Grantee Research Project Results
2020 Progress Report: Practical PFAS Treatment with Sawdust
EPA Grant Number: SU839820Title: Practical PFAS Treatment with Sawdust
Investigators: Tu, Maobing , Lu, Mingming
Current Investigators: Tu, Maobing , Lu, Mingming , Hall, Cara , Gentry, Sincerrai , Nead, Carissa , Doll, Graci , Harrison, Canaan , Liu, Jinqi , Zhang, Yu
Institution: University of Cincinnati
EPA Project Officer: Page, Angela
Phase: I
Project Period: October 1, 2019 through September 30, 2020 (Extended to September 30, 2021)
Project Period Covered by this Report: October 1, 2019 through September 30,2020
Project Amount: $24,952
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2019) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
PFAS are a group of man-made chemicals that are extremely persistent in the environment. They have been considered to be emerging contaminants by U.S. Environmental Protection Agency (EPA) and pose potential threat to human health and the environment. The objective of this project is to develop a practical and cost-effective approach to treat PFAS in drinking water using functionalized sawdust (FS). This will improve the awareness of PFAS in our water systems and to encourage public participation.
The rationale of this research is cellulose-based sawdust would be much more cost-effective and practical than activated carbon (AC), commercial anion exchange resin or membrane filtration in removing PFAS from drinking water. The expensed sawdust is expected to be incinerated after use instead of the expensive regeneration process.
Progress Summary:
This project aims to develop a new functionalized sawdust anion exchange resin for PFAS removal and to develop new cost-effective treatment processes using FS. In this reporting period, the commercial sawdust has been functionalized by reaction with epichlorohydrin and dimethylamine. Fourier transform infrared spectroscopy (FTIR) was used to characterize the functional groups changes along with the functionalization reactions. It can be observed that functional groups (such as hydroxyl group) have been significantly changed after functionalization, which indicated the occurrence of functionalization reactions. The kinetic and isothermal adsorption experiments with FS have been performed and samples have been collected for liquid chromatography coupled to quadrupole time-of-flight mass spectrometry (LC-QTOF) analysis. The major output of the effort in the period is the functionalization of sawdust into biomass-based anion exchange resin. Undergraduate students (Carissa Nead, Graci Doll, Jinqi Liu, Sincerrai Gentry and Cara Hall) and graduate student (Yu Zhang) at University of Cincinnati have been recruited to working on the project in year 1. Due to coronavirus pandemic, the tasks 3 and 4 have been delayed and will be completed in year 2.
Figure 1. Synthesis of functionalized sawdust
The first task was to functionalize sawdust into biomass-based anion exchange resin. To achieve this, the commercial sawdust has been functionalized by reactions with epichlorohydrin and dimethylamine and 20 g of FS has been collected (Fig. 1). The products have been analyzed with FTIR to characterize the functional groups. The result showed that the intensity of the peak around 3500 cm-1 decreased from the original sawdust, which indicated a decrease of hydroxyl groups after functionalization. Besides, the increased intensity of 3000 cm-1 indicated the reacted amine group in the FS products. The FTIR result demonstrated that the functional groups (such as hydroxyl and amine group) have been significantly changed after functionalization, which indicated the occurrence of functionalization reactions.
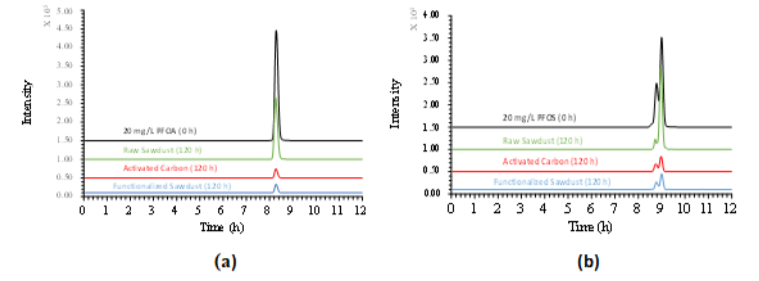
Figure 2: Adsorption of PFOA (a) and PFOS (b) with raw sawdust, AC and FS
The second task was to determine the adsorption kinetic and adsorption isotherm of PFOA and PFOS on FS. To achieve this, the adsorption kinetic experiments of PFOA and PFOS have been conducted with raw sawdust, commercial granular activated carbon (AC1240C), and FS. PFOA and PFOS were quantified by an Agilent LC-QTOF. It has been observed that FS, AC and raw sawdust adsorbed 93.07%, 92.41%, and 42.08% of PFOA, respectively (Fig. 2a). Similarly, FS, AC and raw sawdust also adsorbed 84.24%, 83.85%, and 36.61% PFOS, respectively (Fig. 2b). The results suggested that FS will be an excellent material to remove PFOA and PFOS in drinking water. The functionalized sawdust can help EPA to develop innovative methods to treat and manage FPAS in the drinking water and wastewater.
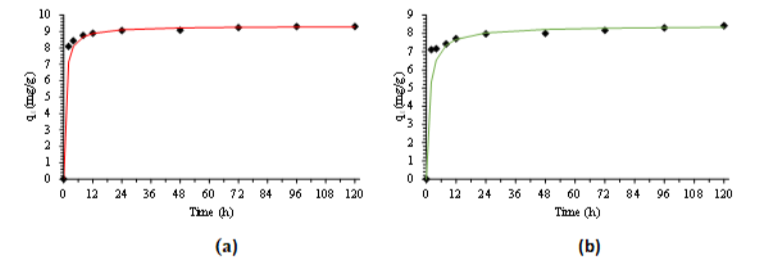
For the adsorption kinetics, the adsorption of PFOA and PFOS on FS was carried out in a batch process using a bottle-point technique. As showed in Fig. 3, the adsorption rate constant and initial adsorption rate were determined by fitting the experimental data to the pseudo second order reaction. The adsorption sorption rate constant of PFOA and PFOS is 0.1739 g/mg/h and 0.1022 g/mg/h respectively. The initial adsorption rate of PFOA and PFOS is 15.12 mg/g/h and 7.25 mg/g/h, respectively. The results suggested that the adsorption of PFOA and PFOS on FS was very fast and majority of adsorption can be completed within 2 h.
Figure 3: Adsorption kinetics fo PFOA (a) and PFOS (b) on FS and modeling using the pseudo-second-order equation
For the adsorption isotherm, series concentrations (e.g., 5, 10, 50, 100, 150, and 250 mg/L) of PFOA and PFOS solutions were mixed with 0.2 g FS. The bottles were maintained on the shaker (200 rpm) for 120 h. The samples have been stored at 4 ℃ until LC-QTOF analysis in year 2. The Task 3 was to assess PFOA and PFOS removal with FS column tests, the column test device has been designed and set up. It is expected the sampling running will be tested in year 2.
To ensure the quality of our data results, the FS synthesis experiments have been conducted and characterized with FTIR to confirm the reproducibility of sawdust functionalization process. The kinetic and isothermal adsorption experiments have been performed in duplicates. The PFAS analysis by LC-QTOF was carried out at the Environmental Analysis Center at University of Cincinnati. The standard PFAS samples have been used to build a standard curve for qualification measurement of PFAS water samples. The expenditures of this project has been comparable to the originally estimated.
Future Activities:
The experimental plan is on the right track. Task 3 and Task 4 will be completed in subsequent year. The project team will continue PFOA and PFOS sample analysis for adsorption isotherm, evaluation of PFOA and PFOS removal with FS column tests and determination of air emission from combustion of PFOA and PFOS containing FS. The detailed future activities will include:
-
PFOA and PFOS samples will be analyzed by LC/QTOF and adsorption isotherm of PFOA and PFOS on FS will be determined and evaluated.
-
Column test of PFAS removal in drinking water will be assessed and compared with GAC column test.
-
Preliminary test on combustion of PFOA and PFOS containing FS will be conducted and major products will be quantified.
Supplemental Keywords:
PFAS, anion exchange resin, functionalized sawdust, treatment, practical methods, wastewaterProgress and Final Reports:
Original AbstractP3 Phase II:
Practical PFAS Treatment with Functionalized Sawdust | 2022 Progress Report | 2023 Progress ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.