Grantee Research Project Results
2017 Progress Report: Initial Annual CEMALB Progress Report: Human Health Effects of Environmental Pollutants
EPA Grant Number: CR835785Subproject: this is subproject number R835785 , established and managed by the Center Director under a main grant
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Center for Environmental Medicine, Asthma, lung biology
Center Director: Peden, David B
Title: Initial Annual CEMALB Progress Report: Human Health Effects of Environmental Pollutants
Investigators: Peden, David B , Jaspers, Ilona , Hernandez, Michelle L
Current Investigators: Peden, David B , Jaspers, Ilona , Hernandez, Michelle L , Alexis, Neil , Caughey, Melissa , Rebuli, Meghan E
Institution: University of North Carolina at Chapel Hill
EPA Project Officer: Schmitt, Michael
Project Period: April 1, 2015 through March 31, 2022
Project Period Covered by this Report: April 1, 2017 through March 31,2018
Project Amount: $9,800,000
RFA: Human Health Effects of Environmental Pollutants (2014) RFA Text
Research Category: Human Health
Objective:
(1) Discussion of the research performed during the reporting period and results (outputs/outcomes) that have been generated.
As originally proposed, this center is organized to be responsive to the research areas outlined in the funding announcement, which included: a) Multi-pollutant Interactions, b) Health Effects Associated with Inhaled Particulate Pollutants Derived from Specific Sources, c) Susceptibility to Air Pollutants, d) Social Determinants of Health, e) Intervention Studies, and f) Biological Mechanisms.
As noted in an earlier progress report (submitted 6/30/16) for the period beginning on 4/1/2015, this center has been revised and is focused on three projects. Project 1 is led by David Peden and is entitled “Development of Interventions to Mitigate Inflammatory Response to Pollutants”. Project 2 is led by Ilona Jaspers and is entitled “Health Effects Associated with Inhaled Particulate Pollutants Derived from Specific Sources”, but has been revised during the reporting period to “Development of Methodologies to Assess Health Effects Associated with Pollutant Exposures”. Project 3 is led by Michelle Hernandez and is entitled “Effect of bio-aerosols (LPS) on systemic and cardiovascular responses”. These three projects respond to the areas “Health Effects Associated with Inhaled Particulate Pollutants Derived from Specific Sources”, “Susceptibility to Air Pollutants”, and “Intervention Studies”. It should be noted that while each project is led by Drs. Peden, Jaspers and Hernandez respectively, there is robust cross-collaboration by the PIs across each project.
Additionally, the funding announcement outlines the potential for additional program specific supplements to base funding to targeted program needs. During this initial year, three “above base” projects were proposed and agreed upon by the steering committee, all of which led by Dr. Jaspers (UNC) and Dr. Diaz-Sanchez (EPA). They are entitled “Large Dataset Analyses to Determine Pollutant- induced Adverse Health Effects”, “Environmental Health Education”, and “Autophagy as a Key Determinant of Cell Fate in Air Pollution Health Effects Among Healthy and Type 2 Diabetics”. Listed below are project summaries from the project year 4/1/17 to 3/31/18. Detailed descriptions of each project are provided in the detailed project reports, which are provided with this overall report.
Project 3: Establish whether LPS inhalation induces Systemic inflammation and CV inflammation
- Overview and Objective of Research: There is a growing body of literature supporting that short term exposure to LPS, a biological constituent of particulate matter, is associated with increased blood pressure in controlled human exposures (Zhong et al, Endotoxin and β-1,3-pDp-Glucan in concentrated ambient particles induce rapid increase in blood pressure in controlled human exposures, Hypertension 2015). The innate immune protien IL-1β, a byproduct of LPS-induced inflammation, appears to serve as a central mediator of systemic and CV responses. Short-term changes in air pollution are also associated with changes in stiffness of the conduit arteries and with exacerbations of heart failure. Moreover, inhibition of IL-1β activity with anakinra improves vascular endothelial function and left ventricular contractile function in patients with chronic inflammatory diseases. Thus, it is plausible that through IL-1β release, LPS adversely affects CV outcomes by modifying heart rate variability through autonomic control of heart rate, impairing endothelial function, increasing vascular stiffness, and depressing myocardial contractility.
The objective of Project 3 is to examine the role of biological components of PM on cardiovascular outcomes. To accomplish this overall objective, two protocols were developed using inhaled Clinical Center Reference Endotoxin as a model PM exposure.
Progress Summary:
Project 1: Development of Interventions to mitigate Inflammatory Response to Pollutants:
The aims of Project 1 were modified in year 1, and outlined in previous years project report. As noted at the time of modification in year 1, the primary activities of this project are to: 1.) to identify characteristics of people at risk for adverse response to pollutants; 2.) develop specific interventions to mitigate the inflammatory r esponse to pollutants; 3.) to develop personalized wearable sensors to allow individuals to understand their personal exposures and physiological state (e.g. lung function, pulse, accelerometry) in conjunction with the ASSIST center at NCSU: and 4.) to develop programs in environmental precision medicine and health using big data approaches (with funding of an above base supplement to address some aspects of this aim). Each aim is outlined below, with progress in the protocols underway to address these goals are summarized below:
Progress in each aim:
Aim 1.) To identify characteristics of people at risk for adverse response to pollutants:
Protocol 15-1775 (D.Peden, PI): To identify persons who are susceptible to WSP-induced inflammation and examine the role of GSTM1 and other factors in this susceptibility. This is a single challenge protocol to identify responsive and non-responsive volunteers to 500 mg/m3 of woodsmoke particles (WSP). Thirty-seven volunteers underwent exposure to 500 μg/m3
WSP for 2 hours with alternating 15-minute periods of exercise and rest to achieve minute ventilation of 20 L/min/m2. Induced sputum samples were obtained prior to, 6 and 24 hours after exposure for assessment of inflammatory cells and cytokines. As of 6/25/18, sputum data from 29 of 31 challenged volunteers have been studied.
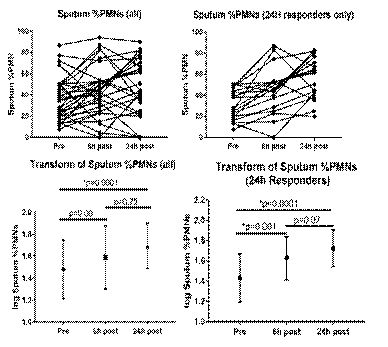
Using our established criterion that change in PMNs of at least 12% defined a responder 24 hours after challenge, we observe that 33% of volunteers are non-responders, reasonably consistent with what we have observed for ozone and LPS. Figure 1 depicts PMN responses at baseline, 6 hours and 24 hours following WSP challenge, with significant effects at 24 hours seen across the entire cohort (Top Left Panel, with statistical significance of log transformed data shown in the Lower Left Panel) as well as in those identified as responders
(Top Figure 1: Response of volunteers to woodsmoke particle challenge
Right Panel, with statistical significance of log transformed data shown in the Lower Right Panel). Thus far, GSTM1 does not appear to be a determinant of response to WSP challenge. Across the entire cohort, baseline levels of IL-8 (but not IL-1b or IL-6) correlate well with 24-hour %PMN responses (r=0.52, p=0.0096), similar to our observations with ozone. Baseline levels of PMNs (expressed as %PMNs) also marginally correlate with response to WSP (r=0.36, p=0.06). Recruitment continues, though these data suggest that WSP responsiveness is not influenced by GSTM1, though baseline inflammation may be important in subsequent response to WSP.
Protocol 15-2677 (D. Peden PI): Human biological responses to low level ozone: This study is designed to determine if exposed volunteers have cardiovascular, systemic and respiratory tract response to low level ozone which varies between 0.06 and 0.08 ppm O3 vs. filtered air. Additionally, ozone monitors developed in Aim 3 are being tested in this protocol. To date, 19 have completed the study.
Data analyses of LPS response (D. Peden, N Alexis): We have currently reviewed data from 38 volunteers challenged with LPS and defined as responders (27) and non-responders (11) with regard to PMN influx into the airway. We had sputum mRNA from these volunteers. We observed that responders versus non- responders had significantly lower % PMN levels at baseline (29% vs. 62%) and greater post minus pre exposure changes (delta) in %PMN levels (delta: 30 vs 9). At baseline, 13 genes were identified as being differentially expressed in responders and non-responders. Ten of the 13 genes (77%) displayed increased expression in responders, among them cyclooxygenase 1 PTGS1, the enzyme that converts free arachidonic acid to prostaglandin p. Three of the 13 genes (23%) displayed decreased expression in responders, including IL-8, a potent PMN chemoattractant, and IL18RAP, an IL-18 receptor binding enhancer that mediates downstream NF-kB activation. We anticipate submission for publication in the near future.
Aim 2.) Develop specific interventions to mitigate the inflammatory response to pollutants;
Protocol 15-1938: Effect of gamma tocopherol enriched supplementation on response to inhaled O3 exposure (D. Peden, MD, PI). This is a double-blinded placebo controlled study of the effect of 1200 mg of oral gamma tocopherol for 4 doses over 48 hours on inflammatory response to 2-hour exposure to 0.25 ppm O3. To date 17 volunteers have been enrolled, with 12 completing the protocol, 2 in study and 3 failing to complete study for voluntary reasons. After 15 volunteers have been enrolled, a planned interim analysis will be undertaken. If there are definitive results (either there is a clear-cut effect of gT or study futility is recognized) the study will be terminated and results published.
Protocol 17-2303: (Allison Burbank, MD, PI). A phase II randomized, double blinded, placebo-controlled study of gamma tocopherol-enriched supplement on lower airway responses to inhaled wood smoke in healthy adults. This study will examine 20 volunteers exposed to 500 micrograms/m3 woodsmoke particles to determine if 2 doses of 700 mg of gamma tocopherol will mitigate the impact of WSP on airway inflammation. This protocol was the focus of substantial regulatory review, and all required reviews have been completed (IND, UNC Scientific Review Committee, EPA and IRB reviews) and the first volunteer has been recruited, having been identified as responsive in protocol 15-1775.
Protocol 1529: Efficacy of Fish Oil or Olive Oil Supplementation on the Health Effects of Ozone Exposure in Healthy Young Subjects (James Samet, PhD, EPA, PI). As noted in last year’s report, CEMALB investigators are co-investigators in this study, and FDA approval was obtained for this protocol as an amendment to Dr. Peden’s FDA IND for exposure to ozone (IND 71475) in 2/2017. The study is underway, but still relatively early in the study and no results are available
Aim 3.) To develop personalized wearable sensors to allow individuals to understand their personal exposures and physiological state
Protocol 14-1849 (D. Peden, PI): Exercise training and monitoring for environmental research This protocol is used to undertake rapid cycle improvement for testing of personal monitors and sensors for physiologic parameters (ECG, respiratory rate, accelerometry) developed by engineer-investigators at the NCSU-based
assist center. To date, algorithms to estimate minute ventilation from accelerometry data are being developed, to be able to estimate the minute ventilation term in pollutant dosing in ambient settings. To date, 57 volunteers have completed this protocol, in which 2 sensor arrays have undergone development.
Aim 4.) To develop programs in environmental precision medicine and health using big data approaches
Additionally, Dr. Peden, is a key investigator in the UNC Biomedical Data Translator research team (led by Stan Ahalt of RENCI at UNC), which is part of a multi-site national consortium funded by the NIH (the National Center for Advancing Translational Sciences) to examine how various large data sets can be leveraged together to address medical, health and basic science questions. A unique element of the UNC proposal, which has now been adopted by the overall consortium, is inclusion of environmental data in big data assessments of health data. The UNC team has done initial assessments with 10,000 asthma patients showing increased exposure to PM exposure in patients with increased acute episodes and severity. These data remain under analyses and publications are being developed, and new environmental health outcomes are being considered. This project also led to an above base study by EPA and UNC scientists that are outlined under Above Base projects.
Modified Title: Project 2 Development of Methodologies to Assess Health Effects Associated with Pollutant Exposures
In an era of personalized and precision medicine, efficacy data of therapeutic agents for airway disease should take into account effect modifiers, such as BMI and GSTM1 genotype, to optimize treatment strategies.
- Overview and Objective of Research: Oxidant pollutants derived from the incomplete combustion of organic matter, such as burning of biomass or woodsmoke, are of increased global public health concern and associated with increased susceptibility to respiratory infections, including viral infections in children. More than two billion people in the world use biomass (including indoor wood burning) as their main source of energy for domestic heating and cooking. Epidemiological evidence suggests that reducing exposure to biomass smoke exposure is associated with a decrease in upper and lower respiratory tract infections. Collaborative studies among investigators from UNC and EPA developed a human in vivo exposure and viral challenge study to translate these findings into human. These studies were built on human in vivo experimental models developed by investigators at UNC using the FDA-approved live attenuated influenza virus (LAIV) vaccine as a model for viral infections in human volunteers. Inoculation with LAIV causes a self-limiting replicative infection, which induces similar cytokine and antiviral host defense responses as community-acquired infections. This human LAIV inoculation model was previously used to assess interactions between exposure to diesel exhaust and viral infections, which demonstrated that underlying allergy rhinitis enhances the interaction between DE and viral infection, leading to increased viral replication and markers of allergic inflammation.
Due to the global health concerns related to adverse health effects induced by exposure to smoke derived from biomass fuel burning or woodsmoke, we hypothesize that exposure to woodsmoke or biomass smoke enhances susceptibility to viral infections by modifying mucosal immune responses.
Modifications Made to the Original Objectives: While the controlled exposure of humans to DE or woodsmoke facilitate establishing cause-and-effect relationships, these observations need to be translated into the community and populations naturally exposed to environmental contaminants. To address this gap, we propose to develop, optimize, and validate novel non-invasive tools to sample the respiratory mucosa. The goal of these studies will be to establish methodologies enabling investigators to assess markers of exposure and biomarkers of health effects in naturally exposed populations.
- Progress Summary/Accomplishments (Outputs/Outcomes): Using our established model combining human in vitro and in vivo models of viral infections, we propose to address this hypothesis with the following aims:
Objective 1: Determine the Effects of Controlled Woodsmoke exposures on markers of viral replication and mucosal immune responses in healthy human volunteers.
Summary of Experimental Design:
Subjects (n=20 for each group) were randomized for exposure to either air or 500 ug/m3 WSP and inoculated with LAIV shortly after completion of the exposure. Baseline and post-inoculation nasal lavages, nasal biopsies, collection of epithelial lining fluid (ELF), and collection of peripheral blood were done by CEMALB personnel, similar to our previous studies using DEE exposures (i.e. FLAIR). Clinical protocol development, IRB approval, recruitment of subjects, and proper maintenance of databases were done in collaboration with the Medical, Physiology and Study Coordination Core (led by Drs. Noah and Jaspers).
All samples collected as part of these protocols will be used to analyze the endpoints identified above in collaboration with the Biorepository and Cell Biology Core (led by Drs. Alexis and Jaspers).
Objective 2: Develop, Optimize, and Validate Non-invasive, Field-deployable Sampling Techniques, Allowing Investigators To Sample The Respiratory Mucosa For Pulmonary Biomarkers
Summary of Experimental Design
The Proposed Specific Activities to Optimize, Validate, and Apply this Methodology Include the Following:
- Compare Mediator Profiles in ELF, Induced Sputum, and BALF
To do so, ELF, induced sputum, and BALF collected from the same subject will be examined for the relative amount of cytokines/chemokines (using multiple ELISA platforms), neutrophil elastase, CC16, total protein as well as pH. Mediator profiles will be compared within each individual to determine whether there are respiratory tract region-specific mediators.
- Assess Potential Health Disparities In Environmental Triggers Of Asthma
To do so, ELF will be collected from a small cohort of adolescent patients (age 12-18) with asthma, who are currently enrolled into a clinical trial assessing asthma symptom control in Dr. Hernandez’ clinic in Raleigh. ELF samples will be assessed for soluble markers indicative of 1) overall inflammation (IL-1β, IL-8, TNFα, IL-6, prostaglandin E2), 2) allergic inflammation (eosinophil cationic protein, IL-4, IL-13, mast cell tryptase), and 3) neutrophilic inflammation (neutrophil elastase, myeloperoxidase) before and after asthma control adjustments. Data will be stratified based on race and ethnicity to determine any potential health disparities in asthma control.
- Determine Pollutant-induced Changes in ELF
To do so, we envision incorporation of ELF collection all ongoing controlled exposure studies as well as community-based field studies. For example, a longitudinal ELF collection study could be assessed for changes in inflammatory markers and association with recent air pollution episodes.
Progress:
We have demonstrated that collection of ELF is a validated and valuable novel and non-invasive tool to gain information on biomarkers of exposure and disease severity. A manuscript describing the methodology has been published.
Originally Proposed Milestones:
We predicted that this methodology could be included or added to ongoing cohort studies examining the link between environmental parameters and adverse health effects in the community. These milestones are ongoing and not time-limited.
Achieved Milestones:
The ability to adapt this technique to other studies has been recognized by other groups, who are currently using collection of ELF to a) determine the effects of flavored e-cigarettes on inflammation, b) determine biomarkers of inflammation after controlled exposures to air pollutants, c) examine effects of second-hand exposure to e-cigarette aerosols, and d) identify air-pollutant effects on disease exacerbations in COPD patients.
Objective 1: Determine the Effects of Controlled Woodsmoke exposures on markers of viral replication and mucosal immune responses in healthy human volunteers.
Sample collection and analysis has been completed. Future efforts will focus on summarizing and publishing findings from the controlled woodsmoke exposure study
Objective 2: Develop, Optimize, and Validate Non-invasive, Field-deployable Sampling Techniques, Allowing Investigators To Sample The Respiratory Mucosa For Pulmonary Biomarkers
Focus on implementing the sampling technique in to other ongoing studies and expand the scope of sample analyses. We have conducted initial pilot experiments to determine exposure-dependent changes in the nasal microbiome.
Protocol 17-3351: Effects of 20,000 EU of Clinical Center Reference Endotoxin (CCRE) versus placebo on systemic and cardiovascular inflammatory responses in mild asthmatics and healthy volunteers (M. Hernandez, PI) was approved by the UNC IRB.
Individuals who exhibit a systemic inflammatory response to inhaled LPS (identified in EndoScreen) will be recruited for this study. Systemic inflammatory response to inhaled endotoxin will be determined by measuring change in peripheral blood neutrophil counts, a biomarker of systemic inflammation, following inhaled CCRE vs saline placebo. Blood pressure, heart rate variability (HRV), vascular stiffness (by flow mediated dilation, or FMD) and left ventricular strain (LVS) will be measured before and after CCRE and placebo exposure to investigate the effect of endotoxin-induced systemic inflammation on cardiovascular function. UNC IRB and EPA approval were obtained. Devices to measure HRV were ordered to begin this project; the project will begin pending delivery of these devices followed by training sessions of study coordinator and physician staff.
While proceeding with these approval processes, we also queried our Biorepository and Data Base and identified data from healthy volunteers and allergic asthmatics who participated in our past endotoxin studies using 20,000 EU CCRE within the past 10 years. We have previously undertaken 2 studies of a nutritional
anti-inflammatory therapy, gamma tocopherol (γT), the dietary isoform of vitamin E, and have shown that it has anti-inflammatory properties against LPS-induced inflammatory responses. Biological data include neutrophil counts and cytokine levels in the blood and sputum at baseline and 6 hours after challenge. We used the data from both studies to address the role of specific risk factors (GSTM1 status and BMI) in response to LPS and effectiveness of gamma tocopherol
In these studies, a total of 28 subjects (13 healthy volunteers and 15 subjects with mild asthma) underwent cross-over studies where they received treatment 1200 mg gamma Tocopherol and placebo treatment. γT supplementation reduced post-challenge sputum neutrophilia compared to placebo in both HV and asthmatics
(p=0.03 and p=0.04, respectively). Pooling both healthy volunteers and asthmatics, the effects of γT supplementation in mitigating LPS-induced sputum neutrophil recruitment were still evident (p=0.01). Using linear regression modeling, the effect of BMI and GSTM1 genotype on the γT treatment response to inhaled endotoxin was assessed. We found that neither BMI nor GSTM1 genotype had any effect on γT treatment response to inhaled endotoxin.
This work was presented at the AAAAI/WAO Congress meeting in March 2018 in Orlando, FL. A Sood, AJ Burbank, CG Duran, K Enders, H Zhou, DB Peden, ML Hernandez. Gamma Tocopherol (γT) Supplementation Reduces Endotoxin-Induced Sputum Neutrophilia in Healthy Volunteers and Asthmatics Regardless of BMI or GSTM1 genotype. The resulting manuscript is currently in preparation for submission to the Journal of Allergy & Clinical Immunology.
Protocol 12-1588: Observational assessment of baseline asthma control as a susceptibility factor for air pollution health effects in African-American children with persistent asthma (Teen AIRE Study)
Asthma status was assessed using NHLBI guideline criteria, with 14 volunteers having poorly controlled asthma and 11 having well- controlled asthma. These volunteers were followed over a course of 6-8 weeks through 6 visits. Endpoints included weekly measurements of pulmonary function, assessment of asthma control through the Asthma Control Questionnaire, autonomic control of heart rhythm and vascular markers of inflammation, and clotting/coagulation and exposure. We specifically focused on the health effects attributed to low levels of ozone in the 5 days preceding each participant’s clinic visits in their residential locations, controlling for exposure to ambient fine particle (PM2.5) and coarse particle (PM-10) exposures. Because BMI has been previously associated with differential responses to ambient air pollution, we stratified health effects by BMI status (<25, normal weight v. >25, overweight).
All participants were exposed to low levels of ambient ozone (below the NAAQS of 70 ppb) throughout the course of the study and received a high level of clinical care, including stepping up of asthma controller medications as needed based on asthma control status. Despite this level of medical management, we still observed an association between increased ambient ozone concentration and reduced lung function measurements (FVC and FEV1). For % predicted FVC, the strongest effect was observed for ozone concentrations in the 24-hour period preceding clinic visits (lag 0), where there was a decrease of 2.7% predicted FVC (p =0.02). This decrement diminished for the preceding 24 to 48-hr period (lag 1) to 1.3%, and for the previous 5 day moving average a decrease of 2.9% was seen near significance (p=0.07). Similar results were observed for % predicted FEV1, where there was a 2.5% decrease at lag 0 (p=0.07) and a decrease of 1.6% at lag 1. In a two-pollutant model, we examined whether ozone effects were confounded by PM2.5. The effect estimates were minimally altered with a decrease of 2.5% and 1.4 % seen at lag 0 and lag 1, respectively.
Additionally, we provide what we believe is the first report of systemic changes by short-term ozone exposure in children. In particular, a highly significant (p<0.001) increase in total cholesterol levels of 5.56 mg/dL at lag 1, per IQR of ozone was observed. Changes in LDL followed the same pattern. At the same lag 1, an association with a 3.6 mg/dL increase in LDL was also observed (p=0.06). No changes in triglycerides or VLDL were observed. Additionally, cholesterol and LDL were elevated at lag 1 among those with high BMI.
Our results suggest that low-level ozone can still impact respiratory outcomes regardless of baseline control status or daily controller treatment, and implies that environmental awareness or novel interventions that are not currently used in asthma may need to be employed in medical evaluation and treatment guidance for environmental asthma.
This work was presented at the March 2018 AAAAI/WAO meeting in Orlando, FL, and the resulting manuscript, “Low level Ozone has both systemic and respiratory effects in African-American adolescents with asthma” was favorably reviewed by the Journal of Allergy & Clinical Immunology, and was resubmitted in a revised version: Michelle L. Hernandez, MD, Radhika Dhingra, PhD, Allison J. Burbank, MD, Krista Todorich, MD, Celia E Loughlin, MD; Marcia Frye MD, Kelly Duncan, PhD, Carole Robinette, MS; Katherine Mills, BA, Robert B Devlin, PhD, David B. Peden, MD, David Diaz-Sanchez, PhD.
Above Base Project: Large Dataset Analyses to Determine Pollutant-induced Adverse Health Effects Project 4:
- Overview: Increasingly, large datasets generated through emerging “omics” technology, including epigenomis, metagenomics, genomics, proteomics, and metabolomics are used to provide unbiased assessment of pollutant-induced adverse health effects. These technologies can identify biomarkers of exposure, biomarkers of health effects, predict susceptibility, understand biological mechanisms, and indicate preventative strategies. Investigators in CEMALB have used these technologies to understand responsiveness to pollutants, such as ozone, investigate epigenetic markers of susceptibility, and identify novel biological pathways. The trainees supported through this project are Virginia Bass, Emma Bowers, and Kimberly Stratford
Using an in vitro model of ozone exposure, work has been conducted to identify the underlying mechanisms of adaptation to repeated exposures that occurs in areas such as Los Angeles. In addition, studies are underway to identify potential mechanisms mediating differential responsiveness in humans. For this project, the following goals have been accomplished (Emma Bowers):
-
- Characterized responses to single and repeated ozone exposure in 13 different primary cell donors
- Investigated antioxidant contributions to the ozone adaptive response
- Identified differences in cellular signaling between ozone-sensitive and insensitive primary cell donors
- Successfully defended doctoral dissertation and published 3 peer-reviewed manuscripts with additional papers in submission/under review.
Using existing blood samples collected from subjects exposed to particulate air pollutants, analyses were conducted to correlate blood micronutrient levels (e.g. Vitamin D) with clinical biochemical endpoints, which indicate adverse responses in the body. The goal of this study is to determine if 1) vitamin D levels vary at baseline between the subjects exposed to clean air and particulate pollutant exposures, 2) vitamin D levels differ due to exposures to clean air or particulate pollutant exposures, and 3) vitamin D levels correlate with increased adverse effects as indicated by changes in common clinical endpoints. For this project, the following goals have been accomplished (Kimberly Stratford):
-
- Identified circulating Vitamin D levels as an important determinant of cardiopulmonary responses caused by exposure to particulate air pollutants in rodent models
- Characterized the role of Vitamin D deficiency in acrolein-induced cardiovascular health effects
- Successfully defended doctoral dissertation and published 3 peer-reviewed manuscripts with additional papers in submission/under review.
Oleic acid (OA) is frequently used as a representative fatty acid, found for example in meat-cooking fumes or biodiesel exhaust. Vascular damage and acute lung injury has been observed with OA infusion in models of acute respiratory distress, but it is not yet established whether OA deposited in the lung is able to produce adverse pulmonary and cardiovascular effects associated with exposure to ambient air pollution.
Using human in vitro models, this research will examine the role of OA on pulmonary and cardiovascular endpoints. For this project, the following goals have been accomplished (Virginia Bass):
-
- OA exposure inhibited relaxation of rings to the vasodilator acetylcholine (which acts via endothelial cell mechanisms). This inhibition of vasodilation is similar to the finding that exposure of volunteers to either diesel or biodiesel exhaust attenuated the response of endothelial cell mediated vasodilation.
- OA changed lung function parameters in rats in vivo, increased markers of inflammation and lung injury
- OA increase iron load in lung macrophages in vivo and co-administration of OA and ferric ammonium citrate (FAC) tended to increase the responses (neutrophilia, cytokines) relative to oleic acid alone
Above Base Project: Autophagy as a Key Determinant of Cell Fate in Air Pollution Health Effects Among Healthy and Type 2 Diabetics Project 5:
- This project supports Andres Henriquez working with Urmila Kodavanti (his EPA mentor). The original focus was the role that autophagy in mediating resolution (or lack of resolution) of ozone- induced lung inflammation and metabolic responses. However, while it was possible to reliably establish techniques for assessing critical markers of autophagy using Western blotting, it was problematic to find right the correct analytical tools antibodies that provided consistent results when performing repeat assessments. Also, after overcoming these difficulties, it was found that ozone-induced cellular changes in vitro did not involve autophagy changes that could be detectable. BEAS-2B cultures were used and highly variable results were obtained during each repeat led to difficulty in reliably assessing this process. As a result, the project was modified to assess the contribution of stress hormones in ozone-induced lung injury and inflammation. Since the other ongoing projects in the lab involving the role of stress hormones has resulted in novel insights, these are being further explored in relation to the roles of epinephrine and corticosterone in mediating ozone-induced lung injury and inflammation in this project. This project now consistent with the EPA program focus of Biological Mechanisms outlined in the Funding Announcement.
- Progress on Required Project
In pursuing the new focus, the involvement of immune cell changes and pulmonary inflammation were assessed at protein and gene expression levels in the lungs, BALF and serum samples from a study involving ozone exposure in rats with SHAM, adrenal demedullation or total bilateral adrenalectomy surgeries to examine the contribution of stress hormones. Additional assessment of samples involving lung tissue gene expression profiling in SHAM and adrenalectomized rats using global RNASeq is completed as the specific aim 1. Studies proposed for specific aim 2A involving pharmacological means of intervening stress hormone receptors have been completed. Aim 2B study, which will include adrenalectomy in conjunction with pharmacological interventions, has also been completed. Andres Henriquez successfully defended his doctoral dissertation and this work has already resulted in 7 publications with additional publications currently submitted/under review.
Above Base Project: Environmental Health Education Project 6:
- Both the EPA and CEMALB have a long and distinguished track record in support of activities that promote environmental public health education. Environmental Health is a fast moving field that encompasses toxicology, environmental medicine, microbiome, epidemiology, genomics, epigenetics, and many other fields. In addition, community engagement in public health research and in the dissemination of scientific facts is of increasing interest. Currently, CEMALB and UNC-CH at large has several initiatives to train students/postdocs by familiarizing them in the latest developments in these areas so as to enable them to understand better current and future environmental challenges. Linking state-of-the-art biomedical training with opportunities to actively engage in community-based public health research will better prepare our trainees for careers in Environmental Health.
In order to foster these initiatives, following goals are proposed:
-
- To attract potential trainees to the field of Environmental Public Health.
- To provide the opportunity for trainees to travel to meetings to further their education in Environmental Public Health
- To provide the opportunity for trainees to interact with experts in Environmental Public Health
- To enhance community-based research skills by supporting additional training opportunities
- Progress on Required Project
Opportunities for trainees to travel to meetings to further their education in Environmental Public Health
During the last reporting period, we received 4 applications and awarded 1 travel award. All four applications were reviewed by a committee composed of the members of the Curriculum in Toxicology executive committee and ranked based on their merit. The awarded trainee traveled to the 2018 Society of Toxicology meeting in San Antonio, TX to present her research and interact with other environmental health scientists. The following applicant and her research were awarded:
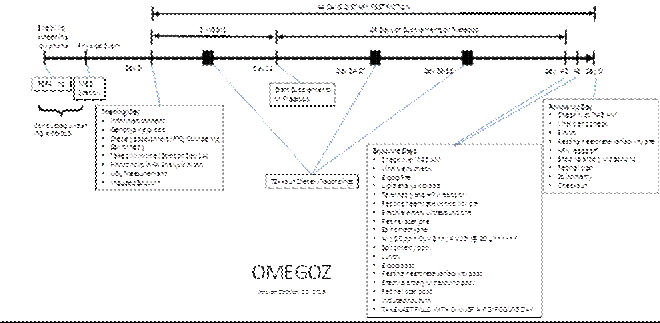
Above Base Project: Efficacy of Fish Oil or Olive Oil Supplementation on the Health Effects of Ozone Exposure in Healthy Young Subjects (OMEGOZ) Project 7:
Overview: A growing body of epidemiological data suggests an increased risk of cardiovascular events associated with air pollution. One of the common air pollutants, ozone, has been shown to induce oxidative stress and inflammation in the cardiovascular and respiratory systems. This proposal is to examine the efficacy of fish oil and olive oil in protecting cardiovascular and pulmonary functions after ozone exposure. The objective is to understand the mechanistic basis for the health effects of ozone in relation to previously studied particulate matter. Healthy young adults will be given dietary supplementation of fish oil, olive oil, or placebo for 4 weeks, then exposed to ozone for 2 hours in an exposure chamber. Cardiac rhythm, pulmonary function, vascular responses, endothelial function, and markers of coagulation and airway inflammation pre- and post- ozone exposure will be measured. This study is designed to build on the previous nutritional supplement interventions PM studies OMEGACON (IRB # 07-0190) and CAPTAIN (IRB # 11-1807), in order to understand the mechanism of action of particulate pollutants in comparison to that of ozone, a known oxidant air contaminant.Participants: Sixty healthy 18-35 year-old male and female subjects will be involved in the study. Methods: The subjects will be exposed to clean air for 2 hours on the first day, then ozone (0.3ppm) for 2 hours on the second day after being randomly supplemented for four weeks with omega-3 fatty acids, olive oil, or placebo. The study outline appears in the below schematic:
2. Progress on Required Project
While working on Omegoz, Liz optimized a protocol to analyze the bioenergetic profile of induced sputum samples using the Seahorse Extracellular Flux Analyzer. The analysis measures several aspects of both mitochondrial and glycolytic function. Following optimization, Liz processed the post- air and post-ozone exposure sputum samples (N= 4) collected from four Omegoz subjects. Seahorse analysis requires a minimum number of viable sputum cells for successful analysis. Sputum cell recovery has been variable to date with Omegoz and thus has presented a challenge with this part of the study.
Future Activities:
Endpoints:
Primary endpoints were markers of viral replication. Secondary endpoints will include 1) gene expression profile of NLF cells (measured by qRT-PCR and NanoString technology) and 2) Cytokines/chemokines in NLF (including IL-1ß, IL-6, IFNa, IFNb, IFNg, TNFa, IP-10, IL-8, RANTES, eotaxins, TARC). In addition, we collaborated with Dr. Wolberg, who determine markers of coagulation and clotting in plasma collected before and after the different exposures.
Progress:
Sample collection and analysis is complete and a manuscript has been written.
The following sample analyses have been completed: 1) Gene expression of NLF cells and 2) cytokines/chemokines (27 analytes) of NLF. These data are currently being analyzed in collaboration with the CEMALB Biostatistics Core (Dr. Haibo Zhou), as well as UNC collaborators with expertise in bioinformatics.
Our study demonstrated that WSP exposure did not significantly alter markers of viral replication but decreased antiviral host defense responses as marked by reduced levels of IP-10 in NLF and reduced expression of IL-10 in NLF cells. More importantly, our analysis indicated a Sex*Exposure interaction, prompting us to separately analyze WSP-induced changes in males and females. In addition, our data indicate that inflammation-related gene expression profiles significantly differ in males in females, at baseline, after LAIV inoculation, and after WSP exposure, demonstrating that WSP-induced changes in antiviral defense responses in the nasal mucosa occur in a sex-specific manner.
These data were presented at the annual American Thoracic Society meeting and are currently being summarized for publication.
Originally Proposed Milestones:
Completion of the analyses of “Woodsies” is anticipated by the end of 2016
Achieved Milestones:
We achieved the completion of the analyses of “Woodsies” and are currently finalizing the manuscript for publication.
Addition to Objective 1: After further consideration of existing data, it was decided to add additional experimental arms to SA1 to examine the effect of woodsmoke on allergic inflammation as well as real-life viral infections. These are additions to the original plan, which focused on examining the effects of ambient pollutant exposures on live-attenuated influenza virus (LAIV)-induced responses in healthy non-allergic volunteers. To do so, we will develop a new IND protocol to laboratory-grade human rhinovirus in normal healthy volunteers, with the goal to compare pollutant-induced alterations of virus-induced host responses in a real-world circulating respiratory virus.
Note: Dr. Hernandez provides an update on the progress of this additional objective in the summary for Project 3.
- Summary/Accomplishments Project 3 (Outputs/Outcomes):
Protocol 15-1458:Systemic inflammatory response to 20,000 EU Clinical Center Reference Endotoxin in Normal Adults (M. Hernandez, PI)
The goal of this study (also known as “Endoscreen”) is to assess 18 healthy volunteers with an inhaled LPS challenge to first establish responsiveness to inhaled LPS. It is estimated that up to 30% of the population may not be responsive to inhaled LPS. Inhaled LPS response will be established by detecting at least a 20% increase from baseline levels of neutrophils in the blood, and/or by a 10% increase from baseline levels of neutrophils in induced sputum samples. In addition, as secondary endpoints, we will evaluate inflammatory cytokine levels in systemic circulation and access for the presence of genes thought to be related to endotoxin response. Subjects will wear a cardiac monitor known as Faros 180 to monitor heart rate variability, an autonomic index that has been previously shown to be affected by high dose ozone exposure (Devlin RB et al, Circulation, 2012). The Endoscreen study will provide not only data on LPS responsiveness, but will also provide preliminary associations between changes in LPS-induced heart rate variability.
This study required us to submit an amendment (#55) to our CCRE IND: Protocol 15, Amendment #55 of IND BB-9998 “Airway response to clinical center reference endotoxin in healthy adults”. On 5/5/2016, we received notification from the FDA review coordinator no clinical hold on proceeding with this study, though minor revisions were requested to the document. We submitted the revised protocol on 5/23/16 to FDA, and an IRB application was submitted to UNC on 5/26/16. The IRB 15-1458 was approved on 6/28/2016. In the interim, the NIH chose to use a new source of endotoxin for inhalation challenge studies. NIH investigators then went through the FDA approval process to test the new endotoxin source. They received approvals in August 2017, and sent us new vials of endotoxin for inhalation in September 2017. Recruitment for this study began in November 2017.
Thus far, 6 volunteers have completed the Endoscreen Protocol with recruitment to continue.
Protocol 17-3351
We will continue recruitment for both inhaled LPS protocols, focused on examining the role of IL-1β on airway and systemic cardiovascular responses. We do not anticipate any significant delays in recruitment.
Protocol 12-1588
Development of Human Rhinovirus Infection Model, “pStudy of airway inflammatory responses to experimental rhinovirus infection”
In collaboration with Project 2 (Jaspers, PI), Drs. Hernandez and Burbank developed an IND application to the FDA to bring the experimental rhinovirus infection model to UNC. This will support Project 2’s additional experimental aims to compare pollutant-induced alterations of virus-induced host responses in a real-world circulating respiratory virus. This protocol was approved by the FDA, the UNC IRB (16-8283)and Institutional Biosafety Committee, and pending EPA approval for this reporting period.
The majority of severe exacerbations of asthma and need for hospitalizations are triggered by infection with respiratory viruses. Of these, rhinovirus is the most commonly implicated virus. Experimental infection with human rhinovirus (HRV) is a valuable tool for examining the pathophysiology of viral-induced exacerbations of underlying airway disease. This model has been safely used in both mild and moderate asthmatics, and also in subjects with COPD. Our primary objective is to establish the HRV-model in our center, to enable future work examining pollutant-induced alterations of virus-induced host responses.
The primary objective of this new observational study is to characterize in detail the clinical, physiologic, and inflammatory features of HRV infection in normal volunteers, while also evaluating the safety of HRV administration. This study will provide the pilot data needed for the design of subsequent studies evaluating innate immune responses to HRV infection in asthmatics, and also modulation of HRV-induced responses by inhaled pollutants. The duration of Protocol 1 will be approximately 2 years. The expected duration of subject participation is approximately 2 months.
After EPA approval, investigators will move forward with establishing the infrastructure to support the completion of this project.
Project 4:
Both Emma Bowers and Kimberly Stratford successfully defended their dissertation.
For the studies examining the effects of LCFA and more specifically OA on markers of pulmonary and cardiovascular endpoints (Virginia Bass)
-
- Investigate the effects of OA on endothelial cell function (mitochondrial function) and vascular contractility
- Identify the role of changes in iron metabolism in the effects of OA on pulmonary inflammation and endothelial cell dysfunction
Project 6:
Opportunities for trainees to travel to meetings to further their education in Environmental Public Health
Requests for applications for this award will start in December, 2018 and complete applications need to be received by middle of February, 2019 to be eligible. We anticipate 3-6 applications to make 1-2 awards.
Opportunities for trainees to interact with experts in Environmental Public Health
The organizing committee has ranked potential experts in environmental public health to be invited for a seminar in the fall. We anticipate the invitation to be completed before the end of July for a visit in the fall 2018.
Enhance community-based research skills by supporting additional training opportunities
Since these applications are received and reviewed on a rolling basis, we cannot accurately predict the number of potential applications. However, we anticipate receiving several applications over the next 6 months.
Note: Starting in April, 2018, this Project was amended to include “research and training support for trainees interested in conducting research in the field of Environmental Public Health.” This mechanism will be used to provide short-term support for trainees at any level to conduct research in environmental health research.
Syed Masood, a graduate student at UNC-CH, started a 10-week summer research project on June 4th, 2018 with Dr. Samet and is supported through this mechanism.
Project 7:
This project is in progress and sputum samples continue to be processed in the Alexis lab for analysis of inflammatory endpoints. Elizabeth Cortiselli’s specific involvement however, with the Seahorse (extracellular flux/bioenergetics) analysis of the sputum samples has been discontinued and diverted to another project.
Journal Articles: 50 Displayed | Download in RIS Format
| Other center views: | All 83 publications | 50 publications in selected types | All 50 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Bowers EC, McCullough SD. Linking the epigenome with exposure effects and susceptibility:The epigenetic seed and soil model. Toxicological Sciences 2016;155(2):302-14 |
CR835785 (2016) CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Hickman E, Smyth T, Cabos-Uribe C, Immormino R, Rebuli M, Moran T, Alexis N, Jaspers I. Expanded characterization of in vitro polarized M0, M1, and M2 human monocyte-derived macrophages: Bioenergetic and secreted mediator profiles. PLOS ONE 2023;18(3):e0279037 |
CR835785 (Final) |
Exit |
|
|
Kobernick AK, Peden DB, Zhou H, Zhou Q, Dillon MA, Alexis NE. Reproducibility of the inflammatory response to inhaled endotoxin in healthy volunteers. Journal of Allergy and Clinical Immunology. 2016;138(4):1205-7 |
CR835785 (2015) CR835785 (2020) |
Exit Exit |
|
|
Martin WK, Tennant AH, Conolly RB, Prince K, Stevens JS, DeMarini DM, Martin BL, Thompson LC, Gilmour MI, Cascio WE, Hays MD. High-throughput video processing of heart rate responses in multiple wild-type embryonic Zebrafish per imaging field. Scientific reports 2019;9(1):1-4. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
McCullough SD, Bowers EC, On DM, Morgan DS, Dailey LA, Hines RN, Devlin RB, Diaz-Sanchez D. Baseline chromatin modification levels may predict interindividual variability in ozone-induced gene expression. Toxicological Sciences. 2015;150(1):216-24 |
CR835785 (2015) CR835785 (2017) CR835785 (2020) |
Exit |
|
|
McCullough SD, On DM, Bowers EC. Using Chromatin Immunoprecipitation in Toxicology:A Step‐by‐Step Guide to Increasing Efficiency, Reducing Variability, and Expanding Applications. Current protocols in toxicology 2017;72(1):3-14 |
CR835785 (2016) CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Miller DB, Snow SJ, Henriquez A, Schladweiler MC, Ledbetter AD, Richards JE, Andrews DL, Kodavanti UP. Systemic metabolic derangement, pulmonary effects, and insulin insufficiency following subchronic ozone exposure in rats. Toxicology and applied pharmacology 2016;306:47-57 |
CR835785 (2016) CR835785 (2017) CR835785 (2020) |
Exit Exit |
|
|
Mirowsky JE, Peltier RE, Lippmann M, Thurston G, Chen LC, Neas L, Diaz-Sanchez D, Laumbach R, Carter JD, Gordon T. Repeated measures of inflammation, blood pressure, and heart rate variability associated with traffic exposures in healthy adults. Environmental Health. 2015;14(1):66 |
CR835785 (2015) CR835785 (2020) |
Exit |
|
|
Mirowsky JE, Devlin RB, Diaz-Sanchez D, Cascio W, Grabich SC, Haynes C, Blach C, Hauser ER, Shah S, Kraus W, Olden K. A novel approach for measuring residential socioeconomic factors associated with cardiovascular and metabolic health. Journal of Exposure Science and Environmental Epidemiology 2017;27(3):281 |
CR835785 (2016) CR835785 (2020) |
Exit |
|
|
Perryman A, Kim H, Payton A, Rager J, McNell E, Rebuli M, Wells H, Almond M, Antinori J, Alexis N, Porter N, Jasters I. Plasma sterols and vitamin D are correlates and predictors of ozone-induced inflammation in the lung: A pilot study. PLOS ONE 2023;18(5):e285721 |
CR835785 (Final) |
Exit |
|
|
Walsh DM, McCullough SD, Yourstone S, Jones SW, Cairns BA, Jones CD, Jaspers I, Diaz-Sanchez D. Alterations in airway microbiota in patients with PaO2/FiO2 ratio≤ 300 after burn and inhalation injury. PloS one 2017;12(3):e0173848 |
CR835785 (2016) CR835785 (2020) |
Exit |
|
|
Zetlen H, Cao K, Schichlein K, Knight N, Maecker H, Nadeau K, Rebuli M, Rise M. Comparison of multiplexed protein analysis platforms for the detection of biomarkers in the nasal epithelial lining fluid of healthy subjects. JOURNAL OF IMMUNOLOGICAL METHODS 2023;517(113473). |
CR835785 (Final) |
Exit |
|
|
Thurston GD, Balmes JR, Garcia E, Gilliland FD, Rice MB, Schikowski T, Van Winkle LS, Annesi-Maesano I, Burchard EG, Carlsten C, Harkema JR. Outdoor Air Pollution and New-Onset Airway Disease. An Official American Thoracic Society Workshop Report. Annals of the American Thoracic Society 2020;17(4):387-98. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Bass V. San Antonio Oleic acid induces acute pulmonary injury and inflammation in vivo. Society of Toxicology, 2017;10(1):34 |
CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Miller DB, Karoly ED, Jones JC, Ward WO, Vallanat BD, Andrews DL, Schladweiler MC, Snow SJ, Bass VL, Richards JE, Ghio AJ. Inhaled ozone (O3)-induces changes in serum metabolomic and liver transcriptomic profiles in rats. Toxicology and applied pharmacology 2015;286(2):65-79. |
CR835785 (2017) CR835785 (2020) |
Exit Exit |
|
|
Pawlak EA, Noah TL, Zhou H, Chehrazi C, Robinette C, Diaz-Sanchez D, Müller L, Jaspers I. Diesel exposure suppresses natural killer cell function and resolution of eosinophil inflammation:a randomized controlled trial of exposure in allergic rhinitics. Particle and fibre toxicology. 2015;13(1):24 |
CR835785 (2015) CR835785 (2020) |
Exit |
|
|
Cascio WE, Gilmour MI, Peden DB. Ambient air pollution and increases in blood pressure:role for biological constituents of particulate matter. Hypertension. 2015;66(3):469-71. |
CR835785 (2015) CR835785 (2020) |
Exit |
|
|
Mirowsky J, Gordon T. Noninvasive effects measurements for air pollution human studies:methods, analysis, and implications. Journal of Exposure Science and Environmental Epidemiology. 2015;25(4):354 |
CR835785 (2015) CR835785 (2020) |
Exit |
|
|
Rebuli ME, Speen AM, Clapp PW, Jaspers I. Novel applications for a noninvasive sampling method of the nasal mucosa. American Journal of Physiology-Lung Cellular and Molecular Physiology 2016;312(2):L288-96 |
CR835785 (2016) CR835785 (2020) |
Exit |
|
|
Duran CG, Burbank AJ, Mills KH, Duckworth HR, Aleman MM, Kesic MJ, Peden DB, Pan Y, Zhou H, Hernandez ML. A proof-of-concept clinical study examining the NRF2 activator sulforaphane against neutrophilic airway inflammation. Respiratory Research 2016;17(1):89. |
CR835785 (2016) CR835785 (2020) |
Exit |
|
|
Mirowsky JE, Dailey LA, Devlin RB. Differential expression of pro-inflammatory and oxidative stress mediators induced by nitrogen dioxide and ozone in primary human bronchial epithelial cells. Inhalation toxicology. 2016;28(8):374-82 |
CR835785 (2015) CR835785 (2016) CR835785 (2020) |
Exit |
|
|
Snow SJ, Gordon CJ, Bass VL, Schladweiler MC, Ledbetter AD, Jarema KA, Phillips PM, Johnstone AF, Kodavanti UP. Age-related differences in pulmonary effects of acute and subchronic episodic ozone exposures in Brown Norway rats. Inhalation toxicology 2016;28(7):313-23. |
CR835785 (2017) CR835785 (2020) |
Exit Exit |
|
|
Dieffenderfer J, Goodell H, Mills S, McKnight M, Yao S, Lin F, Beppler E, Bent B, Lee B, Misra V, Zhu Y. Low-power wearable systems for continuous monitoring of environment and health for chronic respiratory disease. IEEE journal of biomedical and health informatics. 2016;20(5):1251-64. |
CR835785 (2015) CR835785 (2020) |
Exit |
|
|
Henriquez A, House J, Miller DB, Snow SJ, Fisher A, Ren H, Schladweiler MC, Ledbetter AD, Wright F, Kodavanti UP. Adrenal-derived stress hormones modulate ozone-induced lung injury and inflammation. Toxicology and applied pharmacology 2017;329:249-58 |
CR835785 (2016) CR835785 (2017) CR835785 (2020) |
Exit Exit |
|
|
Snow SJ, McGee MA, Henriquez A, Richards JE, Schladweiler MC, Ledbetter AD, Kodavanti UP. Respiratory effects and systemic stress response following acute acrolein inhalation in rats. Toxicological Sciences 2017;158(2):454-64 |
CR835785 (2016) CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Miller CN, Dye JA, Ledbetter AD, Schladweiler MC, Richards JH, Snow SJ, Wood CE, Henriquez AR, Thompson LC, Farraj AK, Hazari MS. Uterine artery flow and offspring growth in long-evans rats following maternal exposure to ozone during implantation. Environmental health perspectives 2017;125(12):127005 |
CR835785 (2017) CR835785 (2020) |
|
|
|
Mirowsky JE, Carraway MS, Dhingra R, Tong H, Neas L, Diaz-Sanchez D, Cascio W, Case M, Crooks J, Hauser ER, Dowdy ZE. Ozone exposure is associated with acute changes in inflammation, fibrinolysis, and endothelial cell function in coronary artery disease patients. Environmental Health 2017;16(1):126 |
CR835785 (2016) CR835785 (2020) |
Exit |
|
|
Zhou S, Behrooz L, Weitzman M, Pan G, Vilcassim R, Mirowsky JE, Breysee P, Rule A, Gordon T. Secondhand hookah smoke:an occupational hazard for hookah bar employees. Tobacco control. 2017;26(1):40-5 |
CR835785 (2015) CR835785 (2020) |
Exit |
|
|
Burbank AJ, Peden DB. Assessing the impact of air pollution on childhood asthma morbidity:how, when, and what to do. Current opinion in allergy and clinical immunology2018;18(2):124-31 |
CR835785 (2018) CR835785 (2020) |
Exit |
|
|
Henriquez AR, Snow SJ, Schladweiler MC, Miller CN, Dye JA, Ledbetter AD, Richards JE, Mauge-Lewis K, McGee MA, Kodavanti UP. Adrenergic and glucocorticoid receptor antagonists reduce ozone-induced lung injury and inflammation. Toxicology and applied pharmacology 2018;339:161-71 |
CR835785 (2017) CR835785 (2020) |
Exit Exit |
|
|
Snow SJ, Henriquez AR, Costa DL, Kodavanti UP. Neuroendocrine regulation of air pollution health effects:emerging insights. Toxicological Sciences 2018;164(1):9-20 |
CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Huang MC, Douillet C, Dover EN, Stýblo M. Prenatal arsenic exposure and dietary folate and methylcobalamin supplementation alter the metabolic phenotype of C57BL/6J mice in a sex-specific manner. Archives of toxicology 2018;92(6):1925-37 |
CR835785 (2017) CR835785 (2020) |
Exit Exit |
|
|
Hazari MS, Stratford KM, Krantz QT, King C, Krug J, Farraj AK, Gilmour MI. Comparative cardiopulmonary effects of particulate matter-and ozone-enhanced smog atmospheres in mice. Environmental science & technology 2018;52(5):3071-80. |
CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Stratford K, Haykal-Coates N, Thompson L, Krantz QT, King C, Krug J, Gilmour MI, Farraj A, Hazari M. Early-life persistent vitamin D deficiency alters cardiopulmonary responses to particulate matter-enhanced atmospheric smog in adult mice. Environmental science & technology 2018;52(5):3054-61. |
CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Snow SJ, Cheng WY, Henriquez A, Hodge M, Bass V, Nelson GM, Carswell G, Richards JE, Schladweiler MC, Ledbetter AD, Chorley B. Ozone-induced vascular contractility and pulmonary injury are differentially impacted by diets enriched with coconut oil, fish oil, and olive oil. Toxicological Sciences 2018;163(1):57-69 |
CR835785 (2017) CR835785 (2020) |
Exit |
|
|
Henriquez AR, Snow SJ, Schladweiler MC, Miller CN, Dye JA, Ledbetter AD, Richards JE, Hargrove MM, Williams WC, Kodavanti UP. Beta-2 adrenergic and glucocorticoid receptor agonists modulate ozone-induced pulmonary protein leakage and inflammation in healthy and adrenalectomized rats. Toxicological Sciences 2018;166(2):288-305. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Egorov AI, Converse R, Griffin SM, Styles J, Klein E, Sams E, Hudgens E, Wade TJ. Environmental risk factors for Toxoplasma gondii infections and the impact of latent infections on allostatic load in residents of Central North Carolina. BMC infectious diseases 2018;18(1):421. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Lyerly HK, Peden DB. Health and the Environment in North Carolina. North Carolina medical journal2018;79(5):302-5 |
CR835785 (2018) CR835785 (2020) |
Exit Exit Exit |
|
|
Peden DB. The Unexpected Health Effects of Air Pollution. North Carolina medical journal2018;79(5):309-11 |
CR835785 (2018) CR835785 (2020) |
Exit Exit |
|
|
Brooks JL, Berry DC, Currin EG, Ledford A, Knafl GJ, Fredrickson BL, Beeber LS, HAPPI Community Partnership Committee, Peden DB, Corbie‐Smith GM. A community‐engaged approach to investigate cardiovascular‐associated inflammation among American Indian women:A research protocol. Research in nursing & health201942(3):165-75 |
CR835785 (2018) CR835785 (2019) CR835785 (2020) |
Exit |
|
|
Fecho K, Pfaff E, Xu H, Champion J, Cox S, Stillwell L, Peden DB, Bizon C, Krishnamurthy A, Tropsha A, Ahalt SC. A novel approach for exposing and sharing clinical data:the Translator Integrated Clinical and Environmental Exposures Service. Journal of the American Medical Informatics Association 2019;26(10):1064-1073 |
CR835785 (2018) CR835785 (2020) |
Exit |
|
|
Bass VL, Snow S, Soukup J, Schladweiler M, Ghio A, Kodavanti U, Madden MC. 12-hydroxy oleic acid impairs endothelium-dependent vasorelaxation. Journal of Toxicology and Environmental Health, Part A 2019;82(5):383-6. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Cromar KR, Duncan BN, Bartonova A, Benedict K, Brauer M, Habre R, Hagler GS, Haynes JA, Khan S, Kilaru V, Liu Y. Air Pollution Monitoring for Health Research and Patient Care. An Official American Thoracic Society Workshop Report. Annals of the American Thoracic Society 2019;16(10):1207-14. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Miller CN, Kodavanti UP, Stewart EJ, Schaldweiler M, Richards JH, Ledbetter AD, Jarrell LT, Snow SJ, Henriquez AR, Farraj AK, Dye JA. Aspirin pre-treatment modulates ozone-induced fetal growth restriction and alterations in uterine blood flow in rats. Reproductive Toxicology 2019;83:63-72. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Stevens EL, Rosser F, Forno E, Peden D, Celedón JC. Can the effects of outdoor air pollution on asthma be mitigated?. Journal of Allergy and Clinical Immunology 2019;143(6):2016. |
CR835785 (2019) CR835785 (2020) |
Exit |
|
|
Corteselli EM, Gibbs-Flournoy E, Simmons SO, Bromberg P, Gold A, Samet JM. Long chain lipid hydroperoxides increase the glutathione redox potential through glutathione peroxidase 4. Biochimica et Biophysica Acta (BBA)-General Subjects 2019;1863(5):950-9. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Zhang XA, Yates A, Vasilevsky N, Gourdine JP, Callahan TJ, Carmody LC, Danis D, Joachimiak MP, Ravanmehr V, Pfaff ER, Champion J. Semantic integration of clinical laboratory tests from electronic health records for deep phenotyping and biomarker discovery. NPJ digital medicine 2019;2(1):1-9. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Hargrove MM, Kim YH, King C, Wood CE, Gilmour MI, Dye JA, Gavett SH. Smoldering and flaming biomass wood smoke inhibit respiratory responses in mice. Inhalation Toxicology 2019;31(6):236-47. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Sood AK, Burbank AJ, Lawson M, Zhou H, Wells HB, Peden DB, Hernandez ML. Systemic inflammatory response to inhaled endotoxin does not correlate with airway response. Respiratory research 2019;20(1):1-4. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
|
|
Kim YH, King C, Krantz T, Hargrove MM, George IJ, McGee J, Copeland L, Hays MD, Landis MS, Higuchi M, Gavett SH. The role of fuel type and combustion phase on the toxicity of biomass smoke following inhalation exposure in mice. Archives of toxicology 2019;93(6):1501-13. |
CR835785 (2019) CR835785 (2020) |
Exit Exit |
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2020 Progress Report
- 2019 Progress Report
- 2018 Progress Report
- 2016 Progress Report
- 2015 Progress Report
- Original Abstract
50 journal articles for this center