Grantee Research Project Results
2013 Progress Report: Toxicity Screening with Zebrafish Assay
EPA Grant Number: R835168Title: Toxicity Screening with Zebrafish Assay
Investigators: Tanguay, Robyn L.
Institution: Oregon State University
EPA Project Officer: Aja, Hayley
Project Period: May 1, 2012 through April 30, 2016
Project Period Covered by this Report: May 1, 2013 through April 30,2014
Project Amount: $1,199,999
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The objectives of our project have not changed and are:
- Expose zebrafish embryos to at least 1,000 chemicals.
- Study the morphology and behavior of the embryos to look for evidence of developmental toxicity.
- When an adverse effect occurs, conduct whole-transcriptome RNA sequencing studies to observe gene expression patterns, identify the initial biochemical target, and begin to elucidate the toxicity pathway.
- Conduct statistical modeling to compare the results of the zebrafish assay with the results of in vitro assays and other in vivo assays where they exist.
- Share our data with EPA and the broader research community.
Progress Summary:
In the second year of the grant, we focused on completing the analysis of the 24 hpf morphology data, refining bench mark dosing (BMD) analysis of morphology data, and completing all chemical screening.
- Drs. Tilton and Waters have completed the BMD analysis and have a draft of the manuscript that will be submitted to Toxicological Sciences after final team editing. The analysis in the manuscript is much more extensive as the ToxCast phase 2 in vitro data was made available for the cross species analysis.
- The primary results have been disseminated in several venues by the PI.
- Using the LEL “HITs” from the ToxCast phase 1 and phase 2, we established criteria for the selection of the 25 compounds for deep sequencing. We first mined the data for known (or suspected) endocrine disruption pathways, including estrogenic, androgenic, and thyroidogenic. The next criteria was to select compounds that did not produce significant mortality 24 hours postfertilization. This is critical because we have selected the 48 hpf time point for RNA sequencing analysis. The final criteria were an observed concentration response for at least one morphological endpoint. From these criteria, the chemicals have been selected for confirmation and RNA isolation (Table 1).
- For the identified 25 compounds, we contacted EPA to confirm chemical sourcing so we are assured that we are using the same chemical that was initially present in the provided talk Plates. All chemicals have been purchased, and the concentration response studies are ongoing to validate the responses. The goal prior to initiating the RNA sequencing studies is to identify more carefully the EC90 reach compound. This is critical as we are trying to identify gene expression changes that are associated with the endpoint. Since we are pooling embryos at a lower response rate, the variation in the displayed phenotypes would diminish gene expression signal and increase the noise. The exposures for gene expression analysis will begin in the third year of this project.
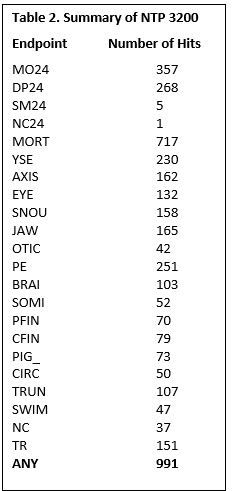
- We have completed the single concentration screening of the 3200 NTP compounds. We screened them all at 64 uM, and an N=24. Overall, 991 of the compounds were identified as bioactive. A breakdown of the individual responses is listed in Table 2. The key for the individual endpoints is: These data have been shared with NTP and for internal analysis. There are plans for additional follow up studies.
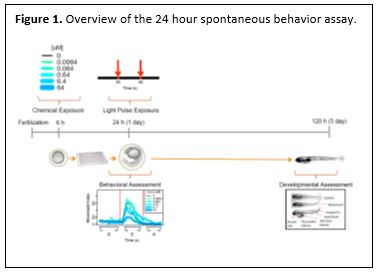
- As stated in the last report, in addition to the morphology phenotypic data, we also collected 24 hour behavioral (spontaneous motion) data for all of the embryos. The analysis of this data is complete, and the manuscript has been submitted. To summarize, we aimed to determine if a high-throughput screening (HTS) system using embryonic zebrafish can characterize chemical-elicited behavioral responses at an early stage (24 hours post-fertilization) that predict adverse developmental consequences. We describe a system where behavior can be both experimentally perturbed (concentration-response across 1060 ToxCast™ chemicals and short pulses of light) and measured (accompanying real-time, spontaneous movement) in a high-throughput fashion. By dividing the experimental interval according to light pulses (Background = prior to first pulse; Excitatory = after first pulse; Refractory = after second pulse), we observed movement responses that classified modulatory chemicals as eliciting light-independent movement alterations or light-dependent photomotor responses. Chemicals eliciting these behavioral responses were mapped against HTS in vitro results to identify specific molecular targets and neurosignalling pathways. As an integrative measure of normal development, significant alterations in movement highlighted active chemicals representing several modes of action. These early behavioral responses were predictive (Relative Risk p < 0.05) for each of 17 specific developmental abnormalities (including notochord defects) and mortality measured at 5 days-post-fertilization (dpf). Our major and exciting conclusion is that this system can provide rapid characterization of chemical-elicited behavioral responses at an early developmental stage that are indicative of significant downstream hazard. An image describing the experimental approach is provided in Figure 1.
- As mentioned in the previous report, we also collected concentration responses for each chemical and its impact on the 5 day light induced motor response. This data has been analyzed and we are in manuscript preparation mode. Initial results indicate that these assays suffer from much more variation and noise, but is useful as part of a screening level view of chemical bioactivity.
- We have modeled gene expression analysis results collected from 48 hour post fertilization animals in preparation of our Arnie seek studies. Using modeling we have determined that we need a minimum of 20 million individual reads in order to maximize data content, while minimizing cost. This manuscript is also under development and should be completed in 2014.
- We have conducted numerous RNA-sequencing studies over the past 2 years (unrelated to this project) and are beginning to be concerned with the experimental, rather than the biological variation issues. This has been reported by other groups. We are currently seeking potentially different platforms to collect this developmental gene expression data and may include exceptionally dense microarrays. We will decide soon whether or not we will pursue RNA sequencing or an alternative platform.
Future Activities:
-
Complete the BMD manuscript and widely disseminate the results.
-
Complete the 24 hpf spontaneous motion manuscript and widely disseminate the results.
-
Complete the validation studies to calculate EC90 for the selected 25 compounds.
-
Complete the re-exposure and RNA isolation for the gene expression analysis.
-
Complete comparative bioinformatic analysis and submit manuscripts.
Journal Articles:
No journal articles submitted with this report: View all 27 publications for this projectSupplemental Keywords:
Zebrafish, toxicity screening, high throughput screening, chemical toxicity, RNA sequencingProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.