Grantee Research Project Results
2016 Progress Report: Cancer MAPs: A 3D Organotypic Microfluidic Culture System to Identify Chemicals that Impact Progression and Development of Breast Cancer
EPA Grant Number: R835737C003Subproject: this is subproject number 003 , established and managed by the Center Director under grant R835737
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Human Models for Analysis of Pathways (H MAPs) Center
Center Director: Murphy, William L
Title: Cancer MAPs: A 3D Organotypic Microfluidic Culture System to Identify Chemicals that Impact Progression and Development of Breast Cancer
Investigators: Beebe, David
Current Investigators: Beebe, David , Murphy, William L
Institution: University of Wisconsin - Madison
EPA Project Officer: Aja, Hayley
Project Period: December 1, 2014 through November 30, 2018 (Extended to November 30, 2019)
Project Period Covered by this Report: December 1, 2015 through November 30,2016
RFA: Organotypic Culture Models for Predictive Toxicology Center (2013) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
To produce a 3D organotypic culture model of ductal breast cancer that is compatible with higher throughput screening and high-content screening approaches to discern toxic effects of chemical substances on breast cancer development and progression. The major goals and aims of this work have not changed since the initiation of this award.
Aim 1. Optimize and automate our microfluidic 3D in vitro ductal breast cancer model to be used for chemical library screening.
Aim 2. Develop an adverse outcome pathway-based model of estrogen-receptor (ER) mediated invasive ductal carcinoma (IDC) by utilizing quantitative physiological and molecular endpoints to identify key steps between the initiating event (estrogen-receptor ligand binding) and the adverse outcome (IDC) in our microfluidic platform.
Aim 3. Conduct high throughput screens using chemicals from the ToxCast library to identify chemicals that promote ER-mediated and non ER-mediated IDC.
Progress Summary:
We have made significant progress in the development of an organotypic breast cancer screening platform within the last year. Since our last update, we have developed, published, and added to AOP Wiki, the adverse outcome pathway (AOP) of ER-mediated breast cancer progression to identify readouts and cell types critical to disease progression (Morgan, et al., 2016). We originally proposed to use the AOP to help guide our platform and endpoint development and have continued that strategy. We have optimized AOP-based readouts that allow us to characterize ER-driven molecular (ER transactivation), cellular (proliferation and apoptosis) and tissue (ductal hyperplasia) level responses within our system.
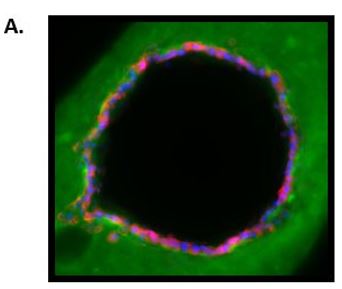
For endpoint optimization, we evaluated ER-driven readouts when a 3D collagen-embedded mammary ductal structure lined with MCF7 human breast cancer cells was exposed to a vehicle control (0.1% ethanol) or 100 nM of endogenous ER ligand 17-beta estradiol (E2). To evaluate the molecular initiating event ER activation, we utilized a MCF7-derived reporter cell line (MVLN) that was previously stably transfected with an estrogen response element (ERE)-luciferase plasmid. Here, we found that exposure to E2 increased ER transactivation in a concentration-dependent manner (EC50 = 40 nM). On a cellular level, we found that E2 increased the percentage of Ki67+ cells 2-fold and decreased the percentage of activated caspase-3/7 cells three-fold. We developed a technique to slice the ducts on a compresstome for histological analysis and observed the tissue level response, ductal hyperplasia in the estradiol-treated mammary ducts. These estrogen-driven responses agree with what has been previously described in vivo.
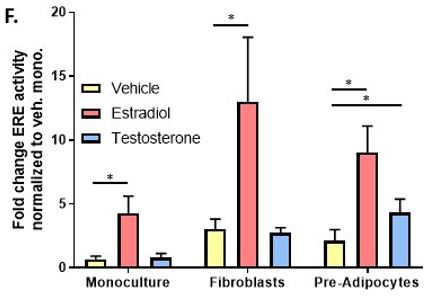
We also have incorporated other cell types of the breast microenvironment into our breast duct model. We evaluated pre-adipocytes and fibroblasts as the AOP identified them as important players in disease progression due to their aromatase production and regulation of estrogen receptor levels, respectively. We predict that chemical toxicants could alter stromal cell function, disrupt stromal-epithelial interactions, or undergo stromal mediated metabolism. Specifically, we hypothesized that mammary stromal cells would modulate breast epithelial response to ER agonists.
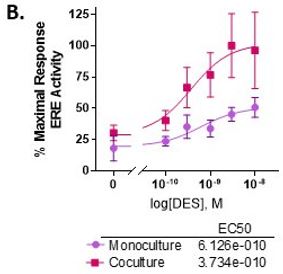
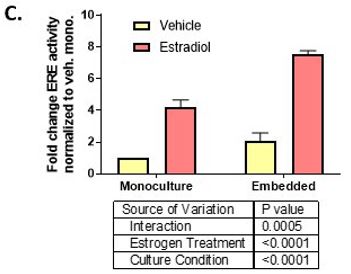
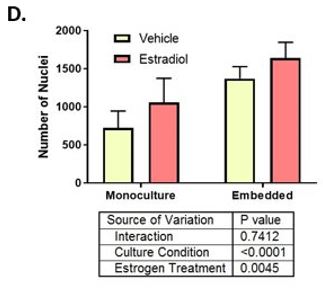
First, we included fibroblasts in the collagen I hydrogel and found the untreated breast ducts had increased ER activity and total cell number. Interestingly, the breast ducts were more sensitive to ER ligands E2 and diethylstilbestrol as indicated by a two-fold lower EC50. Additionally, maximal ER transactivation reached at ER saturation was two-fold higher in co-culture with stromal fibroblasts. On a cellular level, we found that epithelial cells cultured with fibroblasts had reduced apoptosis, which caused E2-induced ductal hyperplasia to occur at earlier time points. Interestingly, we observed no change in estrogen-induced proliferation between the two culture platforms.
Next, we set out to determine the importance of preadipocytes in the model. We exposed both monoculture and fibroblast co-culture to the E2 steroidogenesis precursor, testosterone (T). Here, we found no difference between the vehicle-treated and T-treated fibroblast co-cultures. However, when we replaced stromal fibroblasts with preadipocytes, exposure to T initiated ER-driven molecular, cellular, and tissue level responses. This suggests Cyp19 (aromatase) metabolism in the pre-adipocytes effectively converted T to E2. Altogether, these studies support our hypothesis that stromal cells influence response to ER ligands, and suggest that stromal cells may be important components to incorporate into in vitro breast cancer models.
Future Activities:
For future activities, we plan to conduct screens on chemicals from the Toxcast library and compare responses observed in our monoculture and co-culture systems to traditional in vitro platforms as well as to in vivo findings.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other subproject views: | All 6 publications | 6 publications in selected types | All 6 journal articles |
|---|---|---|---|
| Other center views: | All 215 publications | 82 publications in selected types | All 81 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Morgan MM, Johnson BP, Livingston MK, Schuler LA, Alarid ET, Sung KE, Beebe DJ. Personalized in vitro cancer models to predict therapeutic response:challenges and a framework for improvement. Pharmacology & Therapeutics 2016;165:79-92. |
R835737 (2016) R835737 (2017) R835737C003 (2015) R835737C003 (2016) |
Exit |
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R835737 Human Models for Analysis of Pathways (H MAPs) Center Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R835737C001 Liver MAPs
R835737C002 Brain MAPs
R835737C003 Cancer MAPs: A 3D Organotypic Microfluidic Culture System to
Identify Chemicals that Impact Progression and Development of Breast Cancer
R835737C004 Vascular MAPs: Vascular and Neurovascular Tissue Models
R835737C005 Pathway Analysis Core
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
6 journal articles for this subproject
Main Center: R835737
215 publications for this center
81 journal articles for this center