Grantee Research Project Results
2015 Progress Report: Vascular MAPs: Vascular and Neurovascular Tissue Models
EPA Grant Number: R835737C004Subproject: this is subproject number 004 , established and managed by the Center Director under grant R835737
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Human Models for Analysis of Pathways (H MAPs) Center
Center Director: Murphy, William L
Title: Vascular MAPs: Vascular and Neurovascular Tissue Models
Investigators: Sheibani, Nader
Institution: University of Wisconsin - Madison
EPA Project Officer: Aja, Hayley
Project Period: December 1, 2014 through November 30, 2018 (Extended to November 30, 2019)
Project Period Covered by this Report: December 1, 2014 through November 30,2015
RFA: Organotypic Culture Models for Predictive Toxicology Center (2013) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
1. Develop human models of vascular and neurovascular organotypic cultures.
2. Identify adverse outcome pathways associated with environmental toxins.
3. Define the critical elements for an organotypic response, critical cell types, and tissue architecture.
4. Leverage synthetic matrices and microscale systems to achieve robust tissue assembly and enable high throughput analysis.
5. Determine broad applicability of the model for translational research in predictive toxicology.
Progress Summary:
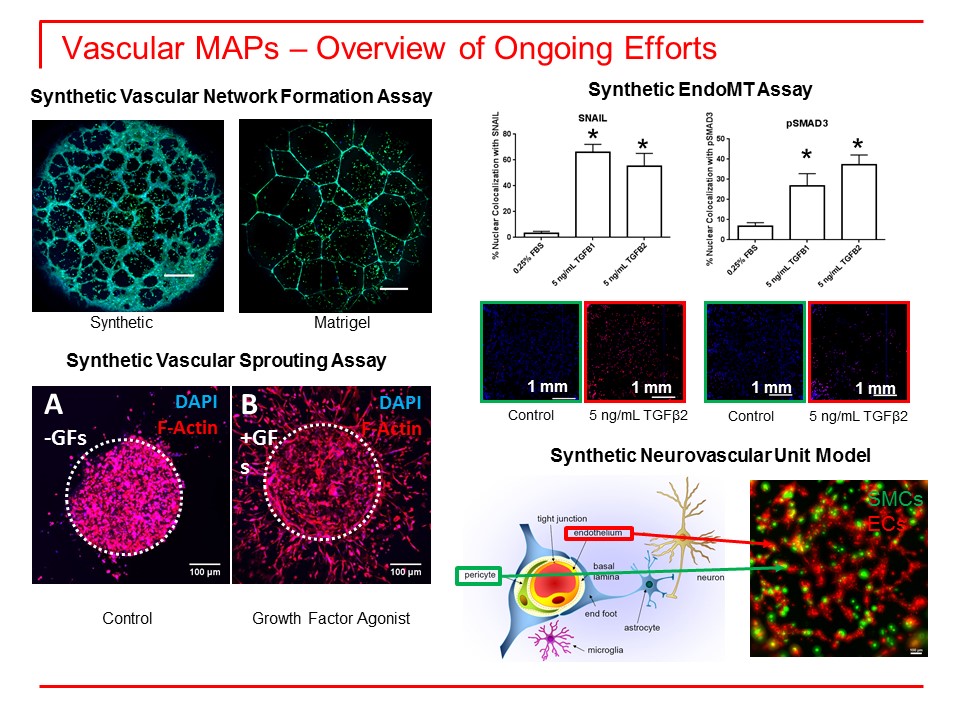
1. Develop human models of vascular and neurovascular organotypic cultures. We have made significant progress in this area. In collaboration with Dr. Murphy’s group we have adapted his synthetic material platform for capillary morphogenesis of human umbilical vein endothelial cells (HUVEC) to human induced pluripotent stem cells (iPSC)-derived microvascular endothelial cells (EC) (Figure 1). The superiority of our new synthetic matrix to commonly used material Matrigel also was confirmed. The complexity of Matrigel and its lot-lot variability results in significant experimental variations, which are almost impossible to control. Other modifications of our synthetic matrix modulating growth factor and cell adhesive signaling mechanisms provide additional flexibility for developing the most suitable platform for screening.
2. Identify adverse outcome pathways associated with environmental toxins. Using our new platform for capillary morphogenesis of iPSC-derived EC, we have begun to evaluate a small initial library of compounds from the U.S. Environmental Protection Agency (EPA) that previously were shown to have adverse effects on capillary morphogenesis of HUVECs on Matrigel. Our preliminary results demonstrated that the synthetic platform developed in Dr. Murphy’s lab is amenable to high throughput screening and can effectively identify chemicals that disrupt capillary morphogenesis in Matrigel, with accuracy and reproducibility superior to Matrigel.
3. Define the critical elements for an organotypic response, critical cell types, and tissue architecture. We have been developing/obtaining cultures of iPSC-derived perivascular supporting cells and astroglial cells, and begun to incorporate them into our EC culture systems. This will allow us to establish vascular networks that more closely resemble in vivo conditions for the identification of adverse effects on the vascular or neurovascular systems developed here. In addition, such systems will help to identify cells that are primary targets of vascular disruption.
4. Leverage synthetic matrices and microscale systems to achieve robust tissue assembly and enable high throughput analysis. Our future goals are to adapt our organotypic culture models with the micropatterning and microfluidic techniques developed in Dr. Beebe’s lab (H-MAP center investigator) in order to further expand our high throughput evaluations. Such systems will allow the study of the impact of flow as well as vascular permeability under various treatment modalities. We also will collect conditioned medium and cell lysates from these systems for biochemical and transcriptome analysis. Determining the production of various inflammatory mediators and investigating their paracrine and autocrine effects on various vascular cells will provide novel insight into regulatory pathways impacted by various chemical insults.
5. Determine broad applicability of the model for translational research in predictive toxicology. Our lab has developed various preclinical models of ocular angiogenesis, and these have been used to study adverse mechanisms that lead to pathological neovascularization, including ischemia, diabetes and inflammation. Identification of various classes of chemicals with potential anti-angiogenic activity using our screening platform may have clinical translational values for many diseases with a neovascular component and can be evaluated in these models.
Significance. The results of our studies demonstrate our ability to synthesize a well-defined synthetic matrix, which efficiently supports capillary morphogenesis of EC, which is amenable to various modifications impacting angiogenesis. Our ability to generate these platforms and include other vascular cellular components, including the perivascular supporting cells and astroglial cells, as well as their adaptation to micropatterning and microfluidic systems will provide novel insight to regulatory mechanisms that keep vascular integrity and function and how these are disrupted by various toxic chemicals with potential adverse effect on the vasculature and tissue/organ dysfunction.
Future Activities:
Our future plans will focus on further developing our 3D vascular network formation and incorporating other vascular cells. The contribution of various vascular cell components to the development, integrity, and function of the formed vasculature will enhance our knowledge regarding primary targets of agents that impact vasculature integrity and function.
Journal Articles on this Report : 4 Displayed | Download in RIS Format
| Other subproject views: | All 21 publications | 17 publications in selected types | All 17 journal articles |
|---|---|---|---|
| Other center views: | All 215 publications | 82 publications in selected types | All 81 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Belair DG, Schwartz MP, Knudsen T, Murphy WL. Human iPSC-derived endothelial cell sprouting assay in synthetic hydogel arrays. Acta Biomaterialia 2016;39:12-24. |
R835737 (2016) R835737 (2017) R835737C004 (2015) |
|
|
|
Belair DG, Miller MJ, Wang S, Darjatmoko SR, Binder BYK, Sheibani N, Murphy WL. Differential regulation of angiogenesis using degradable VEGF-binding microspheres. Biomaterials 2016;93:27-37. |
R835737 (2015) R835737 (2016) R835737 (2017) R835737C004 (2015) R835737C004 (2016) |
Exit |
|
|
Belair DG, Le NN, Murphy WL. Regulating VEGF signaling in platelet concentrates via specific VEGF sequestering. Biomaterials Science 2016;4(5):819-825. |
R835737 (2015) R835737 (2016) R835737 (2017) R835737C004 (2015) |
Exit |
|
|
Zanotelli MR, Ardalani H, Zhang J, Hou Z, Nguyen EH, Swanson S, Nguyen BK, Bolin J, Elwell A, Bischel LL, Xie AW, Stewart R, Beebe DJ, Thomson JA, Schwartz MP, Murphy WL. Stable engineered vascular networks from human induced pluripotent stem cell-derived endothelial cells cultured in synthetic hydrogels. Acta Biomaterialia 2016;35:32-41. |
R835737 (2015) R835737 (2016) R835737 (2017) R835737C001 (2017) R835737C004 (2015) |
|
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R835737 Human Models for Analysis of Pathways (H MAPs) Center Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R835737C001 Liver MAPs
R835737C002 Brain MAPs
R835737C003 Cancer MAPs: A 3D Organotypic Microfluidic Culture System to
Identify Chemicals that Impact Progression and Development of Breast Cancer
R835737C004 Vascular MAPs: Vascular and Neurovascular Tissue Models
R835737C005 Pathway Analysis Core
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
17 journal articles for this subproject
Main Center: R835737
215 publications for this center
81 journal articles for this center