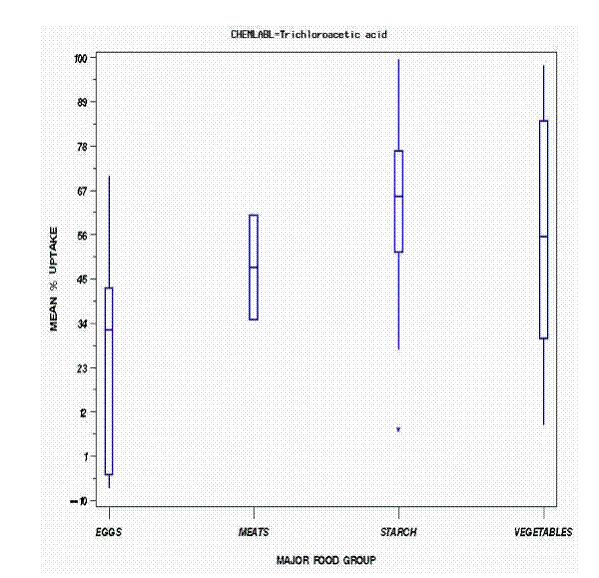
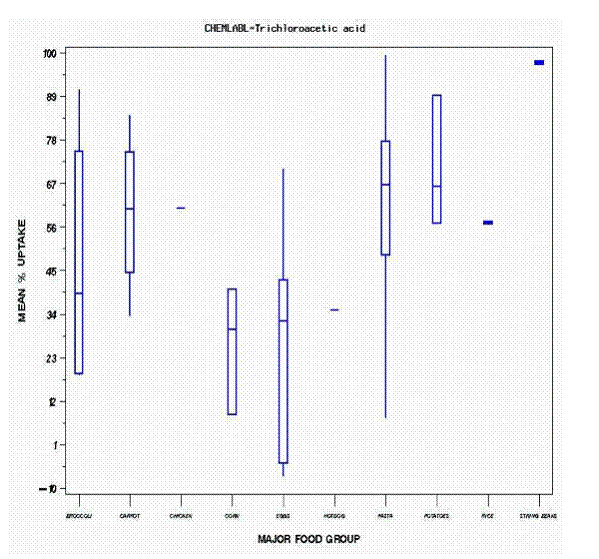
The most commonly measured HAA was trichloroacetic acid. The mean percentage uptake by major food group is shown in Figure 2. The numbers on top of each bar represent the total number of samples in that category. Starches, including pasta of various sorts and potatoes, gave rise to the greatest uptake. Rice was not included because no water remained after cooking. It is interesting to note the uptake by eggs boiled in the shell for varying times. It is likely that the trichloroacetic acid was bound to the shell and did not actually penetrate the portion of the egg consumed; however, because the edible portion of the egg was not extracted and analyzed, this cannot be stated definitively. The same data are shown in Figure 3 where the box defines the first quartile (bottom of box), the third quartile (top of box), the mean (line across box), the minimum value (bottom of line), and the maximum (top of line). Finally, Figure 4 shows the different foods that comprise the major food groups. All of the water values suggest positive uptake into foods. It is important to note that the mean uptake of trichloroacetic acid by carrots was approximately 60 percent, compared to 24 percent in the controlled cooking data of Table 1. Similarly, pasta showed an approximate uptake of 65 percent as reflected by the water measurements, compared to 14-15 percent (with rinsing) for food as shown in Table 2. This discrepancy could have arisen from our inability to measure the trichloroacetic acid directly in food within the available funding.
By analyzing only the water, we are assuming a complete mass balance and only two compartments, the water and the food. Our more detailed work with the carrots for model development suggests that a complete mass balance is not probable. Thus, the approach taken here likely overestimates the uptake into food. Nevertheless, uptake into food has been demonstrated and contributes to human excess dietary exposure.
Additional analyses of the data are planned to take into account the different sources of drinking water and the difference between total masses consumed by adults versus children.
Figure 2. Trichloroacetic Acid Uptake by Major Food Group
Figure 3. Box Chart Showing Uptake of Trichloroacetic Acid Into Major Food Groups
Figure 4. Box Chart Showing Uptake of Trichloroacetic Acid Into Specific Food Types
Expected Results:
The major benefit is that current risk assessments assume that ingestion consists of adding the concentration levels of the specific compounds known to exist in the drinking water (multiplied by the normal assumption of volume of drinking water consumed) to the levels of the specific compounds which exist in the food, as determined by FDA in various surveys. However, this research will attempt to verify that the estimates of exposure related to ingestion actually underestimate the actual exposures, and estimate the amount of underestimation for the two population groups of interest -- children and adults, based on actual measurements of in-home prepared foods and drinking water. Furthermore, this research will provide new information on dietary exposure to a specific set of compounds that is currently not available. The risk assessment process will be improved in that a greater understanding of DBP exposure via food will be obtained. Confirmation of the significance of this exposure pathway for DBPs will lead to more accurate risk assessments, and provide evidence for judging the extent to which children might be included in the portion of the population which has these higher total exposures.Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 8 publications | 1 publications in selected types | All 1 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Raymer JH, Pellizzari E, Childs B, Briggs K, Shoemaker JA. Analytical methods for water disinfection by-products in foods and beverages. Journal of Exposure Analysis and Environmental Epidemiology 2000a;10(6 Pt 2):808-815. |
R826836 (Final) |
|
Supplemental Keywords:
drinking water, water disinfection, nonhalogenated water disinfection, disinfection by-products, DBPs, ozonation, chloramination, chlorination, exposure, children, organics, measurement methods, haloacetic acids, HAAs,, RFA, Health, Scientific Discipline, Water, Toxics, National Recommended Water Quality, Susceptibility/Sensitive Population/Genetic Susceptibility, Health Risk Assessment, Risk Assessments, Chemistry, genetic susceptability, Children's Health, Disease & Cumulative Effects, Drinking Water, DBPs, dietary exposure, environmental hazard exposures, sensitive populations, aldehydes, ozonation, treatment, dissinfection biproducts, water quality, haloacetonitriles, trihalomethane, ketones, risk assessment, human exposure, DBP effects, brominated DPBs, chloramines, dietary ingestion exposures, haloacetic acids, trihalomethanes, food, diet, DBPs , THM, children, drinking water system, drinking water contaminants, exposure, cooking, analytical chemistry, bromate formation, chemical byproducts, disinfection byproducts, disinfection byproducts (DPBs), human health effects, DBP exposure, biomarkers, detection, disinfection biproductsRelevant Websites:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.