Grantee Research Project Results
2022 Progress Report: Integrated blood brain barrier computational model development to predict doses of concern for compound linked neurotoxicity
EPA Grant Number: R840027Title: Integrated blood brain barrier computational model development to predict doses of concern for compound linked neurotoxicity
Investigators: Knipp, Gregory , Sluka, James
Institution: Purdue University , Indiana University
EPA Project Officer: Chung, Serena
Project Period: August 1, 2020 through May 8, 2025
Project Period Covered by this Report: August 1, 2021 through July 31,2022
Project Amount: $790,441
RFA: Advancing Toxicokinetics for Efficient and Robust Chemical Evaluations (2019) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Investigate the bidirectional permeability of marker compounds to determine the permeation of 50 known or purported environmental neurotoxicants across a human cell-based model of the Blood-Brain Barrier. Permeability data will be integrated to rank order neurotoxicant exposure in reference to established BBB marker compounds. The development of physiological-based pharmacokinetic (PBPK) models based on the neurotoxicant permeation rates across the BBB cell-based assays will then be performed to improve accuracy in predicting doses of concern of chemicals that have been implicated in causing perturbations in brain function.
Progress Summary:
Objective 1: Adapt a presently available low throughput triculture model of the human BBB to a higher throughput screen that will measure bi-directional compound permeability.
In the first year, we validated the human layered blood brain barrier triculture (human astrocytes, pericytes, and the HBEC-5i human brain microvessel endothelial cell line) model using several marker compounds to ascertain that significant barrier properties and the ability to discriminate and rank order the permeation rates of marker compounds. The rank order of the permeability coefficients for these compounds matched their observed in vivo exposure. We then began conducting bidirectional permeability studies across the blood-brain barrier (BBB) triculture model with the selected 50 neurotoxicants. To date we have measured the permeability for almost 35 potential neurotoxicants. Representative apparent permeability coefficients (Papp) for several compounds are listed in Table 1 below. We have found that a few of these compounds need to be repeated due to the fact that sink conditions (>90% of the compound in the donor chamber) was not maintained at the first measurement time point. We are repeating these compounds with sampling being adjusted to every five minutes for 30 minutes to better determine the Papp values. It needs to be noted that we have tried to determine the permeation rates for representative highly lipophilic compounds including polyaromatic hydrocarbons (PAH), polybrominated diethyl ethers (PBDE), perfluorooctanoic acid (PFOA), perfluorooctane sulfonate (PFOS) and related compounds with little success. Several of these compounds have limited solubility, which we cannot raise. During our evaluation of the BBB model, we find that if we go beyond 1-2% dimethyl sulfoxide to raise the solubility, the triculture barrier starts to lose some of the barrier characteristics and we are concerned about integrity. We are currently looking at reported solubility values in water and using this as a criterion prior to ordering the lipophilic compounds. Compounds including tris(1-chloro-2-propyl) phosphate (TCEP), phthalic acid, hydrocortisone, and potentially some lower brominated PBDEs are in the process of being investigated to enhance the prediction value of the model. For instance, TCEP has a reasonable water solubility of approximately 3 mg/ml, which should be fine to measure the Papp values.
While these observations hinder the ability to calculate an apparent permeability coefficient, it does indicate that cisplatin, methoxychlor, and perchlorate will readily permeate the BBB and demonstrates that there exists a high probability of a neurotoxic event if these compounds are present in the bloodstream at higher concentrations. We are running additional permeability studies and are acquiring more neurotoxicants to continue throughout the upcoming year with the goal to complete all bidirectional permeability measurements by March 2023. This will enable us to complete Objective 1 of grant focused on adapting a presently available low throughput triculture model of the human BBB to develop the predictive exposure model.
To estimate the exposure rates at steady state concentrations, we are using two parameters: 1) the influx rate of permeation into the brain (Ap→Bl); and 2) the efflux ratio calculated from the permeation rates of compounds out of the brain divided by the permeation rate into the brain
(Bl→Ap/ Ap→Bl). Fast permeability across the BBB would suggest that the compound can reach the brain parenchyma at a relatively quick rate, suggesting that exposure should not be limited by the barrier in vivo. When influx permeation rates rise above a Papp value of ≈10 x 10-6 cm/sec, there is concern that the influx into the brain will be significant in vivo. However, we also need to consider the rate at which the compound is transported out of the brain at steady state during chronic exposure. Thus, it is important to evaluate permeation in both directions by the efflux ratio to predict risk during chronic exposure. If the efflux ratio is greater than ≈1.5-2.0, it suggests that transporters are actively excreting the compound out of the parenchyma and opposing the permeation inward. Conversely, when the efflux ratio is below ≈0.6-0.5, it suggests that transporters are actively transporting the compound into the brain parenchyma that may lead to accumulation. We included a predicted exposure level for the plasma concentration of compounds at steady state (CSS) in Table 1. Higher CSS values will lead to an increased risk of neurotoxicity in subjects who are exposed when the risk is medium or above.
We would like to note that we have encountered some additional delays in receiving tissue culture supplies due to the SARS-CoV2 pandemic over the past year. While these issues did slow some of the progress, it appears that these issues are resolved as of now.
Objective 2: Integrate permeability measures with human-based physiological parameters and compound properties relevant to their disposition in the brain to predict Css,brain of compounds.
Modeling work over the past year has focused on (1) data analysis within the set of compounds, (2) comparison of the results with existing ToxCast data and (3) initial work on a BBB-including PBTK model.
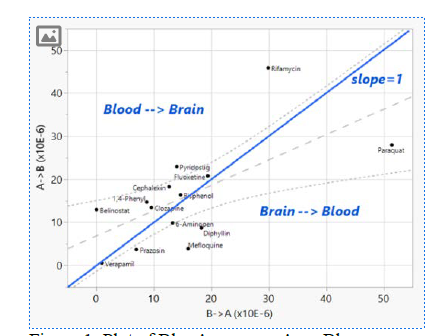
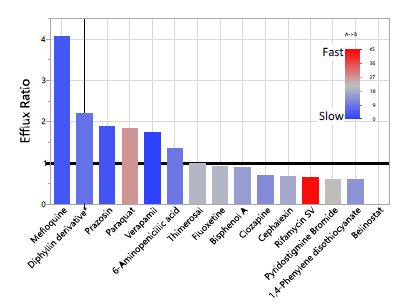
Analysis within the data itself shows that our compound set has a wide range (5 logs) of permeabilities in both the Bl→Ap and Ap→Bl directions. A plot of the two flux directions is given in Figure 1. A plot of the ratios is given in Figure 2. Note that for an acute exposure, the rates (particular the blood to brain rate, Ap→Bl) may be most important, whereas for a chronic exposure, the efflux ratio may matter most. Therefore, the efflux ratio might not give the full story and both the ratio and the two permeabilities give useful information. For example, though Rifamycin SV’s efflux ratio is 0.65 (it is not expected to concentrate in the brain based on our data) it transfers from the blood to the brain very quickly.
We have examined correlations between our measured permeabilities and compound specific PK parameters from the ToxCast database. Using 31 parameters we searched for correlations with Bl→Ap, Ap→Bl and the ratio Bl→Ap/Ap→Bl. For the ratio, we found significant correlations with BIOCONCENTRATION_FACTOR_TEST_PRED p<0.01. For Bl→Ap and Ap→Bl no significant correlations were found.
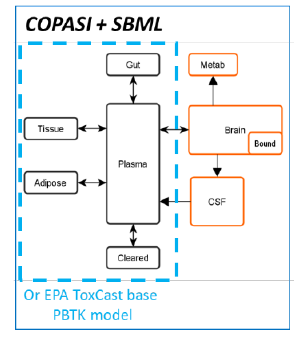
Finally, we have begun work on a BBB-Brain enabled PBTK model. We will extend an existing PBTK model available in EPA’s HTTK R package with a BBB-Brain component. To facilitate this, Dr. Sluka has joined the EPA-HTTK Partners working group. In addition, we have started work on a very basic COPASI+ SBML model shown in Figure 3.
Our final deliverables for the modeling aspect of this project remain unchanged.
1. BBB focused PBTK (perhaps as an extension to the HTTK-R package)
2. Estimates of chronic dose of interest Css,brain informed by estimates of BBB crossing
Figure 1: Plot of Bl→Ap versus Ap→Bl permeabilities. Blue line indicates Efflux Ratio = 1. Gray dashed lines are regression: R^2=0.42,
p=0.009.
Figure 2: Distribution of the Efflux Ratios. Large ratio is active export, small is active import.
(Belinostat, ratio = 0.004, is a CNS drug.) Ap→Bl rate is given by the bar color with red being rapid and blue being slow transfer.
Figure 3: Simplified PBTK layout with BBB and brain.
Future Activities:
See the results described in the Progress Section for Objectives 1 and 2. While the SARS-CoV2 pandemic had initially slowed progress in the laboratory, we are on schedule to meet our deadlines in year 3 of the award.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 10 publications | 6 publications in selected types | All 6 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Holub P, Wittner R, Mascia C, Frexia F, Müller H, Plass M, Allocca C, Betsou F, Burdett T, Cancio I, Chapman A. Towards a Common Standard for Data and Specimen Provenance in Life Sciences. Zenodo 2021. |
R840027 (2022) |
Exit |
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.